2 May 2026 by Shahriar Lahouti.
CONTENTS
- Preface
- PART I: The foundation & systemic approach
- PART II: Lung abnormalities in chest CT
- Essential Lung Anatomy
- Pattern Recognition in Lung Disease
- Focal & Multifocal Opacities
- Atelectasis
- Consolidation
- Nodule: Size and Pattern-Based Approach
- Diffuse & Hazy Opacities
- Ground-Glass Opacity (GGO)
- Mosaic Attenuation
- Cystic & Cavitary Lesions
- Bronchiectasis & Bronchial Wall Thickening
- Linear, Reticular & Fibrotic Patterns
- Focal & Multifocal Opacities
- Spatial Distribution Of Disease
- 📊Rapid Pattern Recognition
- PART III: Putting it all together- Clinical scenarios
- Mediastinal Mass
- Media
- References
PART I: THE FOUNDATIONS & SYSTEMATIC APPROACH
Technical Essentials for the Clinician
For the emergency or critical care physician, a chest CT is not merely a stack of axial images—it is a dynamic, three-dimensional dataset rich with physiological clues. Effective interpretation begins before the scan, with an understanding of how the images were acquired. This knowledge directly impacts diagnostic confidence and the detection of life-threatening pathology.
Hounsfield Units
Every pixel on a CT image represents a precise measurement of tissue attenuation, quantified in Hounsfield Units (HU). This standardized scale is calibrated with water at 0 HU and air at -1000 HU. Understanding key HU ranges transforms qualitative impressions into objective assessments *:
- Fat: -50 to -100 HU
- Simple Fluid (Water): 0 to 20 HU
- Soft Tissue/Muscle: 40 to 60 HU
- Acute Blood/Hyperdense Fluid: 60 to 80+ HU
- IV Contrast: 150 to 300+ HU
- Calcium/Bone: 400 to 1000+ HU
Clinical Application: Actively check the HU of uncertain findings. This simple habit differentiates blood from water, fat from necrosis, and contrast enhancement from intrinsic hyperdensity. For example, a pericardial effusion measuring >40 HU suggests hemopericardium, while one measuring 0-10 HU is likely a simple transudate.
Contrast Timing & Protocols
“The Phase Tells the Story”
◾️Contrast Timing is Physiology: The phase of contrast enhancement is critical.
- Pulmonary Arterial Phase (25-30 sec delay) *
- Optimal for detecting pulmonary embolism. Pulmonary arteries are brightly opacified, while systemic arteries and the left heart remain relatively faint.
- Systemic (Aortic) Phase (60-70 sec delay) *
- The standard “routine” chest CT. The aorta, coronary arteries, and left heart chambers are fully enhanced. This phase is best suited for evaluating aortic pathology, cardiac chambers, and mediastinal structures; however, it is suboptimal for detecting small pulmonary emboli.
- Recognizing the Phase
- Quickly assess the left atrium and ventricle. If they are near water density, you are in an early (pulmonary) phase. If they are brightly enhanced, you are in a systemic phase. This dictates your search pattern.
Contrast Protocols & Timing: What the Clinician Must Know
Quick reference for interpreting CT phase, injection rates, and common pitfalls in critical care imaging.
| Protocol | Injection Rate | Scan Delay | Optimal Visualization | Phase & Indicator | Pitfalls & Clinical Notes |
|---|---|---|---|---|---|
|
PE Protocol (CTPA) |
4–5 mL/s | 25–30 sec | Pulmonary arteries, RV, central clots |
Early (Pulmonary) Phase
PA bright, LV dark
|
LV may appear “dark” – do not mistake for thrombus.
Suboptimal for aortic or cardiac chamber assessment.
|
|
Coronary CTA (CTCA) |
5–6 mL/s | Bolus-tracking from aorta | Coronary arteries, coronary plaque, bypass grafts |
Late Systemic Phase
Coronaries bright, LV bright
ECG-GATED REQUIRED
|
Not an emergency study – requires beta-blockers for heart rate control.
Suboptimal for PE detection. Best for stable chest pain evaluation.
|
| Routine Chest | 2–3 mL/s | 60–70 sec | Heart chambers, aorta, mediastinum, lung parenchyma |
Systemic (Aortic) Phase
Aorta & LV bright
|
Misses small PEs – arteries may be suboptimally opacified.
Best for cardiac contours, pericardium, and mediastinal masses.
|
| Trauma / Aortic | 4–5 mL/s | Bolus-tracking from aorta | Aortic injury, active bleeding, great vessels |
Early Systemic Phase
Arteries maximally enhanced
|
May miss venous or delayed bleeding.
High contrast dose – consider renal function.
|
| Non-Contrast | N/A | N/A | Calcifications, hemorrhage, lung windows only | No contrast phase |
Cannot assess vessels or perfusion.
Useful for follow-up nodules, renal-impaired patients.
|
Key Differences: CTPA vs. CTCA
- CTPA: For PE detection; early phase; pulmonary arteries bright; LV dark; no ECG gating needed.
- CTCA: For coronary assessment; late phase; coronaries bright; ECG gating required; not for emergency PE evaluation.
- Critical: Dark LV on CTPA is normal (not thrombus). Unopacified coronaries on routine CT ≠ coronary disease.
Windows & Level are Your Diagnostic Filters
◾️Raw CT data is neutral. The “window” (width) and “level” (center) settings control which densities are visualized.
- Lung Windows (W: 1500, L: -600) *
- Essential for parenchyma, but makes everything else disappear. Do not assess mediastinum, vessels, or soft tissues here.
- Mediastinal/Soft Tissue Windows (W: 350-400, L: 40-50) *
- Your primary window for the heart, great vessels, lymph nodes, and pleural soft tissues. Evaluate all non-parenchymal findings here.
- Bone Windows (W: 2000, L: 300) *
- For detecting subtle fractures, bone destruction, or calcifications (e.g., in coronary arteries, pericardium, or cardiac valves).
Reconstructions are your 3D Map
◾️Modern PACS workstations allow you to scroll in any plane.
- Coronal & Sagittal Reconstructions
- Instantly clarify anatomy. The coronal view is invaluable for assessing the central pulmonary arteries, tracheobronchial tree, and craniocaudal extent of disease. The sagittal view is key for the aorta, spine, and anterior/posterior compartments *.
- Multiplanar Reformation (MPR)
- A non-negotiable tool for tracing vessels (e.g., confirming a PE, evaluating the coronary artery course) and assessing complex structures like the cardiac valves or aortic root. If you see something odd on axial slices, re-slice through it in another plane *.
◾️The Takeaway: You are not a passive viewer of pre-set images. Actively use windowing and multiplanar reconstructions to interrogate the scan. Knowing the contrast phase frames your diagnostic priorities, ensuring you do not miss a suboptimally seen PE or misinterpret an unopacified left atrium as a mass.
Cardiac Anatomy at a Glance
◾️Background
- The heart is included in all chest imaging and should be systematically evaluated in patients presenting to the emergency department, particularly if a noncardiac cause of the patient’s symptoms is not identified *.
- When reviewing the heart on a routine chest CT, a systematic check of cardiovascular structures can reveal incidental but critical findings.
- A systematic review of cardiac anatomy prevents misinterpretation of normal structures as pathology. For the emergency physician, a pattern-based approach is more practical than memorizing every anatomical detail.
- Know the normal pattern to spot the abnormal. Evaluate the heart from outside to inside (pericardium → coronaries → myocardium → chambers → valves) or vice versa if necessary, catches 95% of critical findings in under 30 seconds *.
- Structures Enhanced by Contrast *
- Coronary arteries (courses visible, calcification evident)
- Valves (aortic, mitral, tricuspid)
- Papillary muscles
- Interatrial and interventricular septa
- Chamber borders and contours
- The 30-Second Systematic Scan (Outside → In)
- Pericardium: Effusion? Thickening?
- Normal pericardium appears as a thin (≤2 mm), non-enhancing line. Circumferential effusion >1 cm or high attenuation (>40 HU) suggests hemopericardium.
- Coronaries: Heavy calcification? Anomalous course?
- Myocardium: Thick/thin? Hypodense? RV strain?
- Chambers: Size? Contour? Filling defects?
- RV should be smaller than the LV. An RV: LV diameter ratio > 0.9 suggests right heart strain.
- The interventricular septum (IVS) normally bows toward the RV. Reversal (bowing leftward) indicates pressure overload.
- RA and LA should be similar in size. LA enlargement (≥45 mm AP diameter) is associated with atrial fibrillation and pulmonary hypertension.
- Valves: Calcified? Masses?
- Pericardium: Effusion? Thickening?
- Key Normal Variants:
- Crista terminalis (CrT) appears as a smooth, linear filling defect along the posterior RA wall—do not mistake it for thrombus.
- Papillary muscles (PM) and left atrial appendage (LAA) should be smooth-contoured without filling defects.
- When to Use Reformats *
- Coronal: Assess craniocaudal chamber relationships
- Sagittal: Evaluate the aortic root and outflow tracts
- Oblique: Trace coronary courses or valve planes
💡Clinical Pearls for the ED:
- Dark left ventricle on early-phase CT = normal (not thrombus)
- Crista terminalis (RA ridge) = normal, not mass/thrombus
- Pericardial recess fluid = normal, not lymphadenopathy
- RV should be smaller than LV (RV: LV ratio ≤0.9)
The 60-Second Systematic Scan: A Survival Checklist
In the resuscitation bay or ICU, time is the most critical resource. A structured, reproducible approach prevents cognitive overload and ensures life-threatening pathology is identified first. This checklist is designed to be performed in under a minute, moving from the most immediate threats to a rapid survey of major systems.
🧠The Philosophy: “Catastrophe First, Context After.”
Do not get distracted by the most obvious finding (e.g., a large consolidation) until you have ruled out conditions that will kill the patient in the next hour. Follow this order relentlessly.
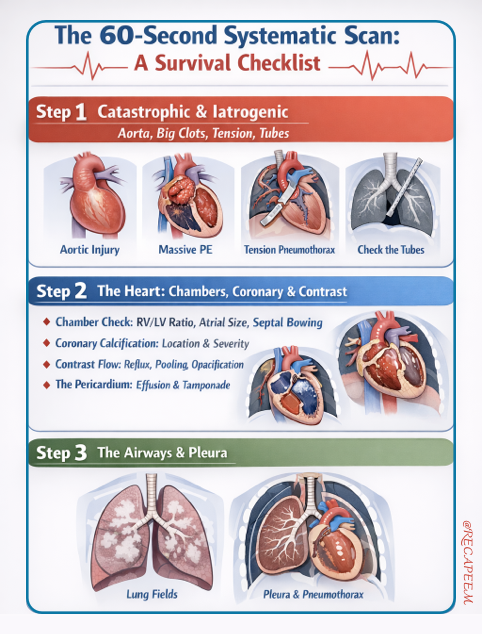
⚠️ Step 1 – Catastrophic & Iatrogenic
This step should take 15–20 seconds. Use soft-tissue windows (W:350-400, L:40-50)
- Aorta: Trace it from arch to diaphragm.
- Look for Dissection flap (linear internal density), intramural hematoma (crescentic high-attenuation wall thickening), rupture (periaortic hematoma, active contrast extravasation).
- Central Pulmonary Arteries: Check the main, right, and left pulmonary arteries.
- Look for a large filling defect (saddle PE), abrupt cutoff, and massively dilated main pulmonary artery (MPA).
- Tension Physiology:
- Mediastinal Shift: Is the trachea/heart pushed to one side?
- Diaphragmatic Inversion: Especially on the left.
- Contralateral Lung Compression: Suggests large tension pneumothorax or massive effusion.
- Tubes & Lines: Confirm placement and identify complications.
- Endotracheal Tube: Tip 3-5 cm above carina.
- Central Lines: Tip at SVC/RA junction.
- 👉Exclude pneumothorax.
- Chest Tubes: Positioned in the pleural space, not the fissure or abdomen.
- NG/OG Tubes: Course through the esophagus into the stomach.
- 👉Exclude bronchial placement.
⚠️ Step 1 – Catastrophic & Iatrogenic Scan
Soft-tissue windows (W:350-400, L:40-50) | Time: 15-20 seconds| Anatomical Target | Critical Findings to Exclude | Immediate Action / Clinical Pearl |
|---|---|---|
|
Aorta & Great Vessels
EMERGENT
Trace from arch to diaphragm
|
|
|
|
Central Pulmonary Arteries
URGENT
Main, right, and left pulmonary arteries
|
|
|
|
Tension Physiology
EMERGENT
Assess mediastinal symmetry
|
|
|
|
Tubes & Lines
CRITICAL
Use scout topogram for overview
|
|
|
Step 2 – The Heart and Large Vessels
This step integrates the “Incidental but Critical” findings (integrate soft-tissue and bone window). * *
Cardiac Chambers
◾️Right ventricular (RV) dilation
- RV: LV transverse diameter
- On non–ECG-gated multidetector chest CT, right ventricular assessment is most commonly performed by comparing the transverse RV diameter with that of the left ventricle (LV) *.
- Measure the maximal dimensions of both ventricles.
- Often requires assessment across multiple CT slices.
- ⎜A normal RV: LV ratio is ≤0.9. RV > LV size suggests RV dilatation
- Limitation: reduced sensitivity in patients with pre-existing LV enlargement *.
- Importantly, this ratio does not represent an absolute measure of RV size; rather, it serves as a surrogate marker of RV dysfunction.
- RV > LV suggests acute pressure overload (e.g., massive PE) or chronic pulmonary hypertension.
- On non–ECG-gated multidetector chest CT, right ventricular assessment is most commonly performed by comparing the transverse RV diameter with that of the left ventricle (LV) *.
- Transverse diameter
- Diameter measured in a plane perpendicular to the septum.
- An RV transverse diameter ≥60 mm in men or ≥57 mm in women demonstrates moderate sensitivity (63–67%) and high specificity (91–94%) for identifying right ventricular enlargement (RVE).
- Interventricular septal bowing
- Bowing toward the LV indicates RV pressure overload
- Seen in acute pulmonary embolism and chronic pulmonary hypertension
◾️Right atrial (RA) dilation
- Right atrial (RA) enlargement reflects chronic right-sided pressure or volume overload and can support the diagnosis of pulmonary hypertension or advanced RV dysfunction.
- Maximal RA transverse diameter:
- ⎮64 mm in women, >67 mm in men
- Measured as the largest diameter parallel to the tricuspid valve plane
- Exclude the right atrial appendage and the coronary sinus
- Maximal RA transverse diameter:
- Performance:
- Sensitivity ~65%
- Specificity ~92%
◾️Left atrial enlargement (LAE)
- Left atrial enlargement is a well-established prognostic marker associated with atrial fibrillation, stroke, heart failure, myocardial infarction, and pulmonary hypertension.
- LAE is an independent risk factor for new-onset atrial fibrillation/flutter and is linked to anticoagulation failure and post-ablation recurrence.
- LA size also correlates with elevated pulmonary capillary wedge pressure and has been associated with pulmonary conditions, including obstructive sleep apnea.
- CT Assessment of LA Size (Non–ECG-Gated)
- Anteroposterior (AP) diameter
- Measured on axial images at the level of the aortic root
- Exclude the LA appendage and pulmonary veins
- LAE thresholds:
- ≥45 mm (general)
- ≥45 mm (women), ≥50 mm (men)
- Performance: moderate sensitivity (~45–55%), high specificity (~92–96%)
- Good interobserver agreement
- Transverse diameter
- Single axial measurement
- Threshold: >73 mm
- Performance: sensitivity and specificity ~84%
- May outperform AP diameter for sensitivity but with greater variability
- Maximal axial cross-sectional area (LA-MACSA)
- Largest axial LA area between the LV outflow tract and the mitral valve level
- Excludes LA appendage and pulmonary veins
- Thresholds:
- 2400 mm² (high specificity for post-capillary PH)
- 2000 mm² with normal RV size strongly suggests group 2 PH
-
- Higher interobserver agreement than linear measurements.
- Limitations
- Linear LA measurements do not directly reflect LA volume
- Accuracy is influenced by sex, body surface area, and the reference standard
- Volume-based standards (MRI or ECG-gated CT) remain the reference, but are often unavailable in acute care CT
◾️Left Ventricle Enlargement (LVE)
- Left ventricular enlargement is associated with cardiomyopathy, myocardial infarction, valvular disease, pulmonary hypertension, and heart failure.
- LVE is a modifiable risk factor for myocardial infarction, stroke, and sudden cardiac death, making early detection clinically important—even in asymptomatic patients.
- CT Assessment of LV Size (Non–ECG-Gated)
- Transverse diameter technique
- Measured as the maximal short-axis intraluminal diameter between the septal and lateral walls (from inner wall to inner wall ), perpendicular to the LV long axis.
- Typically obtained at the level of the papillary muscles
- ⎮Transverse diameter >55 mm (in females) or >60 mm (males).
- Sensitivity is moderate (~60%), and specificity seems to be high (>95%)
- Good interobserver agreement on contrast-enhanced studies
- Limitations
- Thresholds vary due to differing reference standards (TTE vs cardiac MRI)
- MRI-based standards identify larger LV volumes than TTE
- Most studies do not index LV size to body surface area, age, or sex
- Non-contrast CT may limit endocardial definition.
- Transverse diameter technique
◾️Intracardiac masses
- A variety of intracardiac masses can be seen, often presenting as filling defects within the cardiac chambers.
- Thrombus
- Most common intracavitary mass
- Typical locations:
- LV apex (post-infarction, aneurysm)
- LA appendage (atrial fibrillation)
- RA/RV (clot-in-transit for PE)
- CT appearance: Low-attenuation filling defect, often peripheral, may show rim enhancement
- 🔎Thrombus Mimics (Must Exclude)
- Chiari network: Web-like RA structure (benign)
- Crista terminalis: Smooth RA ridge
- Lipomatous hypertrophy: Fat density in the interatrial septum, sparing the fossa ovalis.
- Tumors
- Metastatic (most common cardiac tumor): Lung, breast, lymphoma, melanoma
- CT patterns: Pericardial infiltration with effusion, myocardial nodules, and cavitary masses (especially renal cell, hepatocellular).
- Primary cardiac tumors (20–40× less common): Myxoma (most common primary), sarcoma, lymphoma
- CT clues: Enhancement pattern, multiplicity, invasion, extracardiac primary
- Metastatic (most common cardiac tumor): Lung, breast, lymphoma, melanoma
- Thrombus
💡Key Differentiators:
- Thrombus: Hounsfield units 20–40 HU, no enhancement, associated PE/DVT.
- Clot-in-transit: Mobile, traverses chambers, associated with large central PE.
- Tumor: Variable HU, often enhances, pedunculated or invasive.
- Artifact: Contrast mixing (swirl), incomplete opacification.
Myocardium
The normal left ventricular myocardium is of uniform thickness and enhances homogeneously on postcontrast imaging (right figure below).
◾️Left Ventricular Wall Thickness
- Left ventricular hypertrophy (LVH) reflects increased LV mass and is classically assessed using end-diastolic measurements of LV cavity size and wall thickness. On non–ECG-gated CT, LV wall thickness may be overestimated, as images may not coincide with true end diastole.
- Suggestive CT finding:
- LV free wall or interventricular septal thickness >2.0–2.5 cm on axial images
- Important considerations:
- CT is not definitive for LVH diagnosis
- Findings should prompt further evaluation with echocardiography or cardiac MRI
- 💡When LVH is suspected, assess for underlying causes:
- Aortic or mitral valve disease
- Aortic coarctation
- Left-to-right shunt
◾️Right ventricular wall thickness
- The normal right ventricular (RV) free wall is thin, typically ≤3 mm—and often barely perceptible on CT imaging. Thickening of the RV myocardium reflects chronic pressure overload rather than acute pathology.
- Normal RV wall thickness: ≤3 mm
- Often difficult to appreciate on routine CT
- Abnormal finding:
- RV outflow tract wall thickness >6 mm
- Suggests chronic pulmonary hypertension rather than acute pulmonary embolism.
- Normal RV wall thickness: ≤3 mm
◾️Myocardial Infarction: Acute vs. Chronic CT Findings
- Acute Myocardial Infarction
- Key Finding: Regional myocardial hypoattenuation (perfusion defect)
- Wall Thickness: Preserved (no thinning)
- Attenuation: Hypodense relative to normal myocardium
- Distribution: Follows the coronary artery territory
- Associated Findings:
- ± Intracavitary thrombus
- ± Wall motion abnormality
- Coronary artery calcification may be present
- Pitfall: A beam-hardening artifact can mimic a perfusion defect.
- Chronic (Remote) Myocardial Infarction
- Key Findings:
- Myocardial thinning (<5 mm typical)
- Subendocardial fat deposition (fat metaplasia, -50 to -100 HU)
- Dystrophic calcification (linear or punctate)
- Wall Morphology: Often bulging (true aneurysm)
- Complications:
- Ventricular aneurysm formation
- Mural thrombus (common)
- Arrhythmogenic substrate
- Note: Fat metaplasia is pathognomonic for chronic infarction
- Key Findings:
Coronary arteries
◾️Background
- While coronary luminal stenosis cannot be reliably assessed without ECG gating, non-gated CT provides valuable anatomical and morphological information.
- For more on the anatomy of the coronary arteries in nongated chest CT, see here.
◾️Key visible findings
- Coronary artery courses – Right coronary artery (RCA), left main (LM), left anterior descending (LAD), and circumflex (LCx) can be traced
- Coronary calcification – Readily detected and qualitatively assessed on both non-contrast and contrast-enhanced studies
- Aneurysms & bypass grafts – Focal dilatations or surgical grafts may be visualized
- Anomalous origins/courses – Variant anatomy may be incidentally detected
◾️Limitations
- Motion artifact may obscure segments, especially mid/distal vessels
- No functional assessment – Cannot determine hemodynamic significance
- Optimal evaluation requires ECG-gated CTA for stenosis quantification
This section focuses on coronary calcification assessment; coronary anomalies are discussed separately.
◾️Coronary artery calcification (CAC)
- CAC is not diagnostic of clinical coronary artery disease. However, coronary artery calcification may be used as a risk-stratification tool.
- Absence of any calcium suggests a low risk of coronary artery disease.
- Coronary artery calcification is frequently seen with increasing age (e.g., present in 90% of men and 70% of women by age 70).
- Substantial calcification in younger patients may suggest premature coronary artery disease (e.g., men <55 years old, or women <65 years old).
- Coronary artery calcification (CAC) Screen: Switch to bone windows for 3 seconds.
- Location *
- Heavy calcification in the left main or proximal LAD carries the highest acute coronary risk.
- Severity
- “Heavy” calcification (dense, extensive) suggests significant underlying coronary disease, critical for pre-operative risk stratification or in a patient with undifferentiated chest pain. Severity may be defined qualitatively (figure below) *:
- Mild: Isolated flecks of calcification.
- Moderate: Intermediate severity.
- Severe: Continuous calcification.
- “Heavy” calcification (dense, extensive) suggests significant underlying coronary disease, critical for pre-operative risk stratification or in a patient with undifferentiated chest pain. Severity may be defined qualitatively (figure below) *:
- Contrast-enhanced CT scan has a slightly reduced sensitivity for coronary artery calcification, but overall it remains very good (83% sensitive), especially for more severe calcification.
- Location *
Cardiac Valves
Although motion often compromises the evaluation of cardiac valves at nongated CT, abnormalities such as calcification, valve thickening, or even congenital diseases such as a bicuspid valve are often identifiable. Occasionally, valve vegetations or tumors can be detected at nongated CT.
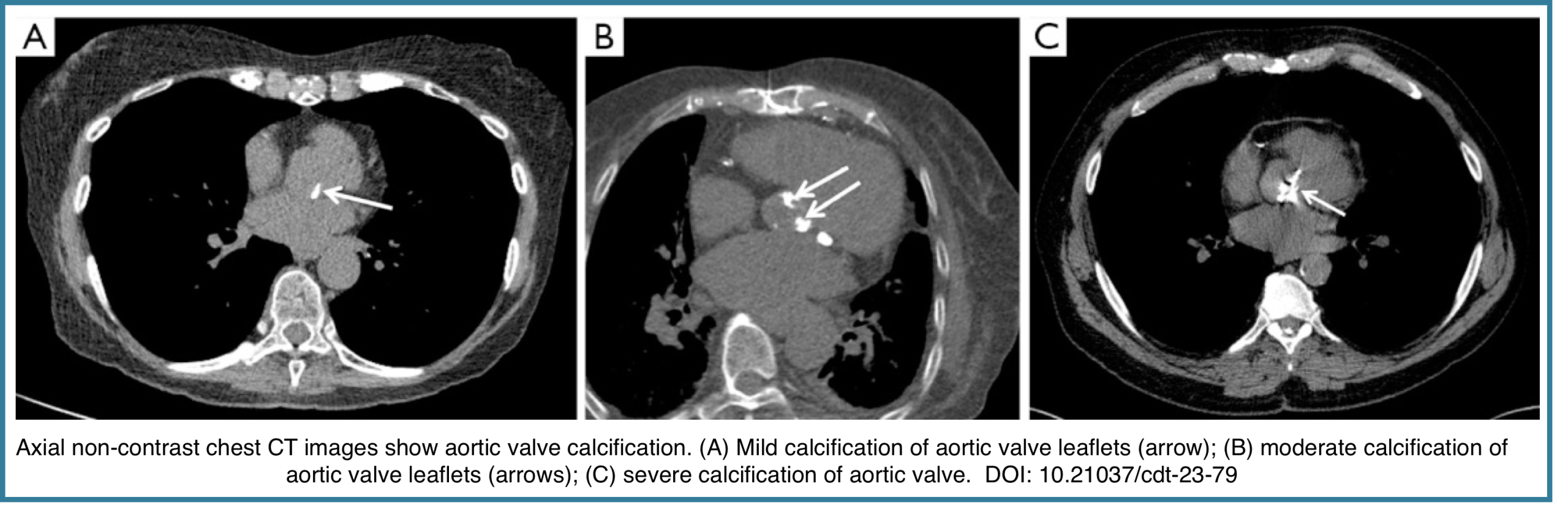
◾️Aortic Valve Calcification (AVC)
- Aortic valve calcification should be distinguished from calcification of the aortic root (which spares the valve leaflets), mitral annulus, and coronary arteries. AVC is a common incidental CT finding, present in up to ~20% of scans, with increasing prevalence in older patients *.
- Qualitative CT severity grading:
- Mild: Small, discrete calcified foci
- Moderate: Multiple, larger calcified foci
- Severe: Extensive, confluent leaflet calcification
- Clinical significance
- Moderate–severe AVC increases the likelihood of aortic stenosis.
- AVC is a marker of systemic atherosclerotic disease.
- Associated CT findings suggestive of aortic stenosis:
- Left ventricular hypertrophy
- Left atrial dilatation
- Post-stenotic dilatation of the ascending aorta (>4.1 cm)
◾️Mitral Annulus Calcification
- Prevalence and Grading
- Mitral annular calcification (MAC) is a common incidental finding on chest CT, present in approximately 8% of scans, significantly more frequently than isolated mitral leaflet calcification.
- Qualitative Grading Scale: While precise quantification can be challenging on non-gated CT, MAC can be graded based on the extent of annular involvement
- Mild: Involves < ⅓ of the annular circumference.
- Moderate: Involves ⅓ – ½ of the annular circumference.
- Severe: Involves > ½ of the annular circumference.
- Clinical Significance:
- MAC is often asymptomatic but serves as a marker of chronic cardiovascular stress and atherosclerosis. Its presence is independently associated with an increased risk of *:
- Coronary artery disease
- Atrial fibrillation
- Stroke
- Cardiovascular mortality
- MAC is often asymptomatic but serves as a marker of chronic cardiovascular stress and atherosclerosis. Its presence is independently associated with an increased risk of *:
- Note for the Clinician: When reporting MAC, specify its severity and note its association with underlying cardiovascular disease. In the context of systemic embolization or unexplained stroke, consider MAC as a potential source, though this is less common than left atrial appendage thrombus.
◾️Mitral Leaflet Calcification
- Mitral Leaflet Calcification, distinct from annular calcification, is less common and often associated with rheumatic heart disease or advanced renal failure.
- It directly correlates with mitral valve sclerosis or stenosis and should prompt echocardiographic evaluation for hemodynamically significant valve disease.
- Notes
- It is subtle and limited to the leaflet tips, whereas mitral annular calcification can be extensive, usually demonstrating a curvilinear morphology in the posterior and outer ring of the valve
- It is also important not to mistake mitral annular calcification for calcification in the left circumflex coronary artery
Mitral Calcification: Annular vs. Leaflet
| Feature | Mitral Annular Calcification (MAC) | Mitral Leaflet Calcification |
|---|---|---|
| Location | C-shaped ring at base of valve | Mobile portions of leaflets |
| Prevalence | ~8% of chest CTs Common |
Less common Uncommon |
| Associations |
|
|
| Significance | Cardiovascular risk marker | Structural valve disease |
| CT Appearance | Curvilinear in AV groove | Nodular in valve orifice |
| Next Step | CV risk assessment | Echo for valve function |
Pericardium
◾️Background
- Anatomy: The normal pericardium is 3 mm or less in thickness and does not show noticeable enhancement. For more on normal epicardial spaces, see here.
- CT provides superior anatomical characterization of pericardial pathology, complementing echocardiography’s functional assessment.
- While echo remains first-line for tamponade evaluation, CT excels in detecting loculated effusions, characterizing fluid composition via HU values, and identifying calcifications.
◾️Pericardial Findings on Chest CT
- Epicardial Fat vs. Pericardial Effusion
-
- Epicardial fat: -50 to -150 HU (fat density)
- Pericardial effusion: 0–80+ HU (fluid to blood density)Differentiation is based on attenuation (HU):
-
- CT advantage: Objective HU measurement and multiplanar visualization provide definitive differentiation, eliminating echo’s acoustic limitations.
- Pericarditis
- Imaging signs: Effusion, pericardial thickening (>2 mm), and enhancement of the pericardial layer. Fat stranding
- Chronicity: Calcification suggests chronic inflammation
- Note: May be dry (without effusion) or infectious (rarely with gas)
- Pericardial Effusion
- Generally, effusions of >1 cm of circumferential fluid are significant.
- Simple/transudative effusion:
- Pure or near water density HU of <20, without or with regular slight enhancement.
- Exudative/Purulent:
- HU 20–40, often with enhancing and irregularly thickened pericardium.
- Hemorrhagic/hematoma:
- A denser pericardial effusion with CT attenuation values of HU 40–80; likely represents a hematoma or haemorrhagic pericardial effusion. It is often seen following thoracic aortic dissection, trauma, or malignancy.
- Cardiac Tamponade
- Physiology: Pericardial fluid under tension impairs diastolic filling.
- CT signs: Chamber compression (especially RA/RV), septal shift, IVC plethora, contrast reflux into IVC
- Note: CT may detect tamponade incidentally, but echo remains the diagnostic modality of choice.
◾️Pericardial masses
- True pericardial masses should be differentiated from a loculated pericardial or pleural effusion or focal pericardial thickening.
- The most common benign pericardial lesion is a pericardial cyst which is recognized by its classic location, most commonly at right cardiophrenic angle, and circumscribed cystic appearance.
- Solid lesions, especially if multiple, should raise concern for metastases especially in the setting of a known primary malignan
Large Thoracic Vessels
Aortic and pulmonary artery pathologic abnormalities are common indications for chest CT, and incidental findings in these and other thoracic vessels are not uncommon in chest CT performed for other reasons.
Aorta and Arch vessels
Normal Aorta: Thin, smooth-walled, tapers as it descends left of the spine.
Normal Diameters (nongated CT upper limits):
- Ascending aorta: ≤4.1 cm
- Descending aorta: ≤3.0 cm
- Note: Varies with age, sex, and body surface area.
💡Key Pathologies Detectable on CT *
- Atherosclerosis: Calcified/noncalcified plaque
- Aneurysm: Focal dilatation (>1.5× normal diameter)
- Dissection: Intimal flap separating true/false lumens
- Thrombus: Mural or luminal filling defect
- Other: Intramural hematoma, penetrating ulcer, rupture
Pulmonary Arteries on Chest CT
Normal Anatomy: Right and left pulmonary arteries branch from the main pulmonary artery (MPA), tapering into lobar, segmental, and subsegmental vessels.
MPA Diameter Threshold (screening):
- >3.0–3.2 cm suggests possible pulmonary hypertension
- MPA > ascending aorta diameter raises suspicion
- Note: Limited specificity; many normotensive patients exceed these values
💡Key Pathologies Detected on CT:
- Pulmonary embolism: Filling defect, arterial cutoff
- Pulmonary artery aneurysm: Focal dilatation
- Arteriovenous malformation: Direct artery-to-vein connection
- Chronic thromboembolic disease: Web, stenosis, occlusion
Major abdominal and Thoracic Veins on Chest CT
Normal Anatomy: Subclavian + jugular veins → innominate veins (brachiocephalic vein) → superior vena cava (below figure).
Pathologies Detected:
- Venous stenosis/occlusion: Luminal narrowing with collateral formation
- Thrombus: Filling defect, often with wall adherence
Contrast as a Hemodynamic Tracer
- Reflux. Contrast reflux into the inferior vena cava (IVC) or hepatic veins suggests elevated right heart pressures *.
Key Pitfall:
- Contrast mixing artifact: Transient, swirling hypodensity from unopacified blood inflow
- Differentiation: Artifact is central, transient, and lacks wall attachment; thrombus is peripheral, persistent, and often enhances peripherally.
Other Incidental but Critical Pathologies
Filling defects in the cardiac chambers
◾️Thrombi
- Most common filling defect in cardiac chambers
- Predilection sites: Left atrial appendage + Left ventricular apex
- Left atrial thrombus: Associated with mitral valve disease, left atrial dysfunction, atrial fibrillation
- Right atrial thrombus: More likely with central venous catheters
- LV thrombus: Increased risk with wall motion abnormalities (e.g., LV aneurysm)
- Other risk factors: Prosthetic cardiac valves, pacemakers
- Complication: Arterial embolism in up to 20% → early identification critical
- CT imaging: Typically non-enhancing; chronic thrombi may be heterogeneous with peripheral fibrous capsule or calcification
- MRI: Helps distinguish thrombus from tumor (e.g., myxoma, tumor thrombus)
◾️Myxomas
- Most common primary benign cardiac tumor (~50% of all primary benign cardiac tumors)
- Location: 75% in left atrium, typically at interatrial septum near limbus of fossa ovalis
- Types: Sporadic (most common); familial; Carney complex
- Presentation: Asymptomatic, obstructive symptoms, embolic phenomena, or constitutional symptoms
- CT appearance: Lower attenuation than contrast-filled chamber; may be heterogeneous (calcification, hemorrhage, thrombus, hemosiderin)
- Differentiation from thrombus: Myxomas are larger and arise from fossa ovalis (not left atrial appendage)
◾️Malignant Cardiac Masses
- Metastases (20–40× more common than primary cardiac malignancies)
- Prognosis: Generally poor
- Common primaries: Lung, breast, melanoma (highest propensity)
- Four pathways of spread:
- Direct contiguous spread
- Hematogenous dissemination
- Transvenous extension
- Retrograde lymphatic invasion
- Manifestations:
- Pulmonary/mediastinal mass with direct invasion
- Myocardial masses (hematogenous)
- Central mass extending into left atrium (venous extension)
- Pericardial effusion or nodularity (lymphatic)
- Primary Cardiac Tumors (rare; incidence <0.02–0.056%)
- Angiosarcoma (most common primary cardiac malignancy in adults)
- Demographics: Middle-aged males
- Location: Right atrium
- Presentation: Right heart failure, hemorrhagic pericardial effusion, tamponade
- CT: Discrete or infiltrating mass with necrosis; highly vascular
- Metastatic disease: 66–89%
- Prognosis: Poor
- Rhabdomyosarcoma (most common primary cardiac malignancy in infants/children)
- Features: May be multiple; occurs on valves; any chamber; involves myocardium; nodular pericardial invasion (not sheet-like)
- Other sarcomas (undifferentiated, leiomyosarcoma, fibrosarcoma, osteosarcoma)
- Location: Typically left heart chambers
- Presentation: Left heart failure
- Prognosis: Poor (average survival ~1 year)
- Primary Cardiac Lymphoma
- Definition: Mostly confined to heart or pericardium (aggressive B-cell lymphoma)
- Location: More frequent in right atrium
- Common finding: Pericardial effusion (sometimes only finding)
- Key feature: Favorable response to chemotherapy (unlike other primary cardiac malignancies)
- CT findings: Non-specific
- Angiosarcoma (most common primary cardiac malignancy in adults)
📍 Takeaway
- Thrombus (most common, non-enhancing, LAA/LV apex) vs. myxoma (left atrium, fossa ovalis, heterogeneous) vs. metastasis (common, lung/breast/melanoma) vs. primary sarcoma (rare, angiosarcoma = right atrium, poor prognosis) — cardiac MRI aids differentiation.
Left Ventricular Outpouchings: True Aneurysm vs. Pseudoaneurysm
Why It Matters
- Pseudoaneurysm → high rupture risk → surgical repair recommended
- True aneurysm → often managed medically
◾️True Aneurysm
- Cause: Post-MI (majority at apex or anterolateral wall)
- Pathology: Thin, scarred, noncontractile myocardium (akinetic or dyskinetic)
- Ostium: Wide, easily visible
- Posterior infarcts are less reported (often lethal due to papillary muscle involvement + severe mitral regurgitation)
◾️Pseudoaneurysm
- Pathology: Rupture of LV free wall contained by overlying adherent pericardium
- Wall composition: No myocardium — only pericardium
- Clinical course: Usually ruptures → immediate death; survivors may present with CHF or emboli (due to slow flow and thrombosis)
- Ostium: Narrow, often difficult to visualize
- Location: Typically posteroinferior
- Causes: Ischemia, infarction, or trauma
💡Imaging Pearls
- Myocardium surrounding cavity → True aneurysm (thinned myocardium)
- Myocardial discontinuity → Pseudoaneurysm
- ECG-gated MDCT or cine MRI can demonstrate form and function noninvasively
Caveats
- True aneurysms are much more common than pseudoaneurysms
- Posterior outpouchings are harder to detect
- Location alone is insufficient for clinical decision-making
📍Takeaway
- Pseudoaneurysm (narrow neck, posterior location, no myocardium, high rupture risk) requires surgery; true aneurysm (wide neck, apical/anterior, thinned myocardium) is managed medically.
- LV outpouchings summary table 👇
| Feature | True Aneurysm | Pseudoaneurysm |
|---|---|---|
| Most common location | Apex or anterolateral wall | Posteroinferior |
| Neck (ostium) | Wide, easily visible | Narrow, often hard to visualize |
| Wall composition | Thinned scarred myocardium | Pericardium only (no myocardium) |
| Contractility | Akinetic or dyskinetic | Noncontractile / dyskinetic |
| Cause | Prior myocardial infarction | Rupture of LV free wall (ischemia, infarct, trauma) contained by adherent pericardium |
| Complications | Heart failure, arrhythmia, blood stasis, thrombus, emboli | Congestive heart failure, embolic events, high rupture risk |
| Management | Medical (HF therapy, anticoagulation if thrombus present) | Surgical repair |
Step 3 – The Airways & Pleura
◾️Shift to lung windows (W:1500, L:-600). This is a rapid survey.
- Airways: Follow the trachea and main bronchi to the hilum.
- Look for: Foreign body, mass, extrinsic compression, pneumomediastinum (air outlining vessels).
- Pleura:
- Pneumothorax: Look for the visceral pleural line, especially at the lung apex in a supine patient. Don’t be fooled by skin folds or bedding.
- Effusion: Assess volume and character. Is it localized? Simple vs. complex (septations, debris).
- Subcutaneous Emphysema: Trace it back to its source (airway, esophageal, or alveolar rupture).
◾️Differentiating Pleural vs. Pulmonary Opacities in Chest CT
- Differentiating a loculated pleural effusion from an intraparenchymal opacity (consolidation, atelectasis, or mass) is a common diagnostic challenge in emergency chest CT. The distinction is critical: a loculated empyema requires drainage, while a pulmonary opacity may demand antibiotics, bronchoscopy, or observation. The key lies in morphology (below figure).
- Loculated effusions typically form a lentiform or biconvex shape with obtuse margins against the chest wall, displace adjacent lung, and lack air bronchograms.
- In contrast, pulmonary opacities make an acute angle with the chest wall, often contain air bronchograms or vessels, and may cause volume loss.
- The following table summarizes the distinguishing features — angle, shape, air bronchograms, split pleura sign, and effect on adjacent structures — to guide rapid, accurate differentiation at the workstation.
⚡ ER Workflow – 10-Second Differentiation: Loculated Effusion vs. Pulmonary Opacity
| Step | Question | Loculated Effusion | Pulmonary Opacity |
|---|---|---|---|
| 1 | Angle with chest wall? | Obtuse (wide, gradual takeoff) | Acute (sharp interface) |
| 2 | Air bronchograms? | No (bronchi displaced) | Yes (branching air-filled bronchi) |
| 3 | Split pleura sign? | Yes (if exudative/empyema) | No |
| 4 | Volume loss? | No (lobe compressed) | Yes (atelectasis, fissure displacement) |
| 5 | Displacement of vessels? | Away from fluid | Preserved or crowded |
60-Second Catastrophic Scan
The table below summarizes the four critical anatomical targets, their corresponding catastrophic findings, and the immediate actions required. This checklist is not for definitive diagnosis but for triage. Its goal is to answer: “Is there an immediately life-threatening problem I must act on now?”
- Once Step 1 is clear, you have bought the time to delve into the parenchymal patterns (Part II) and integrate the full clinical picture (Part III).
⚡ The 60-Second Catastrophic Scan – Emergency Radiology Protocol
Soft-tissue windows (W:350-400, L:40-50) | Time: 15-20 seconds to identify immediately life-threatening conditions
This systematic 60-second scan rules out immediately life-threatening conditions. Any positive finding requires immediate intervention before proceeding to a full detailed read.
| Anatomical Target | Critical Findings to Exclude | Immediate Action / Clinical Pearl |
|---|---|---|
| Aorta & Great Vessels EMERGENT Trace from arch to diaphragm |
|
|
| Central Pulmonary Arteries URGENT Main, right, and left pulmonary arteries |
|
|
| Tension Physiology EMERGENT Assess mediastinal symmetry |
|
|
| Tubes & Lines CRITICAL Use scout topogram for overview |
|
|
Follow this chronological order to avoid missing critical findings. Each step should take 5-10 seconds.
- Step 1 – Scout topogram: Check tube/line positions (ETT, CVC, chest tube, NG tube) before axial scrolling
- Step 2 – Aorta (soft-tissue windows): Trace from arch to diaphragm for intimal flap, crescentic hematoma, or periaortic blood
- Step 3 – Pulmonary arteries: Look for saddle PE or large filling defects at bifurcation
- Step 4 – Tension physiology: Assess mediastinal shift, diaphragmatic inversion, contralateral lung compression
- Step 5 – Pericardium: Check for large effusion or signs of tamponade
- Step 6 – Lung windows (quick pass): Identify massive pneumothorax, hemothorax, or large consolidation
- Step 7 – Upper abdomen (if included): Look for free air, large aortic pathology extending below diaphragm
| Finding | CT Appearance | Action | Don’t Miss |
|---|---|---|---|
| Aortic dissection | Intimal flap (linear filling defect), true and false lumen | Type A → CT surgery STAT; Type B → medical management + BP control | Pericardial effusion = impending tamponade |
| Intramural hematoma | Crescentic high-attenuation (>60 HU) wall thickening without flap | Same management as dissection; often progresses to dissection | Often hyperacute; may have no intimal tear visible |
| Penetrating atherosclerotic ulcer | Focal contrast outpouching from aortic lumen | Risk of rupture if deep or symptomatic | Can mimic dissection on non-contrast |
| Aortic rupture | Periaortic hematoma, active extravasation (blush) | Emergency surgical or endovascular repair | Check for hemothorax or retroperitoneal blood |
| Traumatic aortic injury | Pseudoaneurysm, intimal irregularity at isthmus | Surgical consultation; BP control (beta-blocker) | Associated with mediastinal hematoma |
In acute PE, clot burden does not equal risk. Right ventricular (RV) strain is the key prognostic factor.
| Sign of RV Strain | CT Finding | Clinical Implication |
|---|---|---|
| RV enlargement | RV/LV diameter ratio >1 on axial or 4-chamber view | Increased risk of hemodynamic collapse |
| Septal bowing | Interventricular septum flattened or bowed toward LV | Pressure overload, high-risk PE |
| Contrast reflux | IVC or hepatic veins opacified from right heart | Right atrial pressure elevation |
| Pulmonary artery diameter | Main PA >3 cm | Chronic pulmonary hypertension or acute overload |
| Thrombus burden | Saddle PE, extensive bilateral clots | Less predictive than RV strain; large burden still concerning |
Tension physiology is a clinical diagnosis, but CT can confirm the mechanism and exclude mimics.
| Finding | CT Appearance | Differential | Action |
|---|---|---|---|
| Mediastinal shift | Trachea, heart, great vessels shifted to contralateral side | Massive pleural effusion, large mass, contralateral atelectasis | If pneumothorax present → immediate decompression |
| Diaphragmatic inversion | Hemidiaphragm flattened or inverted (concave upward) | Large effusion, eventration | Indicates high ipsilateral pressure |
| Contralateral lung compression | Lung volume reduced on opposite side | None | Severe tension physiology |
| Deep sulcus sign | Deep or lucent costophrenic sulcus on supine X-ray/CT | Pneumothorax in supine patient | Suspect tension even without mediastinal shift |
Malpositioned tubes and lines are common, easily missed, and potentially fatal. Check these on every scan.
| Device | Normal Position | Malposition | Consequence |
|---|---|---|---|
| Endotracheal tube (ETT) | Tip 3-5 cm above carina (T4 level) | Mainstem intubation (usually right), esophageal intubation | Hypoxia, atelectasis, pneumothorax |
| Central venous catheter (CVC) | Tip at SVC/RA junction (just below carina) | Arterial placement, too deep (cardiac), pneumothorax | Arterial injury, arrhythmia, tamponade |
| Chest tube | Apical (pneumothorax) or basilar (effusion), intrapleural | Intraparenchymal, intra-abdominal, subcutaneous, fissural | Ineffective drainage, organ injury |
| Nasogastric (NG) tube | Course posterior to trachea, tip in stomach below diaphragm | Bronchial placement, coiling in esophagus, esophageal perforation | Pneumonia (if bronchial), mediastinitis (if perforation) |
| Pacemaker wires | Tip in RV apex or septum | Perforation through myocardium, coiling | Tamponade, failure to capture |
| IABP balloon | Tip just below left subclavian, proximal at T7-T9 | Too high (subclavian occlusion), too low (ineffective), malposition | Limb ischemia, visceral ischemia, balloon rupture |
PART II: Lung Abnormalities In Chest CT
The lung parenchyma tells a story through its patterns. In the critical care setting, these patterns must be rapidly recognized and integrated with clinical data to narrow the differential diagnosis. This section provides a systematic framework for interpreting the most common and critical parenchymal findings *.
Before diving into specific patterns, a brief understanding of lung anatomy is essential. The patterns you will learn; e.g., ground-glass, consolidation, crazy-paving, and tree-in-bud, are not random. They localize to specific anatomic compartments within the secondary pulmonary lobule (SPL) (the smallest unit of lung structure bounded by connective tissue septa). Additionally, lobar anatomy and fissures guide localization of consolidation, collapse, and pleural processes. Airway anatomy explains why aspiration favors the right lung and why normal bronchioles are invisible on CT. With this framework, pattern recognition becomes anatomic localization, narrowing your differential before you know the clinical context.
Essential Lung Anatomy for Emergency Chest CT Interpretation
1. Lung Lobes and Fissures
Before interpreting parenchymal patterns, one must first understand the lobar architecture of the lungs. the right has three (upper, middle, lower), the left has two (upper, lower). These boundaries localize disease: aspiration favors the right lower lobe, pneumothorax that crosses a fissure suggests tension, and silhouette signs identify lobar consolidation. The tables below provide a rapid reference 👇.
📚 1. Lung Lobes and Fissures – The Macroscopic Landscape
The foundation of chest CT interpretation: lobes, fissures, and their clinical relevance in the emergency setting
The right lung is divided into three lobes by the horizontal and oblique fissures. Each lobe has distinct anatomic landmarks on CT.
| Lobe | Location | Key CT Landmark | Clinical Pearl |
|---|---|---|---|
| Upper lobe | Above horizontal fissure | Anterior mediastinal border, aortic arch level | Apical consolidation suggests aspiration in supine patient |
| Middle lobe | Between horizontal and oblique fissures | Right heart border (obscured = middle lobe process) | Middle lobe syndrome = recurrent atelectasis/infection |
| Lower lobe | Below oblique fissure | Posterior to oblique fissure, adjacent to hemidiaphragm | Posterior basal segments = most common site of aspiration (supine) |
The left lung has two lobes (upper and lower), separated by the oblique fissure. The lingula is functionally analogous to the right middle lobe.
| Lobe | Location | Key CT Landmark | Clinical Pearl |
|---|---|---|---|
| Upper lobe | Above oblique fissure | Aortic arch, left pulmonary artery, lingula (anterior) | Lingula consolidation mimics right middle lobe process |
| Lower lobe | Below oblique fissure | Descending thoracic aorta, posterior to oblique fissure | Superior segment = classic site for aspiration in recumbent patient |
Fissures are invaginations of visceral pleura that separate lobes. They are critical landmarks for localizing disease and understanding pleural processes.
| Fissure | Occurrence | Orientation | CT Identification | Clinical Pearl |
|---|---|---|---|---|
| Horizontal (minor) | Right lung only | Axial plane (~T4 level) | Thin line from right hilum to chest wall | Separates upper from middle lobe; often incomplete or accessory fissures common |
| Oblique (major) | Bilateral | Coronal oblique plane | Curvilinear line from T4/T5 posteriorly to diaphragm anteriorly | Separates lower lobe from upper/middle lobes |
A pneumothorax that crosses a fissure indicates either tension physiology (complete lobar collapse) or multiloculated pneumothorax. Simple pneumothorax stays within one pleural compartment.
Lobar anatomy directly impacts differential diagnosis for common ER presentations:
| Clinical Scenario | Lobar Predilection | Why? |
|---|---|---|
| Aspiration pneumonia (supine) | Right lower lobe (posterior segments) or right upper lobe (apical segments if supine) | Bronchial anatomy: right main bronchus shorter, wider, more vertical |
| Aspiration pneumonia (upright) | Bilateral lower lobes (basal segments) | Gravity-dependent distribution |
| Pulmonary edema | Perihilar + lower lobe predominance | Hydrostatic forces + dependent lung zones |
| Pulmonary embolism | Lower lobes > upper lobes | Greater blood flow to lower lobes |
| Tuberculosis (reactivation) | Upper lobes (apical/posterior segments) | High oxygen tension favors mycobacterial growth |
| Lobar collapse | Depends on lobe; known silhouette signs (e.g., right heart border = middle lobe) | Fissures become displaced toward atelectatic lobe |
• Right heart border obscured = right middle lobe
• Left heart border obscured = lingula
• Aortic arch or left hemidiaphragm obscured = left lower lobe
• Right hemidiaphragm obscured = right lower lobe
2. Airway Anatomy-From Trachea to Alveolus
The airway divides into the conducting zone (trachea → terminal bronchioles, no gas exchange) and the respiratory zone (respiratory bronchioles → alveoli, gas exchange). On CT, bronchi (>1 mm, cartilage, glands) are visible; bronchioles (<1 mm, no cartilage, no glands) are normally invisible. When visible (tree-in-bud), they are diseased. The right main bronchus is shorter, wider, and more vertical , explaining why aspiration favors the right lung. The tables below provide a rapid reference.
🌬️ 2. Airway Anatomy – From Trachea to Alveolus
The tracheobronchial tree: conducting zone → respiratory zone. What you see on CT and what it means.
The airway is divided into the conducting zone (no gas exchange, trachea → terminal bronchioles) and the respiratory zone (gas exchange begins at respiratory bronchioles).
| Level | Structure | Diameter | Cartilage | Glands | CT Visibility (Normal) |
|---|---|---|---|---|---|
| 1 | Trachea | ~2 cm | C‑rings | Yes | Visible wall |
| 2 | Main bronchi | ~1 cm | Plates | Yes | Visible wall |
| 3 | Lobar bronchi | 5–10 mm | Plates | Yes | Visible as branching |
| 4 | Segmental bronchi | 3–5 mm | Plates | Yes | Visible adjacent to arteries |
| 5 | Subsegmental bronchi | 1–3 mm | Plates | Yes | Visible in lung windows |
| 6 | Bronchioles | <1 mm | None | None | Normally invisible |
| 7 | Terminal bronchiole | 0.5–1 mm | None | None | Invisible (end of conducting) |
| 8 | Respiratory bronchiole | <0.5 mm | None | None | Invisible (begins exchange) |
| 9 | Alveolar ducts | — | None | None | Invisible |
| 10 | Alveolar sacs | — | None | None | Airspace (not discrete) |
This distinction is essential for CT interpretation. They differ in structure, function, and disease patterns.
| Feature | Bronchus | Bronchiole |
|---|---|---|
| Diameter | >1 mm | <1 mm |
| Cartilage | Present (plates or rings) | Absent |
| Submucosal glands | Present | Absent |
| Goblet cells | Present | Absent (Clara cells instead) |
| Wall layers | Complete (mucosa, submucosa, cartilage, adventitia) | Simple epithelium + smooth muscle |
| CT visibility (normal) | Visible | Invisible |
| Disease signature | Wall thickening, bronchiectasis | Tree-in-bud, centrilobular nodules |
Understanding these zones explains why certain diseases present where they do.
| Zone | Structures Included | Function | Volume (adult) |
|---|---|---|---|
| Conducting zone | Trachea → Terminal bronchioles (levels 1–7) | Air transit, warming, humidification, filtration | ~150 mL (anatomical dead space) |
| Respiratory zone | Respiratory bronchioles → Alveolar sacs (levels 8–10) | Gas exchange (O₂/CO₂ diffusion) | ~2.5–3 L |
Trachea (trunk)
/ \
L main R main (wider, steeper → aspiration risk)
| |
L lobars R lobars
| |
segmentals → conduct air (cartilage present)
| |
subsegmentals |
└──────┬────────┘
bronchioles (NO cartilage, NO glands)
|
terminal bronchioles (end of conducting zone)
|
respiratory bronchioles (BEGIN gas exchange)
|
alveolar ducts
|
alveolar sacs
Here is how airway anatomy translates into actionable CT findings in the emergency setting:
| CT Finding | Anatomic Location | What It Means | ER Action |
|---|---|---|---|
| Tree-in-bud | Centrilobular (terminal/respiratory bronchioles) | Mucus, pus, or fluid impacted in small airways | Consider infectious bronchiolitis (TB, MAC, bacterial), aspiration, or panbronchiolitis |
| Bronchial wall thickening | Bronchi (>1 mm, cartilage-bearing) | Airway inflammation/reactive change | Asthma, chronic bronchitis, ABPA, cystic fibrosis |
| Bronchiectasis | Irreversible bronchial dilation | Signet ring sign (bronchus diameter > adjacent artery) | Non-CF bronchiectasis, CF, post-infectious, ciliary dyskinesia |
| Traction bronchiectasis | Peripheral bronchi/bronchioles pulled open | Fibrosis (UIP/IPF, NSIP) | Suspect interstitial lung disease; pulmonary function tests needed |
| Air trapping | Respiratory bronchioles (expiration CT) | Small airway obstruction during expiration | Bronchiolitis obliterans, hypersensitivity pneumonitis, asthma |
| Mosaic attenuation | Lung parenchyma (air trapping vs. vascular) | Heterogeneous lung density | Differentiate air trapping (expiration) from ground-glass (inspiration) or vascular (PE) |
| Crazy-paving | Lobular + septal thickening | Alveolar proteinosis, PJP, edema, DAD | Check history: immunocompromised (PJP), acute (edema/DAD), chronic (proteinosis) |
• Visible airway <1 mm = abnormal (tree-in-bud)
• Wall thickening >1 mm = bronchial disease
• Dilated bronchus > adjacent artery = bronchiectasis (irreversible)
3. Secondary pulmonary lobule (SPL)
◾️The secondary pulmonary lobule (SPL) is the smallest unit of lung structure bounded by connective tissue septa, measuring 1–2.5 cm in diameter. It is the fundamental anatomic framework for interpreting interstitial and small airway disease on high-resolution CT (HRCT).
🔬 3. The Secondary Pulmonary Lobule (SPL) – Your CT "Address System"
The smallest lung unit bounded by connective tissue septa. The fundamental framework for localizing interstitial and small airway disease on HRCT.
The secondary pulmonary lobule (SPL) is the smallest unit of lung structure surrounded by connective tissue septa. It is the fundamental anatomic framework for interpreting interstitial and small airway disease on HRCT.
| Property | Value |
|---|---|
| Size | 1–2.5 cm in diameter (approximately the size of a small grape) |
| Shape | Polygonal (usually 3–5 sides) on axial CT |
| Number per lung | Approximately 5,000–10,000 |
| Primary lobule | Smaller unit (single respiratory bronchiole + its alveoli); not visible on CT |
| Why "secondary"? | The primary lobule (Miller, 1937) was defined first (functional gas exchange unit). The secondary lobule (Reid, 1958) is larger, structurally bounded, and CT-visible. |
Each SPL has three distinct anatomic compartments, each with specific structures and CT correlates when diseased.
| Component | Structure | Normal CT | Abnormal CT Finding |
|---|---|---|---|
| Centrilobular core | Terminal bronchiole + pulmonary arteriole | Invisible (arteriole may be seen as tiny dot) | Tree-in-bud, centrilobular nodules, ground-glass dots |
| Lobular parenchyma | Acinus (respiratory bronchioles + alveolar ducts + alveoli) | Invisible (air-filled) | Ground-glass opacity, consolidation |
| Perilobular region (septa) | Interlobular septa (contain pulmonary veins + lymphatics) | Invisible (<0.1 mm thin) | Septal thickening (smooth, nodular, or irregular) |
On CT, every interstitial or airway abnormality localizes to one of three SPL compartments. Identify the compartment → narrow the differential.
| Feature | Description |
|---|---|
| Location | Within 5–10 mm of the center of the lobule; does NOT touch the pleura |
| Key sign | Tree-in-bud (branching centrilobular opacities) |
| Differential | Infectious bronchiolitis (TB, MAC, bacterial), hypersensitivity pneumonitis, respiratory bronchiolitis (smoker's), pulmonary edema (early) |
| ER pearl | Tree-in-bud + fever → atypical pneumonia or endobronchial infection |
| Feature | Description |
|---|---|
| Location | Along interlobular septa, fissures, bronchovascular bundles, subpleural lung |
| Key sign | Nodular or smooth septal thickening; fissural nodularity |
| Differential | Sarcoidosis, lymphangitic carcinomatosis, silicosis, lymphoma |
| ER pearl | Perilymphatic nodules + young patient + dyspnea → sarcoidosis (most common) |
| Feature | Description |
|---|---|
| Location | Uniform distribution: centrilobular, septal, and subpleural regions equally involved |
| Key sign | 1–3 mm micronodules diffusely distributed |
| Differential | Miliary tuberculosis, hematogenous metastases, fungal dissemination (histoplasmosis, coccidioidomycosis) |
| ER pearl | Random micronodules + immunocompromised host → miliary TB or disseminated fungal infection until proven otherwise |
This ASCII diagram shows how each disease pattern appears within a single SPL:
• Dot alone + fuzzy → Centrilobular (Hypersensitivity pneumonitis, infection)
• Walls only (square border dots) → Perilymphatic (Sarcoidosis)
• Everywhere dots → Random (Miliary TB)
Interlobular septa become visible on CT when thickened (>0.1 mm). The pattern of thickening suggests the etiology.
| Pattern | CT Appearance | Most Likely ER Diagnosis | Key Distinction |
|---|---|---|---|
| Smooth | Thin, uniform linear opacities | Cardiogenic pulmonary edema, lymphangitic carcinomatosis | Cardiomegaly + vascular congestion = edema |
| Nodular | Beaded or irregular thickening | Sarcoidosis, lymphangitic carcinomatosis, silicosis | Young patient → sarcoidosis; older with known cancer → carcinomatosis |
| Irregular | Thick, distorted, often with traction | Interstitial fibrosis (UIP/IPF) | Less urgent in ER; associated with honeycombing |
The tree-in-bud sign is the most important SPL-based sign in chest CT. It represents inflammation, mucus, or fluid within the terminal bronchioles (normally invisible).
| Feature | Description |
|---|---|
| What it looks like | Small centrilobular nodules connected to branching V-shaped or Y-shaped structures |
| What it represents | Inflammation, mucus, or fluid in terminal/respiratory bronchioles |
| SPL localization | Always centrilobular (never touches the pleura because centrilobular bronchiole does NOT reach the pleural surface) |
| Infectious causes | Bacterial pneumonia (TB, MAC, Pseudomonas), viral bronchiolitis, fungal |
| Non-infectious causes | Allergic bronchopulmonary aspergillosis (ABPA), aspiration, panbronchiolitis, cystic fibrosis |
Use this table to rapidly generate a differential based on the SPL pattern you identify.
| Category | SPL Pattern | Classic Disease | Key CT Finding |
|---|---|---|---|
| Airway | Centrilobular | Infectious bronchiolitis | Tree-in-bud |
| Airway | Centrilobular | Hypersensitivity pneumonitis | Ill-defined centrilobular ground-glass nodules |
| Interstitial (lymphatic) | Perilymphatic | Sarcoidosis | Nodular septal thickening, fissural nodules |
| Interstitial (lymphatic) | Perilymphatic | Lymphangitic carcinomatosis | Smooth or nodular septal thickening |
| Interstitial (fibrotic) | Septal + traction | IPF/UIP | Honeycombing (peripheral, basal) |
| Vascular | Centrilobular + septal | Pulmonary edema | Smooth septal thickening (Kerley B) |
| Hematogenous | Random | Miliary TB | 1–3 mm uniform random nodules |
| Inhalational | Centrilobular | Hypersensitivity pneumonitis | Centrilobular ground-glass nodules, mid/upper lung |
| Inhalational | Perilymphatic | Silicosis | Upper lobe perilymphatic nodules, eggshell calcifications |
When you scroll through a chest CT, ask one question: "Where is the abnormality relative to the SPL?"
| If you see... | Then think... | Don't miss... |
|---|---|---|
| Branching centrilobular dots (tree-in-bud) | Airway infection | TB, MAC in immunocompromised |
| Nodules along fissures or pleura | Sarcoidosis / lymphangitic spread | Occult malignancy if older patient |
| 1–3 mm uniform dots everywhere | Miliary process | TB / fungal in immunocompromised |
| Smooth lines reaching pleura (Kerley B) | Pulmonary edema | Cardiogenic vs. non-cardiogenic |
| Thickened septa + crazy‑paving | Alveolar proteinosis / PJP / edema | PJP in HIV patient |
| Centrilobular ground-glass nodules | Hypersensitivity pneumonitis / RB-ILD | Smoking history (RB-ILD) vs. exposure (HP) |
Pattern Recognition in Lung Disease
I. Focal & Multifocal Opacities
◾️Atelectasis vs. Consolidation: The most common differential on ICU CT.
- Atelectasis (Collapse):
- Loss of volume is key. Look for displacement of fissures, mediastinal shift toward the opacity, and compensatory hyperinflation of adjacent lung.
- Often wedge-shaped, subsegmental, or lobar.
- Common in dependent lung zones post-operatively or in ventilated patients.
- Consolidation:
- Represents airspace filling (pus, blood, fluid, cells) without significant volume loss.
- Air bronchograms are a hallmark.
- Consider:
- Infection (Pneumonia): Often segmental/lobar with a geographic border.
- Aspiration: Typically dependent (posterior upper lobes, superior lower lobes), bilateral, and multifocal.
- Hemorrhage: May be patchy, ground-glass, or consolidative. Look for trauma or coagulopathy context.
- Infarction (Hampton's Hump): Wedge-shaped peripheral consolidation with a broad pleural base, often associated with a visible occluding pulmonary embolus.
◾️The Nodule: A Size- & Pattern-Based Approach
- Size Matters: <1 cm = nodule; >3 cm = mass.
- Pattern Dictates Urgency:
- Solid, Smooth, <1 cm:
- Most often benign (granuloma, intrapulmonary lymph node).
- In a septic patient, consider septic emboli (often peripheral, may cavitate).
- Cavitary Nodule/Mass:
- Think necrosis.
- Differential includes infection (abscess, septic emboli, TB, fungal), malignancy (squamous cell carcinoma), vasculitis (GPA), or pulmonary infarction.
- "Tree-in-Bud" Opacities:
- Centrilobular nodules connected to branching linear opacities.
- Highly suggestive of infectious bronchiolitis (e.g., viral, mycobacterial, bacterial). Also seen in aspiration.
- Solid, Smooth, <1 cm:
II. Diffuse & Hazy Opacities
◾️Ground-Glass Opacification (GGO): Hazy increased lung attenuation without obscuration of underlying bronchial and vascular margins. Represents partial airspace filling or interstitial thickening.
- Acute Differential (CRITICAL):
- Diffuse Alveolar Hemorrhage: Diffuse or patchy GGO, often with rapid clearing.
- Acute Interstitial Pneumonia (AIP) / ARDS: Diffuse, symmetric GGO with dependent consolidation. Often has a gravitational gradient.
- Pulmonary Edema (Cardiogenic or Non-Cardiogenic): Symmetric, perihilar/bat-wing distribution, with septal thickening and pleural effusions favoring cardiogenic cause.
- Pneumonia (PJP, viral): Diffuse or mosaic GGO.
◾️Mosaic Attenuation: A patchwork of regions of differing lung density. This can be seen in three predominantly broad categories of lung diseases: small airways disease, vascular lung disease, and infiltrative lung disease
- Key Question: Is it a perfusion defect or an infiltrative disease?
- Vascular Cause (e.g., Chronic PE): Dark areas are hypoperfused; vessels in these areas appear abnormally small.
- Airway Cause (e.g., Constrictive Bronchiolitis): Dark areas are hyperlucent due to air-trapping; vessels are of normal size.
- Infiltrative Cause (e.g., HP): Bright areas are GGO; vessels are of normal size.
III. Cystic & Cavitary Lesions
◾️Cyst: Thin-walled (<2 mm), well-defined, air-filled lesion. Usually implies destructive lung disease.
- Differential: Emphysema, lymphangioleiomyomatosis (LAM – cysts are uniform, diffuse), Langerhans cell histiocytosis (irregular cysts, upper lobe predominance), post-infection.
◾️Cavity: A gas-filled space within a consolidation or mass, with a thick, irregular wall (>4 mm suggestive of malignancy; >15 mm almost always neoplastic or infectious).
◾️Emphysema: No visible wall. Characterized by permanent, abnormal enlargement of airspaces and destruction of alveolar walls. Centrilobular (upper lobe), panlobular (lower lobe in alpha-1), paraseptal.
IV. Bronchiectasis & Bronchial Wall Thickening
◾️Bronchiectasis: Irreversible airway dilation.
- Signs:
- Bronchial lumen diameter > accompanying pulmonary artery ("signet ring sign")
- Lack of tapering (Tram-track sign, representing dilated, nontapered bronchi extending into the lung periphery)
- Visibility of airways within 1 cm of pleural surface.
- Patterns:
- Cylindrical (tram-tracks)
- Varicose (beaded)
- Cystic.
◾️Bronchial Wall Thickening: Common, non-specific finding.
- Acute: Asthma, bronchitis, infection (e.g., bronchiolitis).
- Chronic: Chronic bronchitis, cystic fibrosis, bronchiectasis.
V. Linear, Reticular & Fibrotic Patterns
These suggest interstitial lung disease, which can present acutely (e.g., acute exacerbation of IPF) or be a chronic finding.
◾️Interlobular Septal Thickening: Thickening of the connective tissue septa surrounding secondary pulmonary lobules. Appears as Kerley B lines (peripheral, perpendicular to pleura) on CT.
- Smooth: Think edema (cardiogenic or lymphangitic spread of tumor).
- Nodular/Beaded: Think lymphangitic carcinomatosis.
◾️Honeycombing: Clustered cystic airspaces (usually 3-10 mm) with thick, fibrous walls, located in the subpleural lung. The imaging hallmark of usual interstitial pneumonia (UIP) pattern and end-stage pulmonary fibrosis. Indicates irreversibility.
Spatial Distribution Of Disease
The pattern itself, whether ground-glass, consolidation, nodules, or septal thickening; provides an initial differential. However, anatomic distribution is often more diagnostic than the pattern alone. The same pattern in different locations suggests completely different diseases, for example:
- A predominantly upper lobes GGO is more suggestive for PJP, silicosis; where as a lower lobes GGO are in favor of Edema, NSIP, DIP.
- Key anatomic axes to assess:
- Axial distribution: Central (perihilar) vs. peripheral (subpleural)
- Cranio-caudal distribution: Upper lobe vs. lower lobe predominance
- SPL compartment: Centrilobular, perilymphatic (septal), or random
- Zonal involvement: Anterior vs. posterior (aspiration favors posterior segments)
- Why this matters in the emergency setting:
- Upper lobe predominant GGO in an immunocompromised patient → PJP until proven otherwise
- Peripheral lower lobe predominant GGO in a febrile patient → COVID-19 or viral pneumonia
- Centrilobular nodules → airway-centered disease (infection, hypersensitivity)
- Perilymphatic nodules (fissural, subpleural, septal) → sarcoidosis or lymphangitic spread
- Smooth septal thickening + perihilar GGO → pulmonary edema
◾️Upper vs. Lower Lobe:
- Upper lobe distribution is seen in many perilymphatic and airway centric diseases such as sarcoidosis, silicosis, cystic fibrosis, tuberculosis, and Langerhans cell histiocytosis. Lower lobe distribution classically occurs in a usual interstitial pneumonitis (UIP) pattern of fibrosis and can also occur in gravitationally dependent processes such as aspiration and hematogenous metastatic disease
- Upper: Sarcoidosis, silicosis, Langerhans cell histiocytosis, tuberculosis, and ankylosing spondylitis.
- Lower (Subpleural): Usual Interstitial Pneumonia (UIP), asbestosis, connective tissue disease-related ILD.
◾️Central vs. Peripheral:
- Central distribution suggests diseases related to the bronchovascular bundles; examples include edema and bronchopneumonia in the acute setting, and perilymphatic diseases such as sarcoidosis or silicosis, lymphoma, or Kaposi sarcoma in the chronic setting.
- Peripheral distribution can be seen in acute diseases such as pulmonary infarction, septic emboli, and aspiration; in the subacute or chronic setting, this distribution can be seen in organizing or eosinophilic pneumonia, pulmonary fibrosis, and other diseases.
- Central (Perihilar): Pulmonary edema, sarcoidosis, alveolar proteinosis.
- Peripheral: Organizing pneumonia (OP), chronic eosinophilic pneumonia (CEP), ARDS.
◾️Anterior vs. Posterior (in Supine Patient):
- Dependent (Posterior): Aspiration, atelectasis, edema, pneumonia.
- Non-Dependent: Often indicates a more specific pathology (e.g., OP, eosinophilic pneumonia).
Rapid Pattern Recognition
In the emergency setting, time is limited, and the differential diagnosis must be narrowed quickly. The lung parenchyma tells a story through its patterns, but pattern alone is not enough. The same ground-glass opacity can represent pulmonary edema, PJP pneumonia, or drug toxicity. The same nodules can be infectious, inflammatory, or malignant.
🎯The diagnostic formula:
- ⎮Pattern + Distribution + Time course + Clinical context = Differential diagnosis
This section provides a systematic framework for recognizing the most common and critical patterns encountered in emergency chest CT. For each pattern, the following tables outline the differential diagnosis, key distinguishing features, and emergency actions. Use the step-by-step workflow and quick reference table at the end to rapidly narrow your differential at the workstation.
🩻 Pattern Recognition – Ground-Glass, Consolidation & Crazy-Paving
How to identify, differentiate, and act on the most common parenchymal patterns in emergency chest CT
Ground-glass opacity (GGO) is hazy increased lung attenuation that does not obscure underlying bronchial and vascular margins. It represents partial filling of airspaces, interstitial thickening, or increased capillary blood volume.
| Category | Specific Causes | Key Distinguishing Features | ER Action |
|---|---|---|---|
| Acute / Emergency | Pulmonary edema, PJP, viral pneumonia (COVID, RSV, influenza), DAD/ARDS, diffuse alveolar hemorrhage, hypersensitivity pneumonitis | Edema: smooth septal thickening + cardiomegaly; PJP: upper lobe predominant; COVID: peripheral + lower lobe; DAD: diffuse ± crazy-paving | Oxygen, treat underlying cause; if PJP suspected → initiate steroids + antibiotics; if hemorrhage → check coagulopathy |
| Subacute | Hypersensitivity pneumonitis, organizing pneumonia (COP), drug toxicity (amiodarone, bleomycin), aspiration | HP: centrilobular nodules + mosaic attenuation; COP: peripheral/subpleural band-like; drug: plus fibrosis | Remove exposure, consider steroids for COP/HP; bronchoscopy if no cause found |
| Chronic / Incidental | Desquamative interstitial pneumonia (DIP), nonspecific interstitial pneumonia (NSIP), early IPF, alveolar proteinosis | DIP: lower lobe + smoking history; NSIP: lower lobe ± traction bronchiectasis; IPF: basal + peripheral honeycombing | Outpatient pulmonary referral; IPF requires antifibrotic therapy |
| Focal GGO | Adenocarcinoma (lepidic pattern), organizing pneumonia, hemorrhage, infection | Persistent focal GGO >2 mm wall thickness or solid component → biopsy; resolves on follow-up → infection or hemorrhage | Fleischner Society guidelines for subsolid nodules: follow CT at 3-6 months, then annual for 5 years if persistent |
Consolidation is opacification of airspaces with obscuration of underlying bronchial and vessel margins. Air bronchograms are often visible. It represents replacement of air by fluid, pus, blood, or cells.
| Category | Specific Causes | Key Distinguishing Features | ER Action |
|---|---|---|---|
| Infectious | Bacterial pneumonia (pneumococcal, legionella, Klebsiella), aspiration, TB (primary or reactivation) | Lobar/segmental distribution; air bronchograms; Klebsiella = bulging fissure; Legionella = often bilateral; aspiration = dependent segments | Antibiotics based on local susceptibility; if cavitary or upper lobe → TB workup (AFB, respiratory isolation) |
| Non-infectious | Pulmonary edema (cardiogenic), diffuse alveolar hemorrhage, organizing pneumonia, eosinophilic pneumonia, aspiration (chemical) | Edema: perihilar + cardiomegaly + septal thickening; hemorrhage: diffuse GGO + consolidation; COP: peripheral bands; eosinophilic: peripheral upper lobe | Treat underlying cause; steroids for COP or eosinophilic pneumonia; coagulopathy correction for hemorrhage |
| Chronic / Infiltrative | Lymphoma, adenocarcinoma, lipoid pneumonia (exogenous) | Lymphoma: mass-like consolidation + air bronchograms; adenocarcinoma: persistent focal consolidation with ground-glass halo; lipoid: low-attenuation consolidation (fat density) | Outpatient oncology or pulmonary referral |
| Lobar atelectasis (mimic) | Atelectasis due to bronchial obstruction (tumor, mucus plug, foreign body) | Fissure displacement toward collapse; volume loss; mediastinal shift; no air bronchograms (or crowding) | Bronchoscopy if tumor suspected; chest physiotherapy for mucus plug |
- Air bronchogram = air-filled bronchus surrounded by consolidated lung → commonly infectious or edema
- Signet ring sign = dilated bronchus (≥ adjacent artery) → bronchiectasis (irreversible)
• Supine patient: Posterior segments of upper lobes + superior segments of lower lobes
• Upright patient: Basal segments of lower lobes (gravity-dependent)
• Right lung > left lung (right main bronchus anatomy)
Crazy-paving is a distinctive pattern of ground-glass opacity with superimposed smooth interlobular septal thickening, resembling irregularly shaped paving stones. It is not a specific diagnosis but narrows the differential significantly.
| Disease | Key Features | ER Pearl |
|---|---|---|
| Alveolar proteinosis (PAP) | Most classic cause; crazy-paving often geographic, bilateral, upper/mid lung predominant; usually chronic or subacute | Check for "geographic" distribution + patient with progressive dyspnea; PAS-positive proteinaceous material on BAL; can be autoimmune (anti-GM-CSF) |
| PJP pneumonia | Crazy-paving in immunocompromised; often upper lobe predominant; bilateral GGO with septal thickening | CD4 <200, hypoxia, fever → PJP until proven otherwise; start steroids + TMP-SMX immediately |
| ARDS / DAD (diffuse alveolar damage) | Diffuse bilateral GGO progressing to consolidation and crazy-paving; acute onset within 1 week of insult | ARDS triad: acute onset, bilateral opacities, PaO₂/FiO₂ ≤300, not fully explained by cardiac failure |
| Diffuse alveolar hemorrhage | Crazy-paving + hematocrit effect (dependent layering of blood); GGO + septal thickening | Check for coagulopathy, vasculitis (ANCA), anti-GBM disease; bronchoscopy with serial lavage shows progressively bloodier returns |
| Pulmonary edema | Crazy-paving + cardiomegaly + pleural effusions + perihilar predominant | Cardiomegaly + vascular congestion + septal thickening = cardiogenic edema |
| Lipoid pneumonia (exogenous) | Crazy-paving with low attenuation (fat density) within opacities; often lower lobes; chronic aspiration (mineral oil) | History of chronic laxative or oil-based nasal drop use; oil-red-O stain on BAL |
• PAP (alveolar proteinosis) — classic, geographic
• PJP — immunocompromised, hypoxia
• Pulmonary edema — cardiomegaly, effusions
• Pneumonia (viral or organizing) — clinical history
• Pulmonary hemorrhage — coagulopathy, dropping Hgb
• Pneumonitis (hypersensitivity, drug-induced)
Mosaic attenuation refers to patchy, heterogeneous lung density with regions of lower and higher attenuation. The pattern tells you the cause.


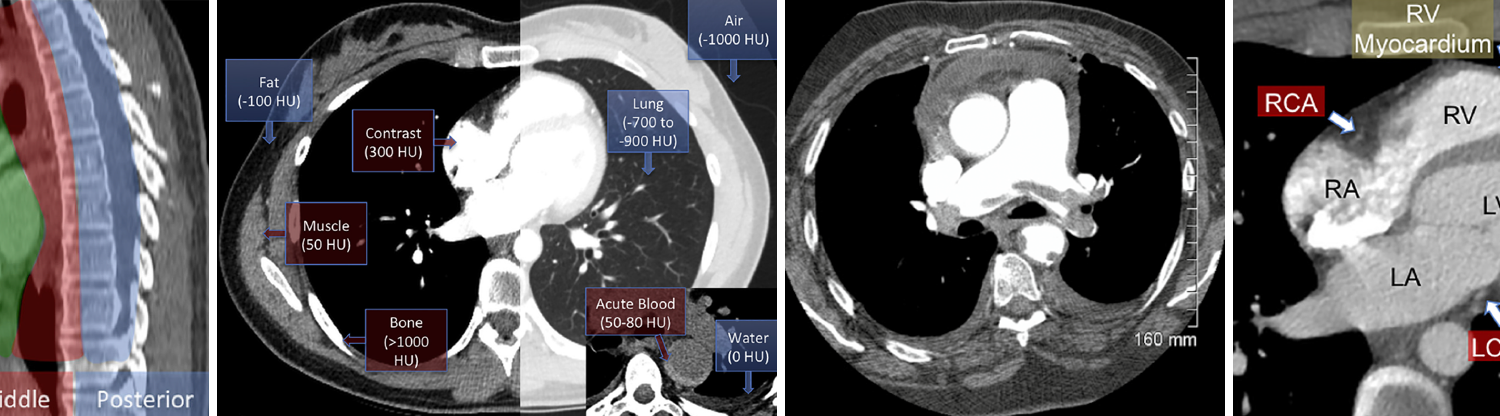
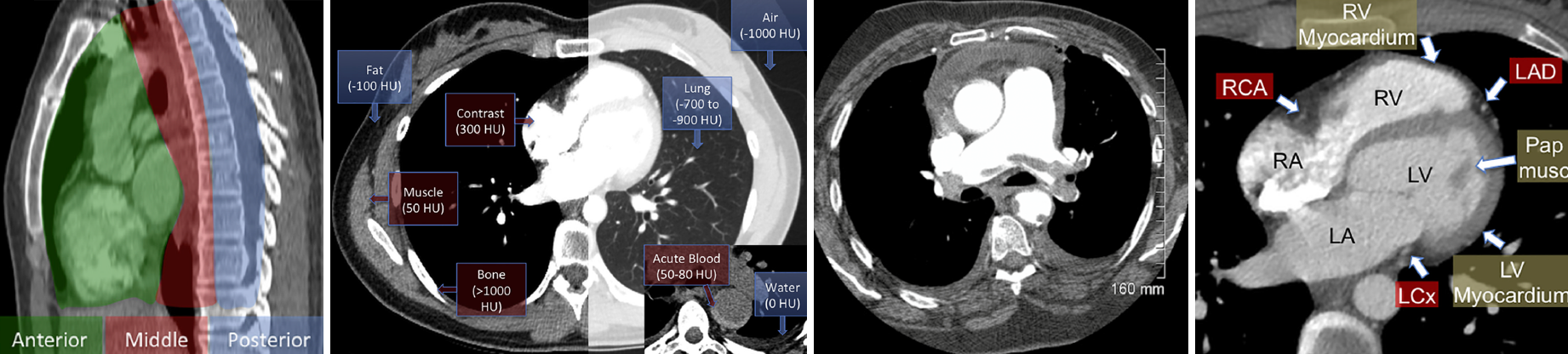
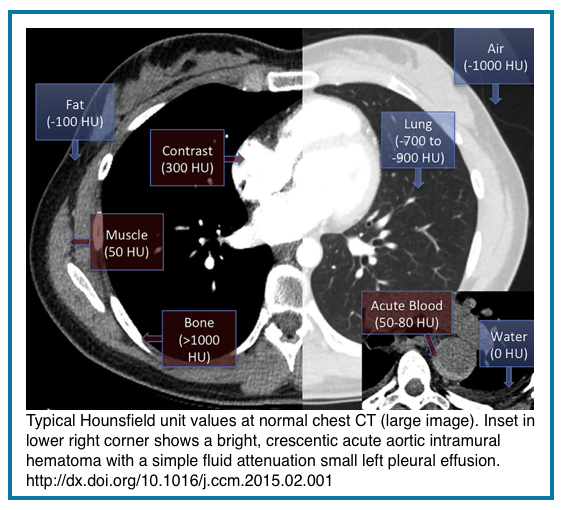
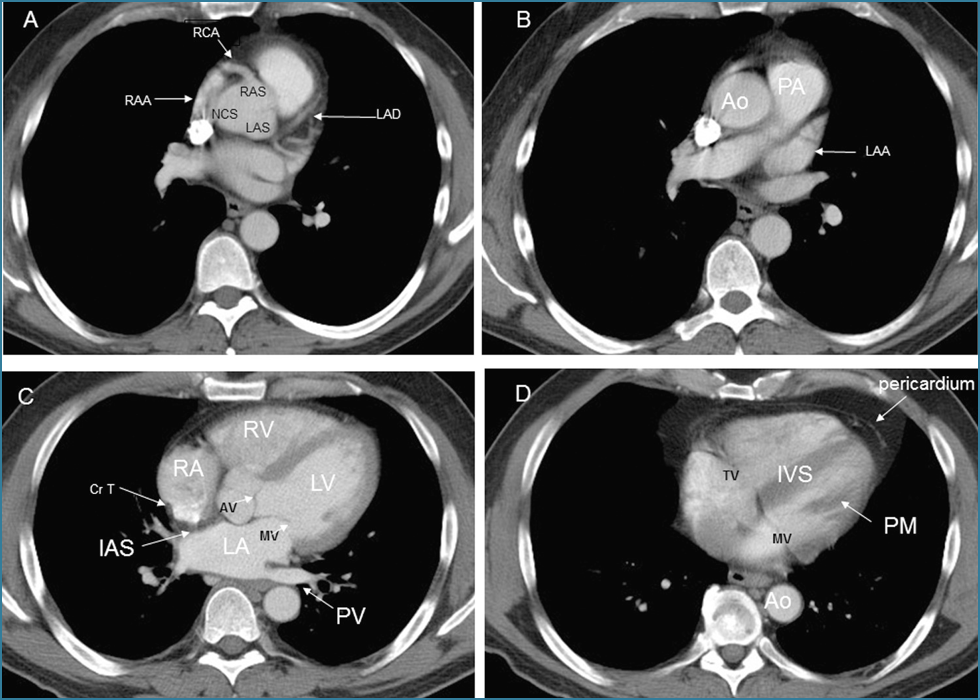
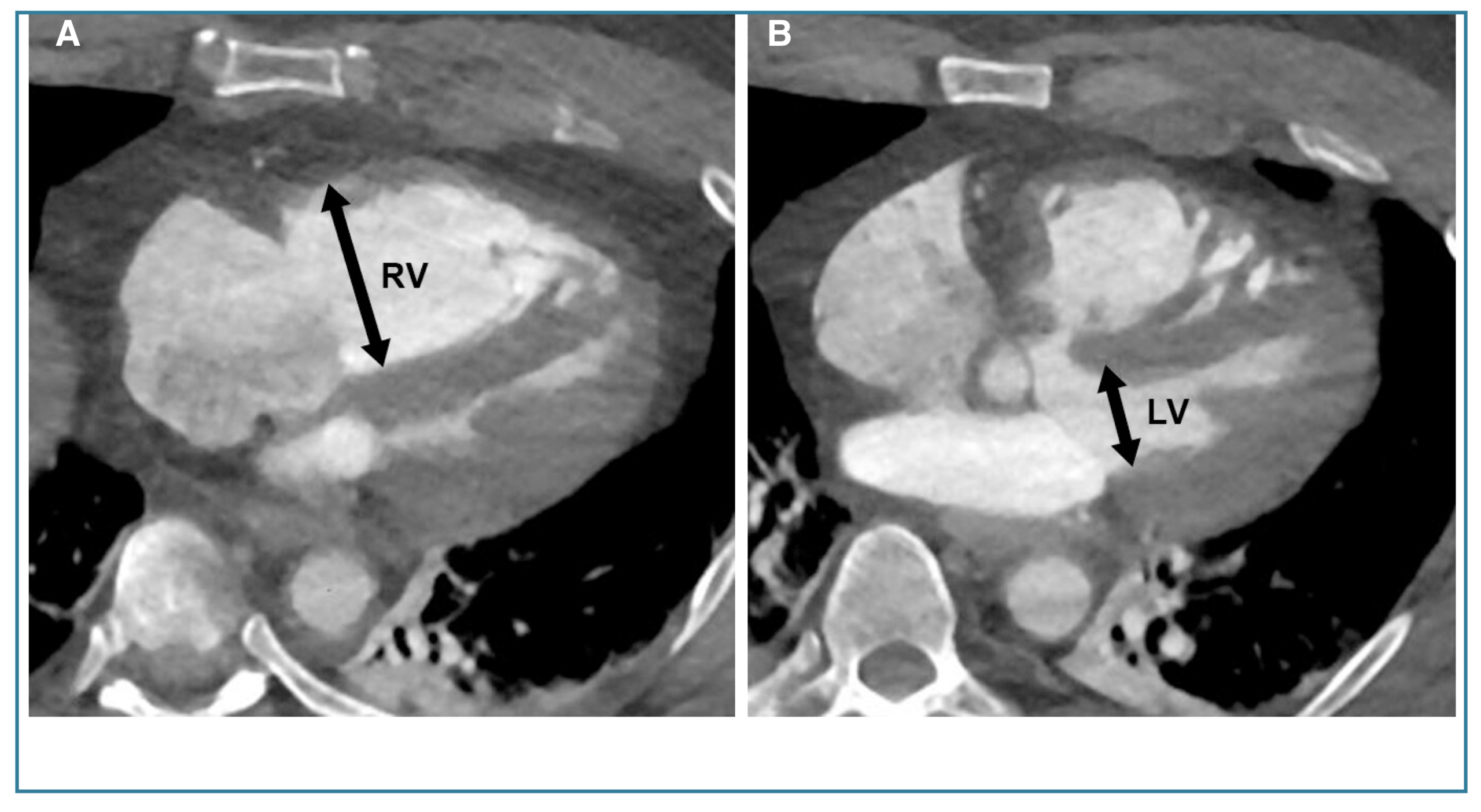
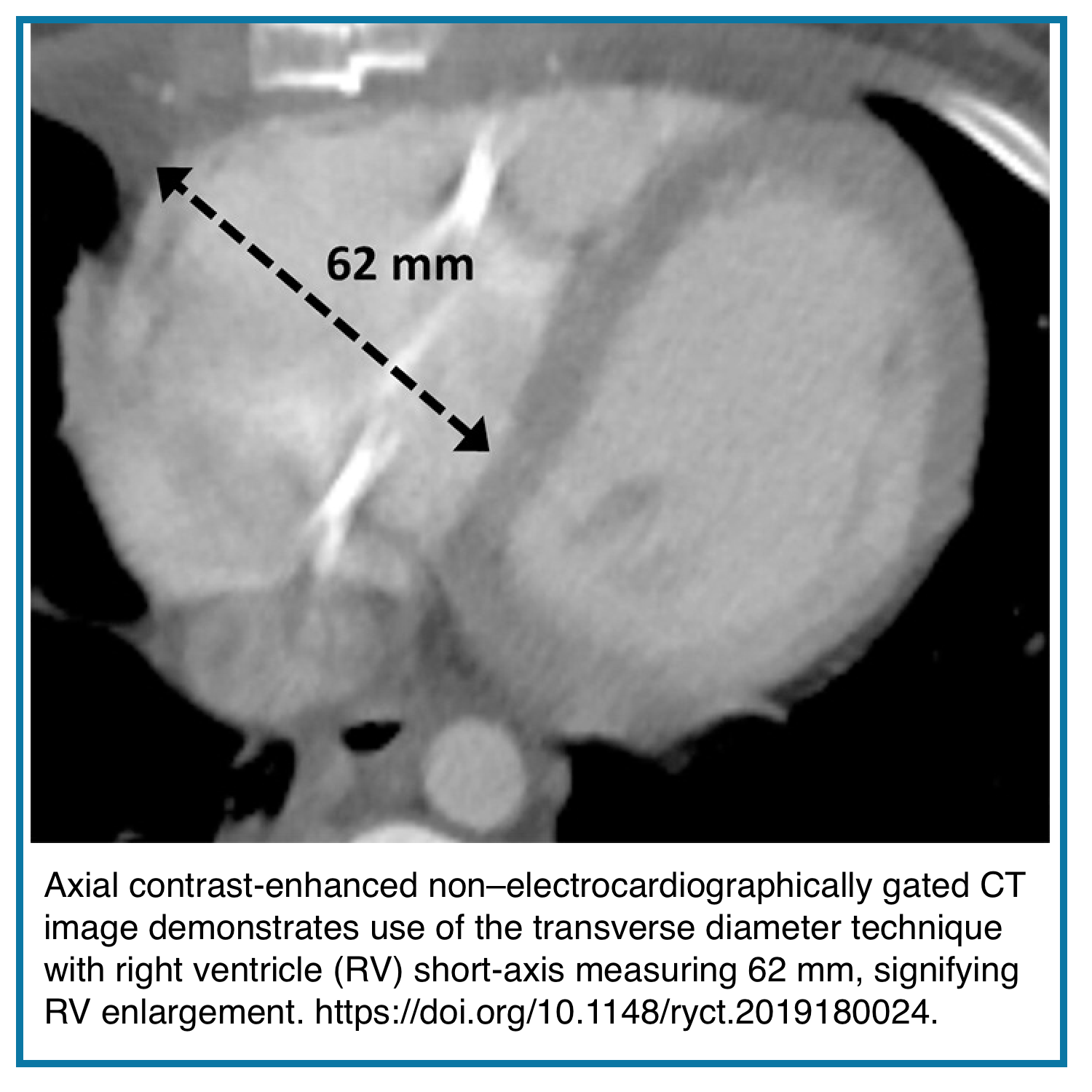
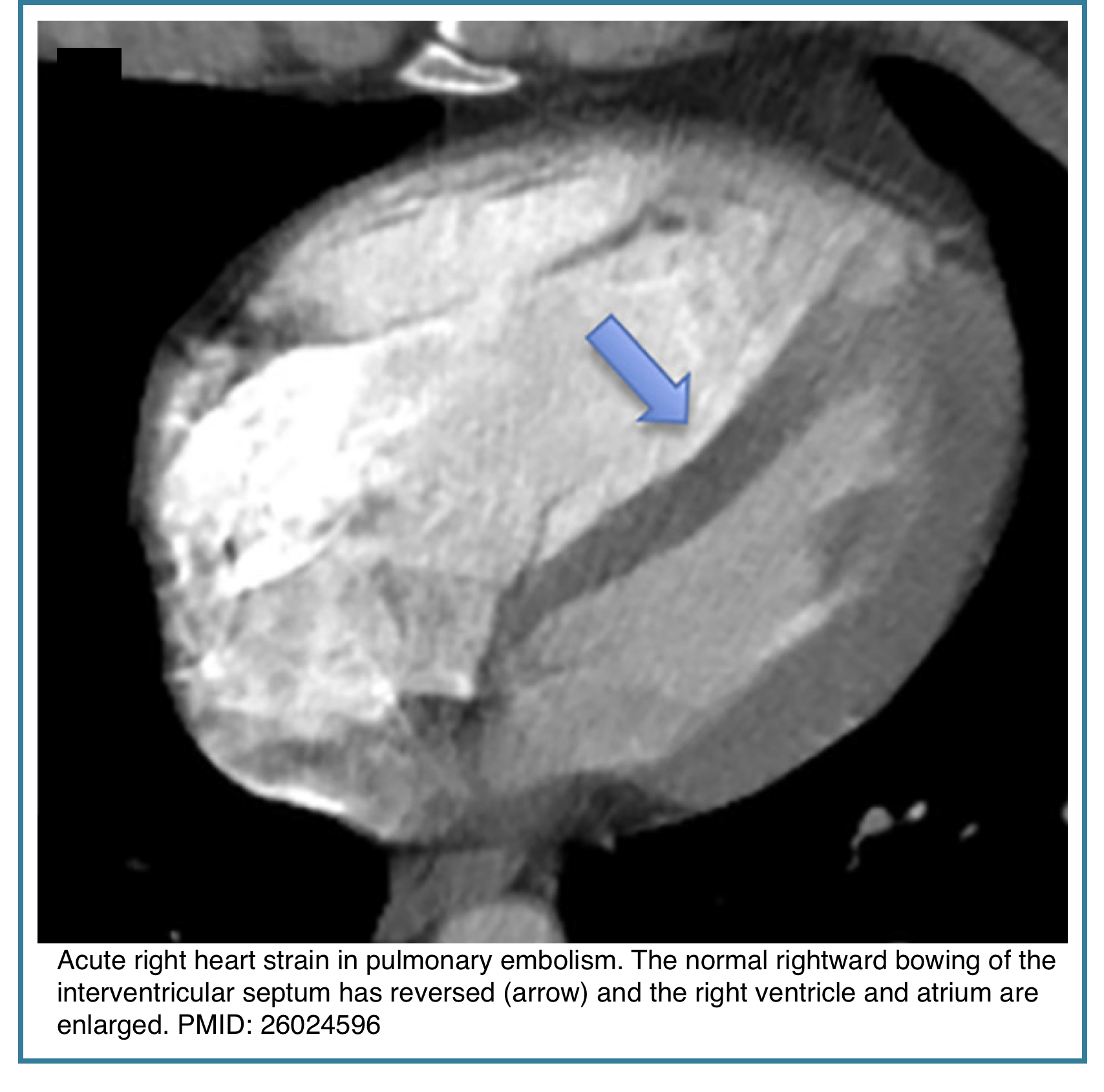
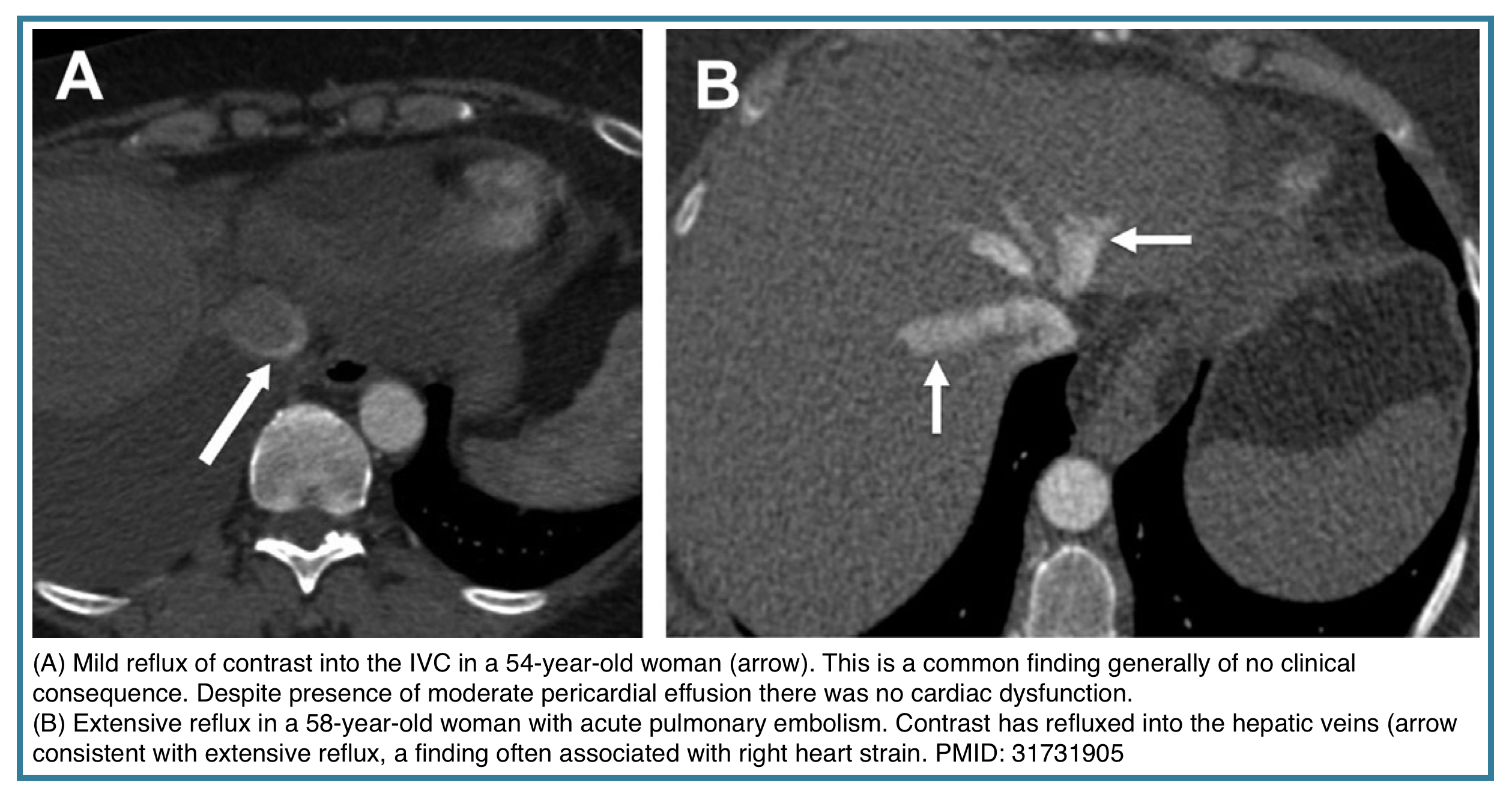
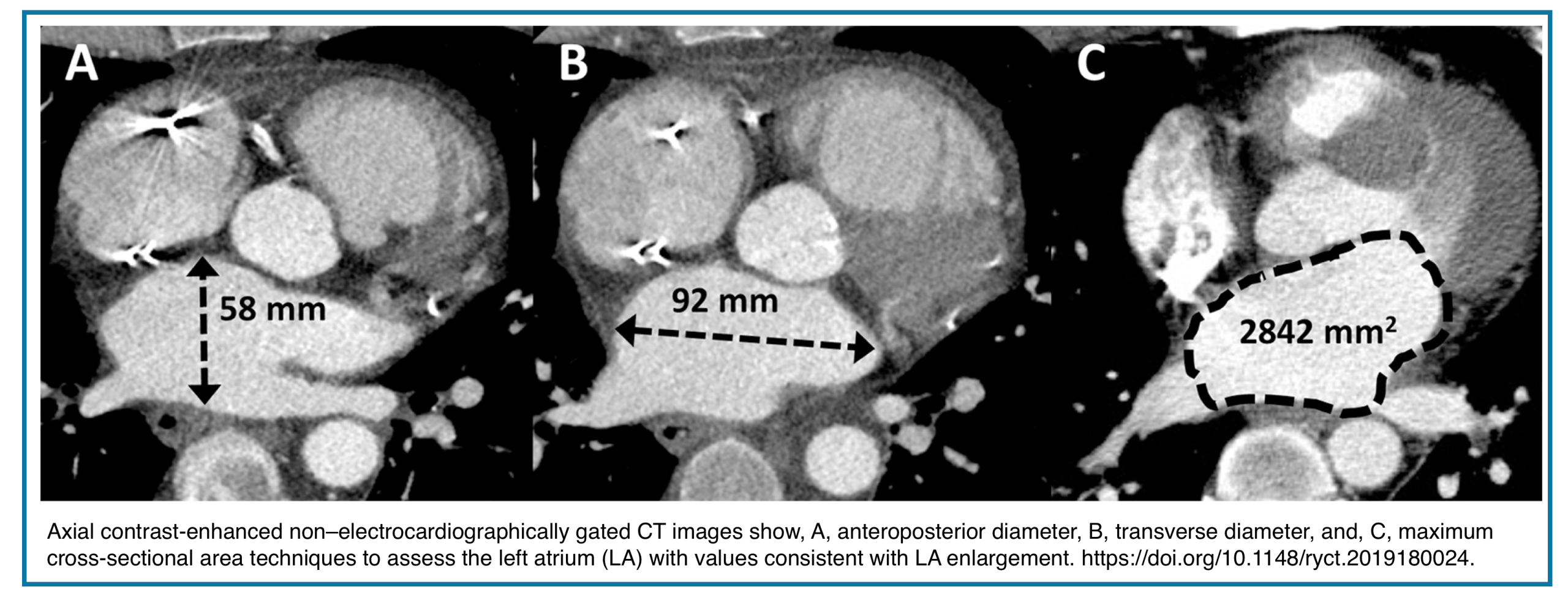
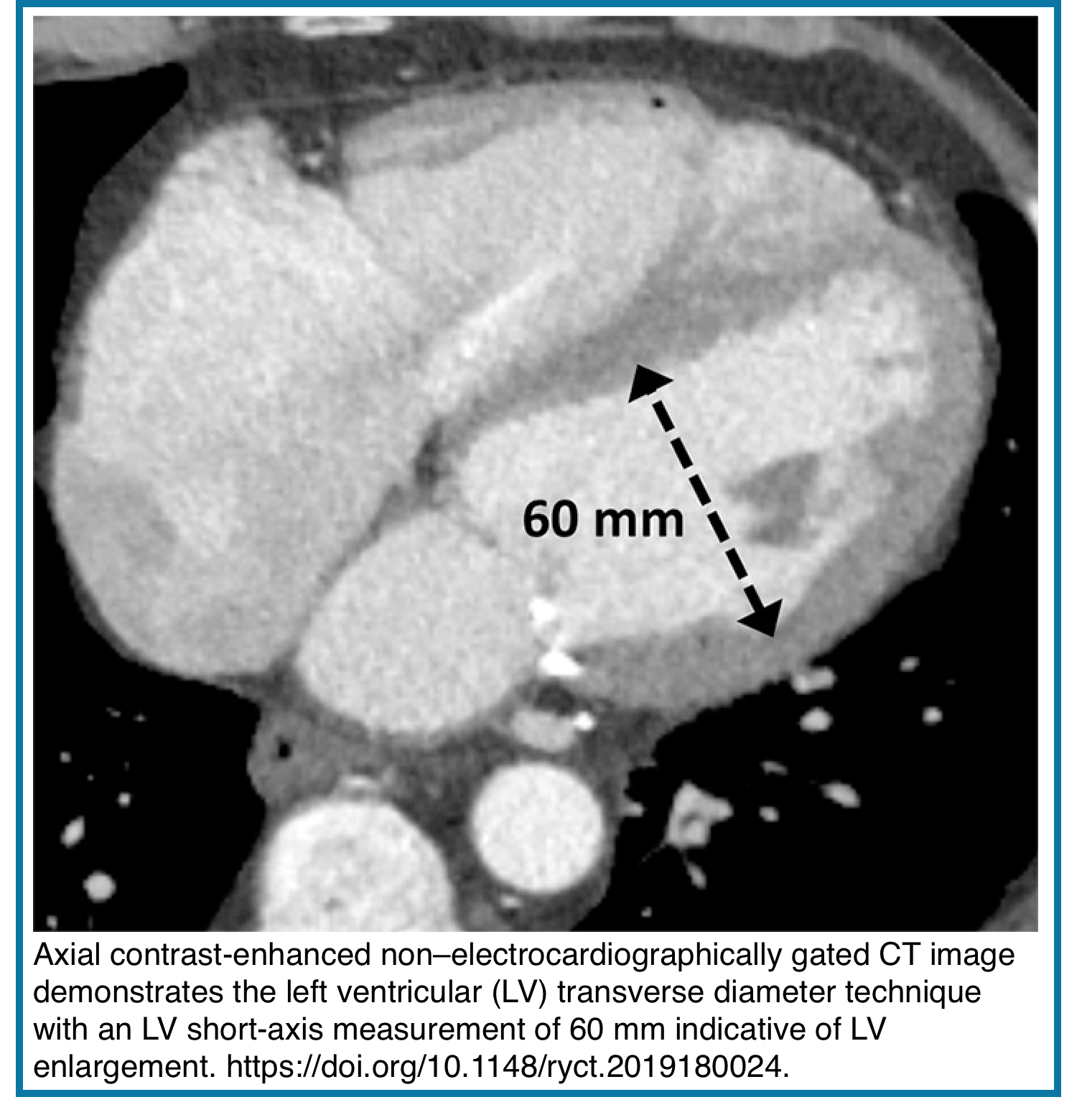
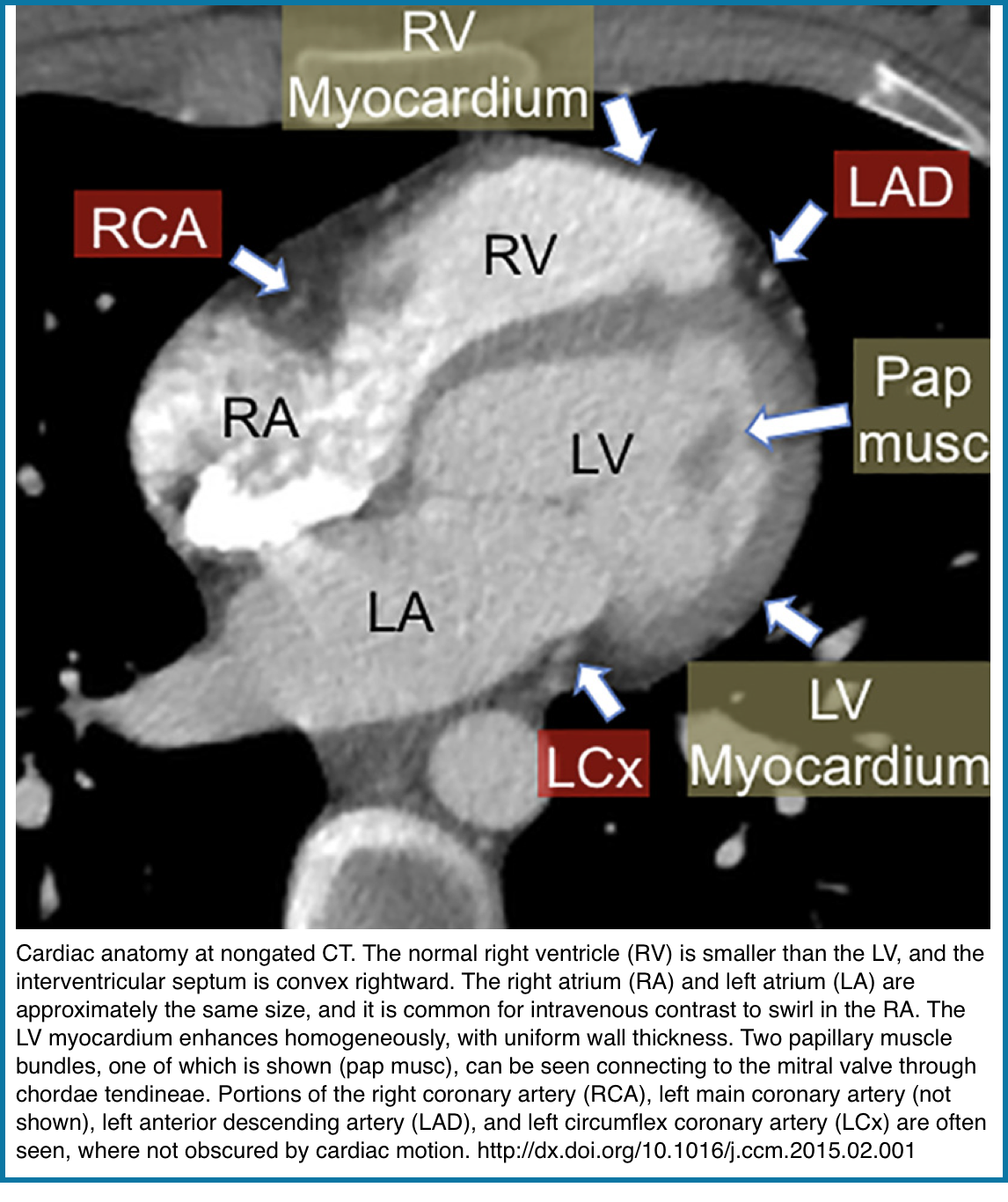
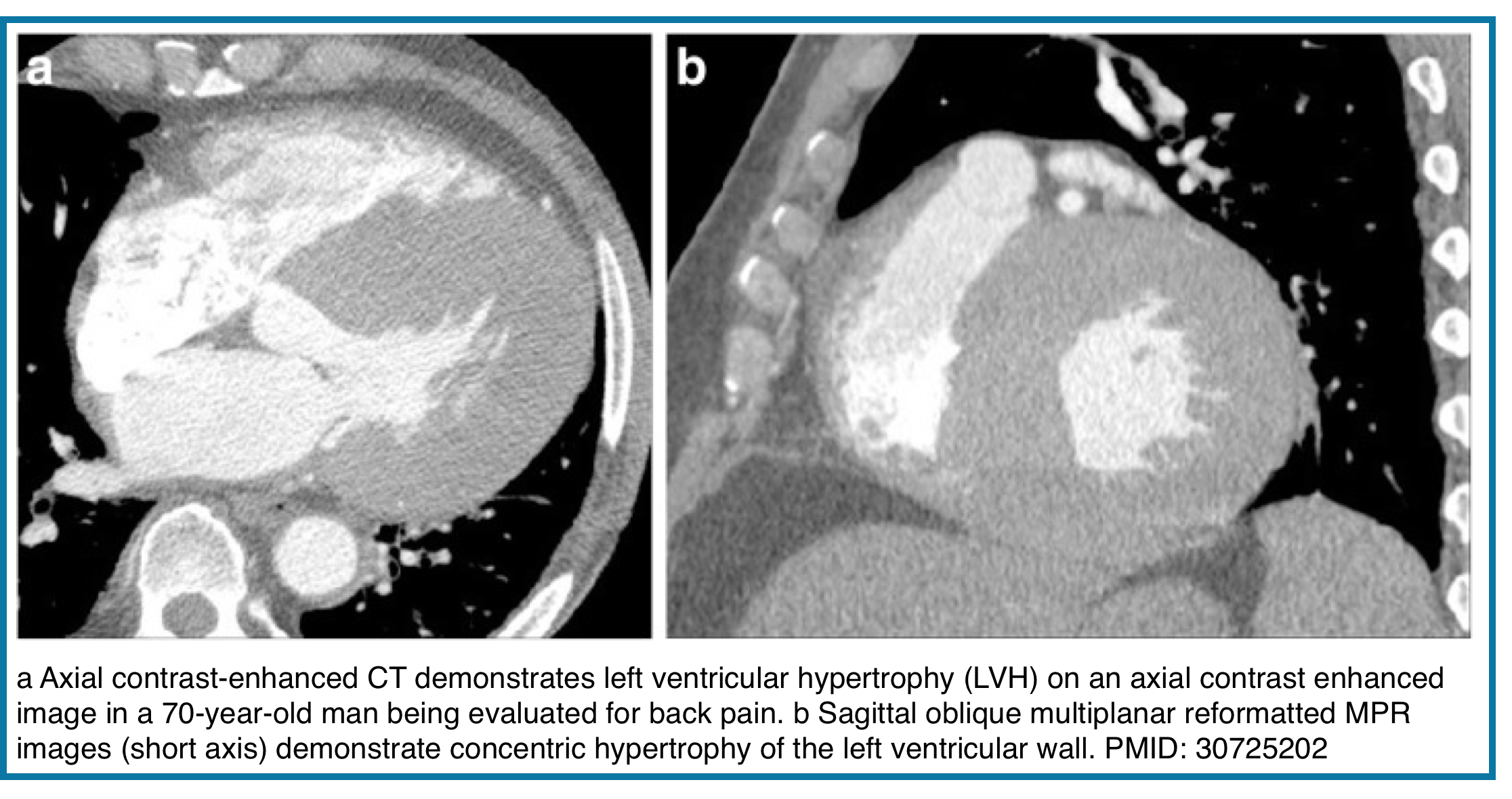
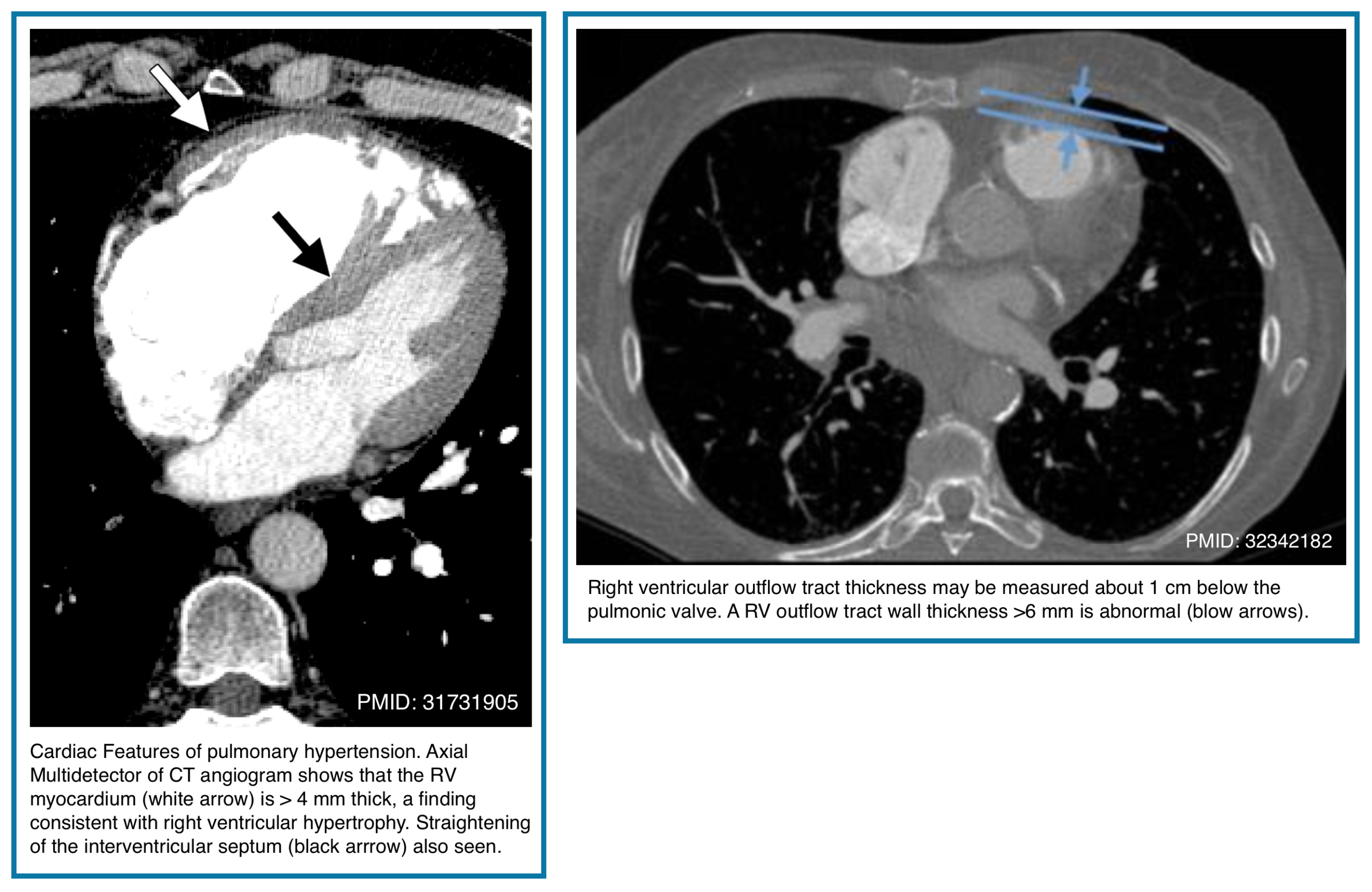
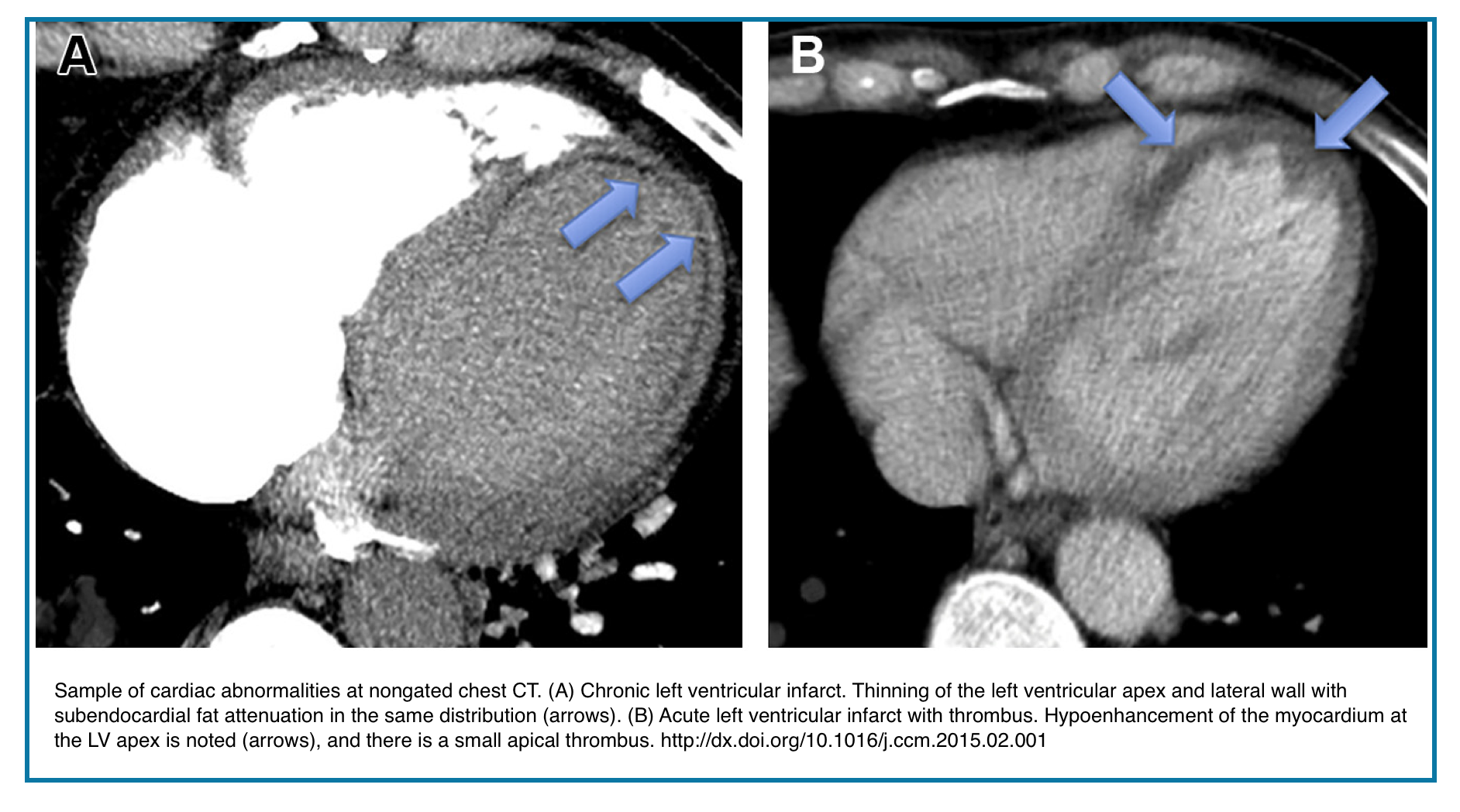
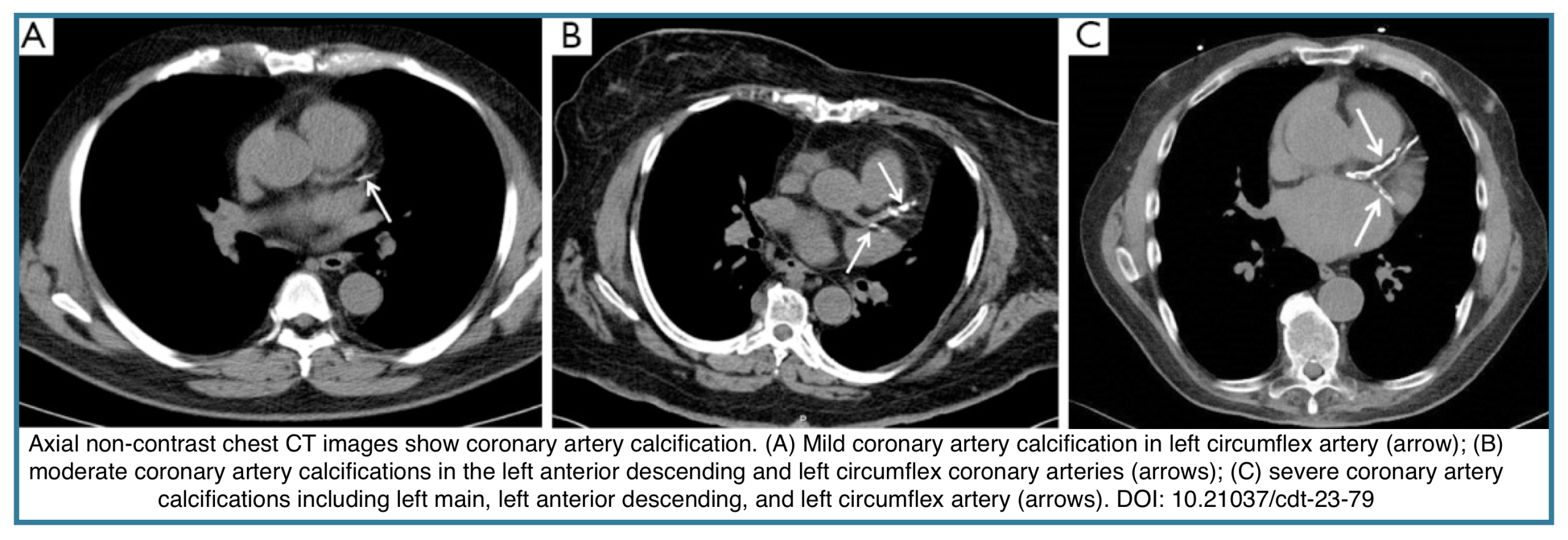
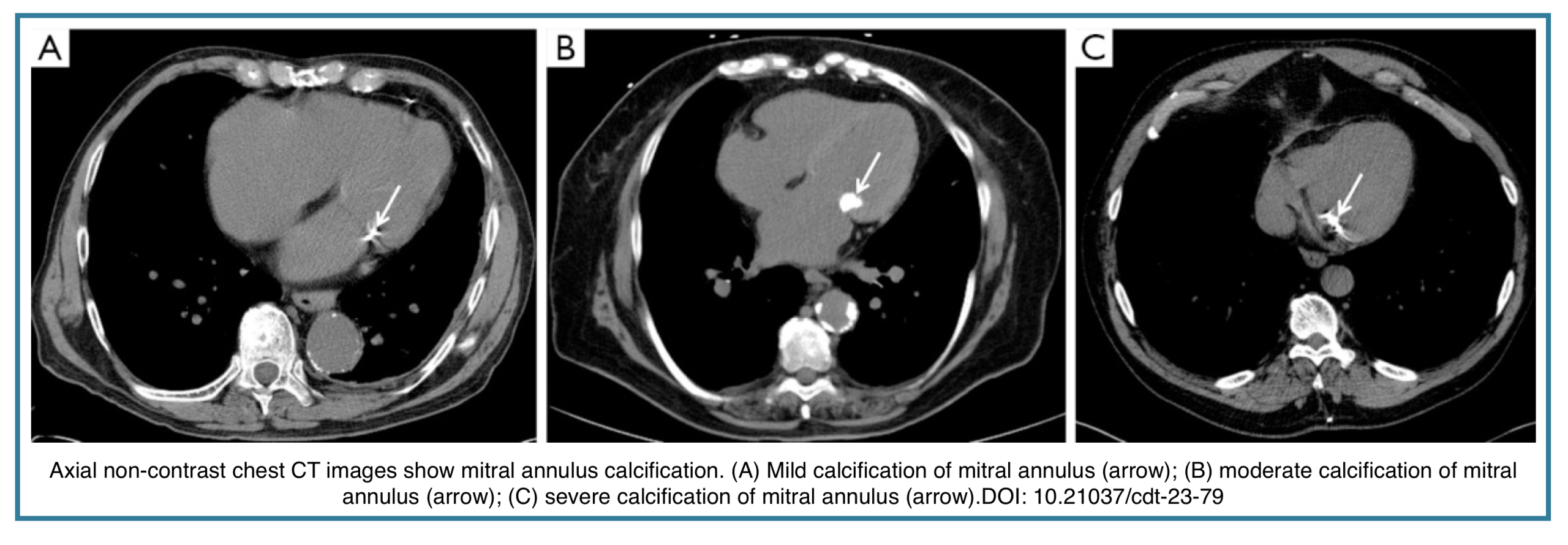
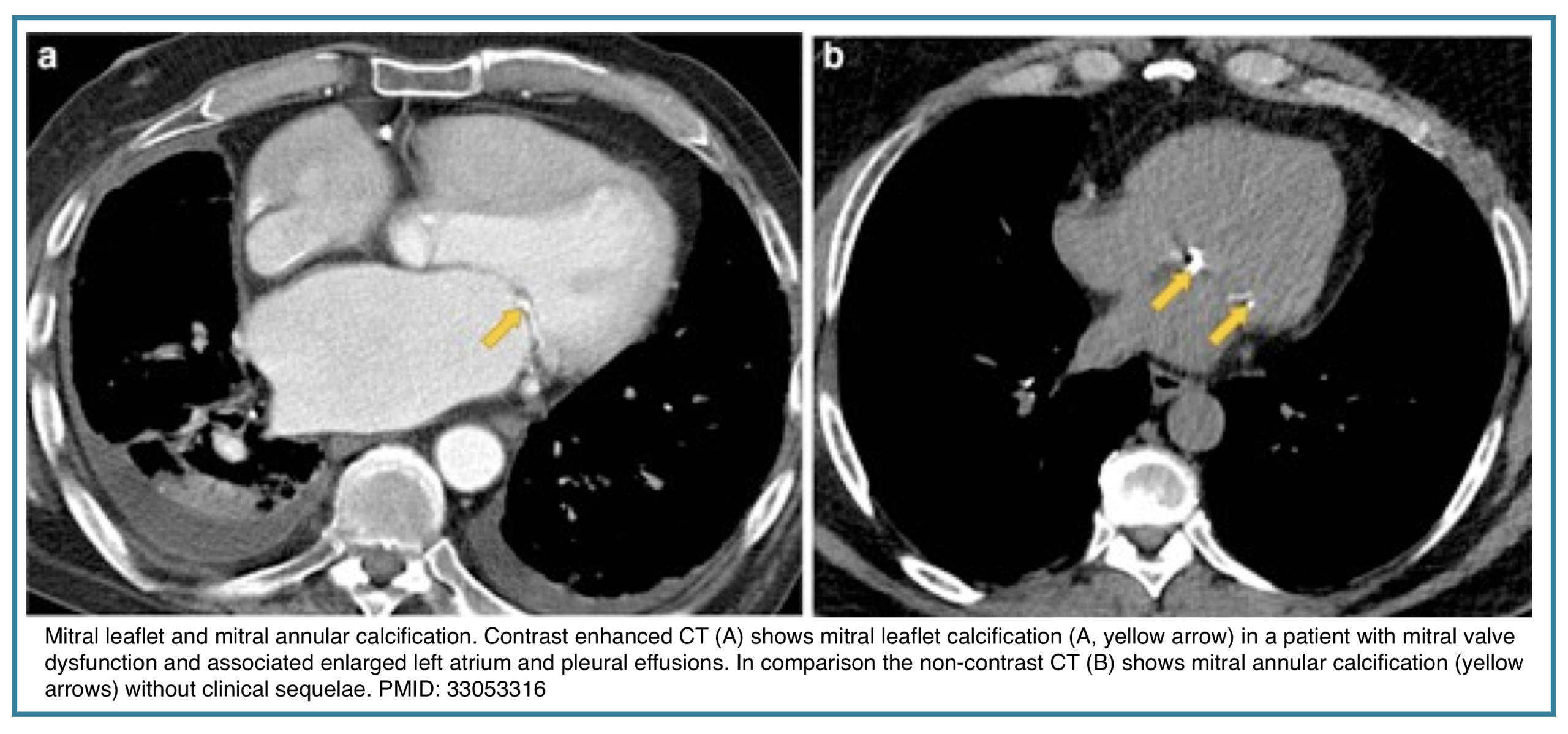
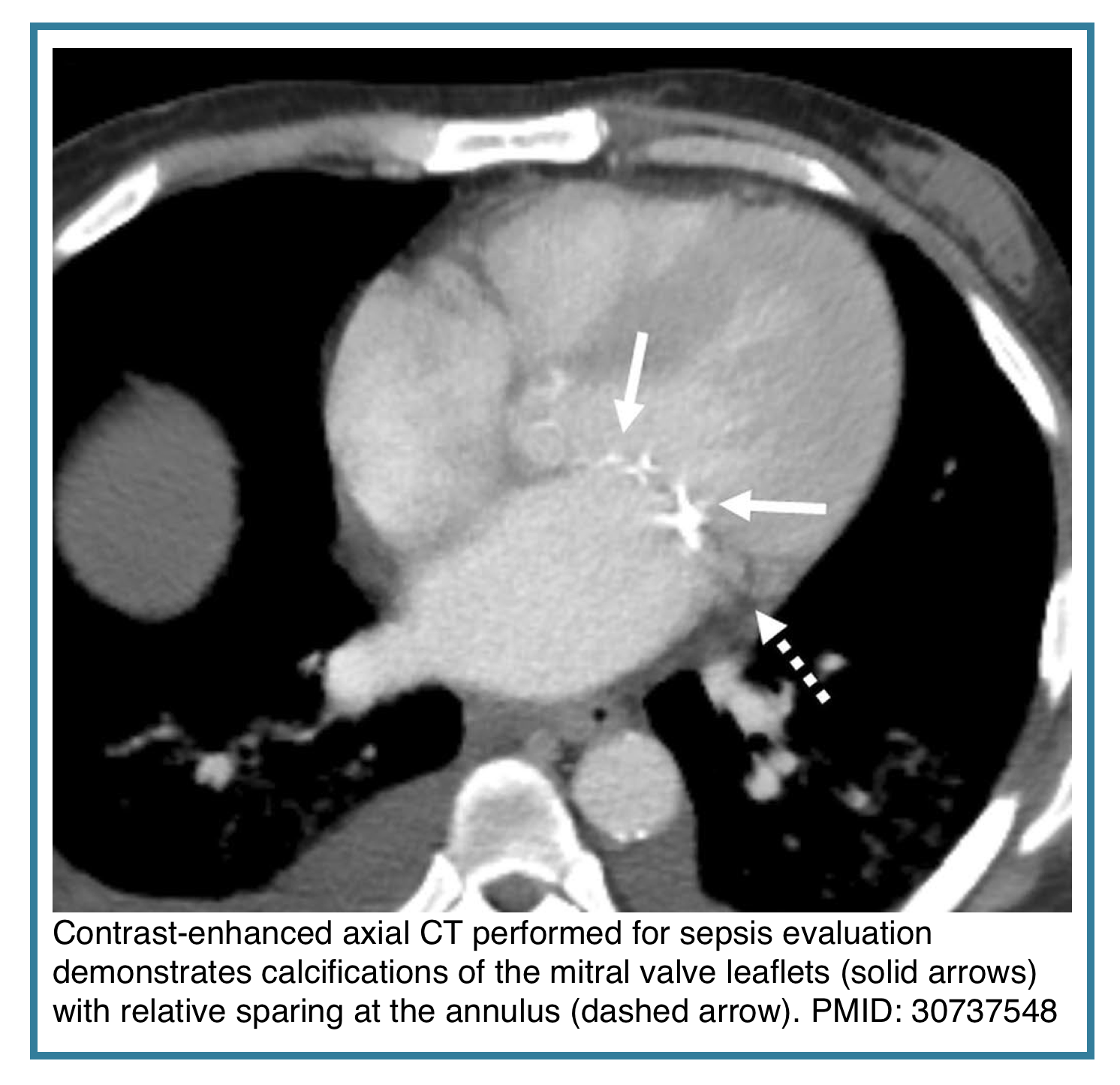
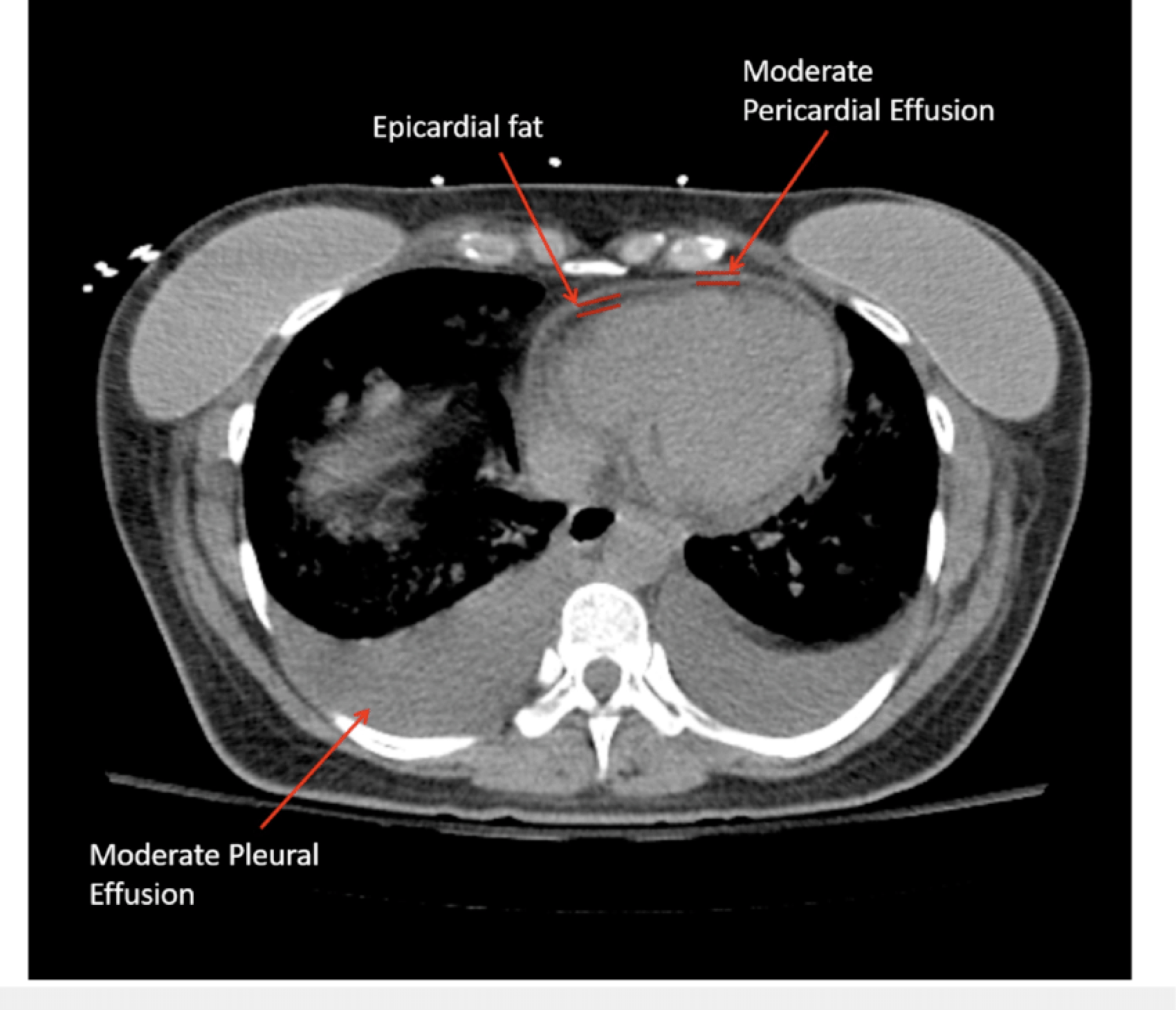
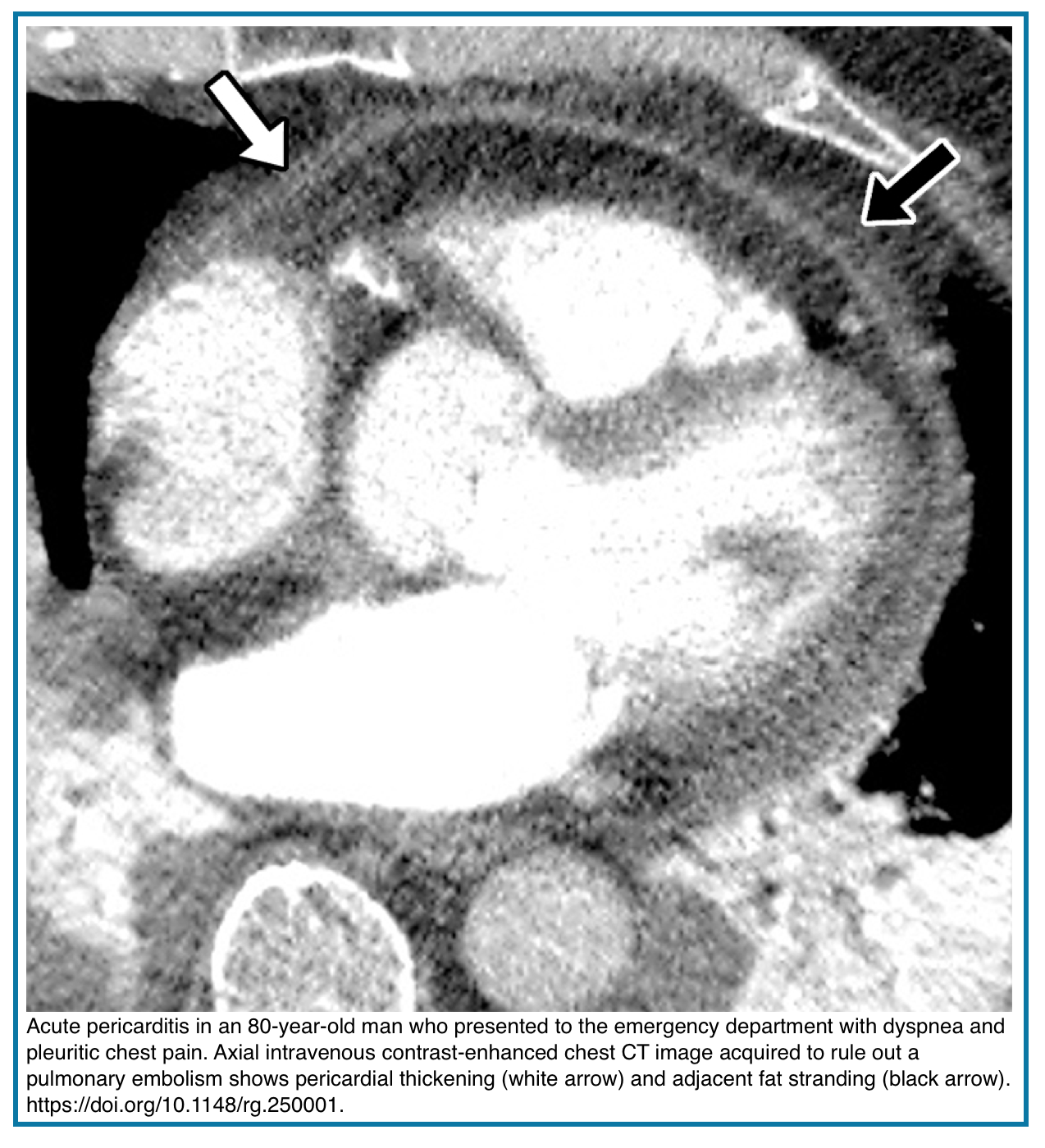
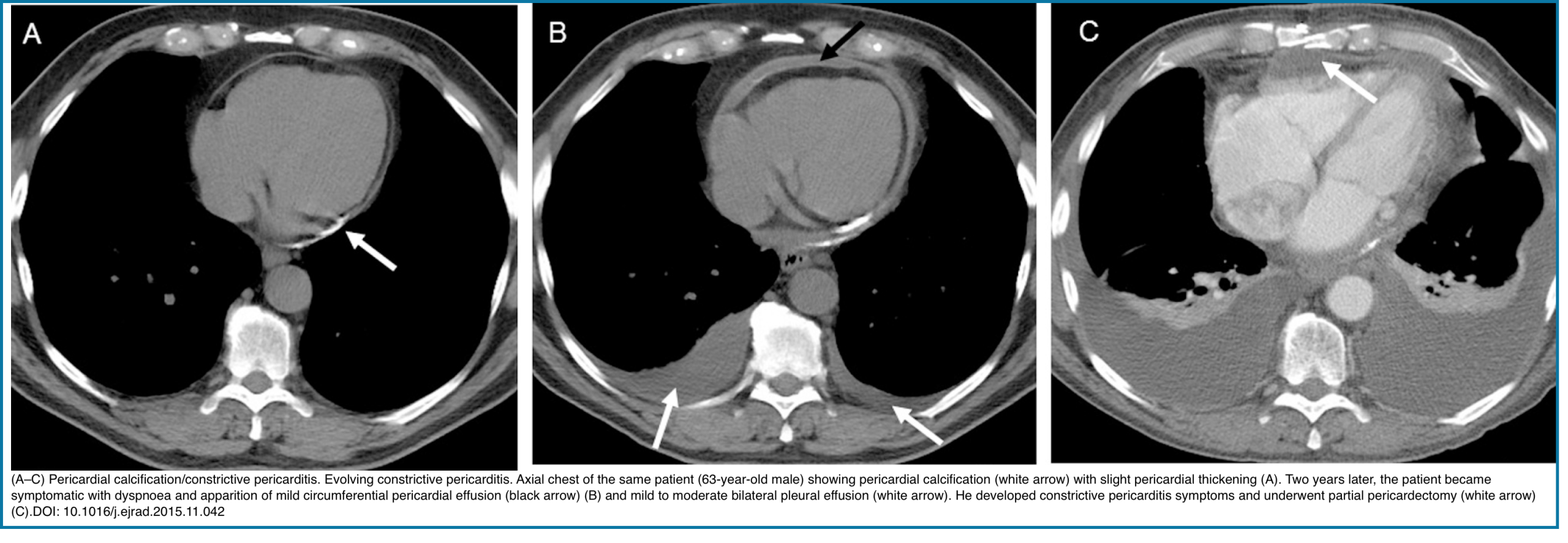
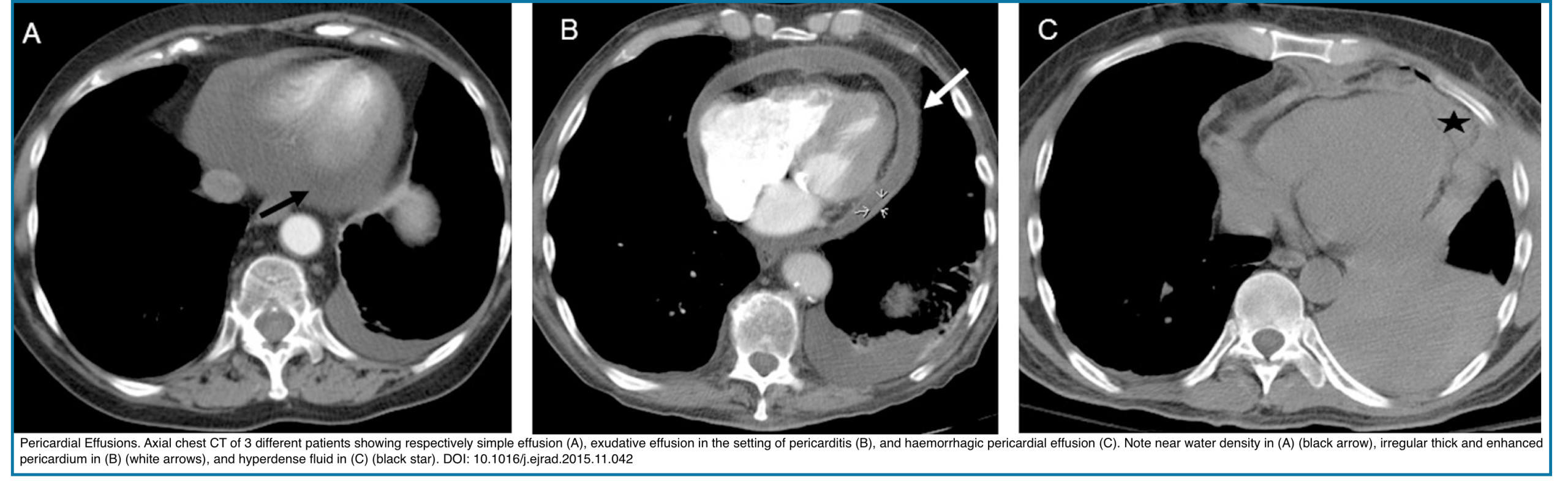
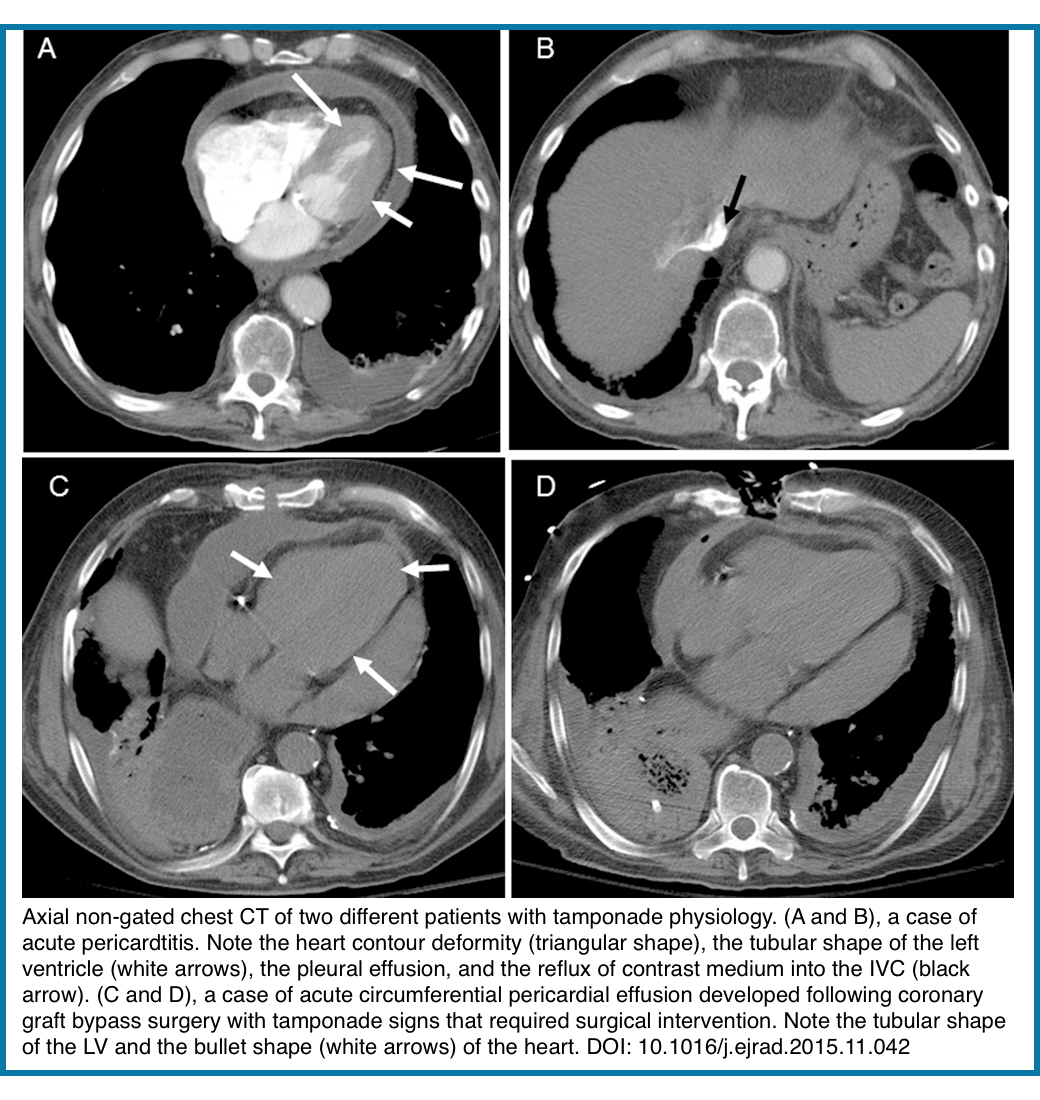
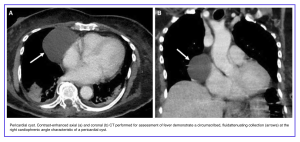
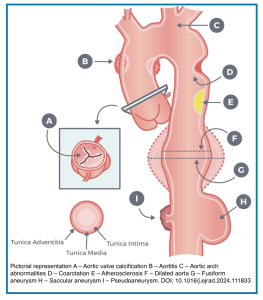
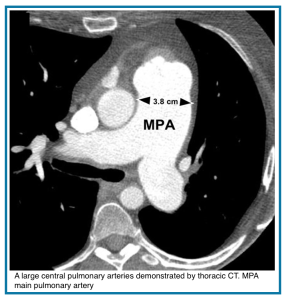
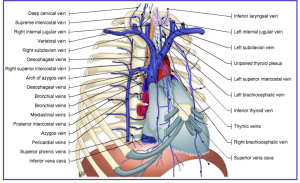
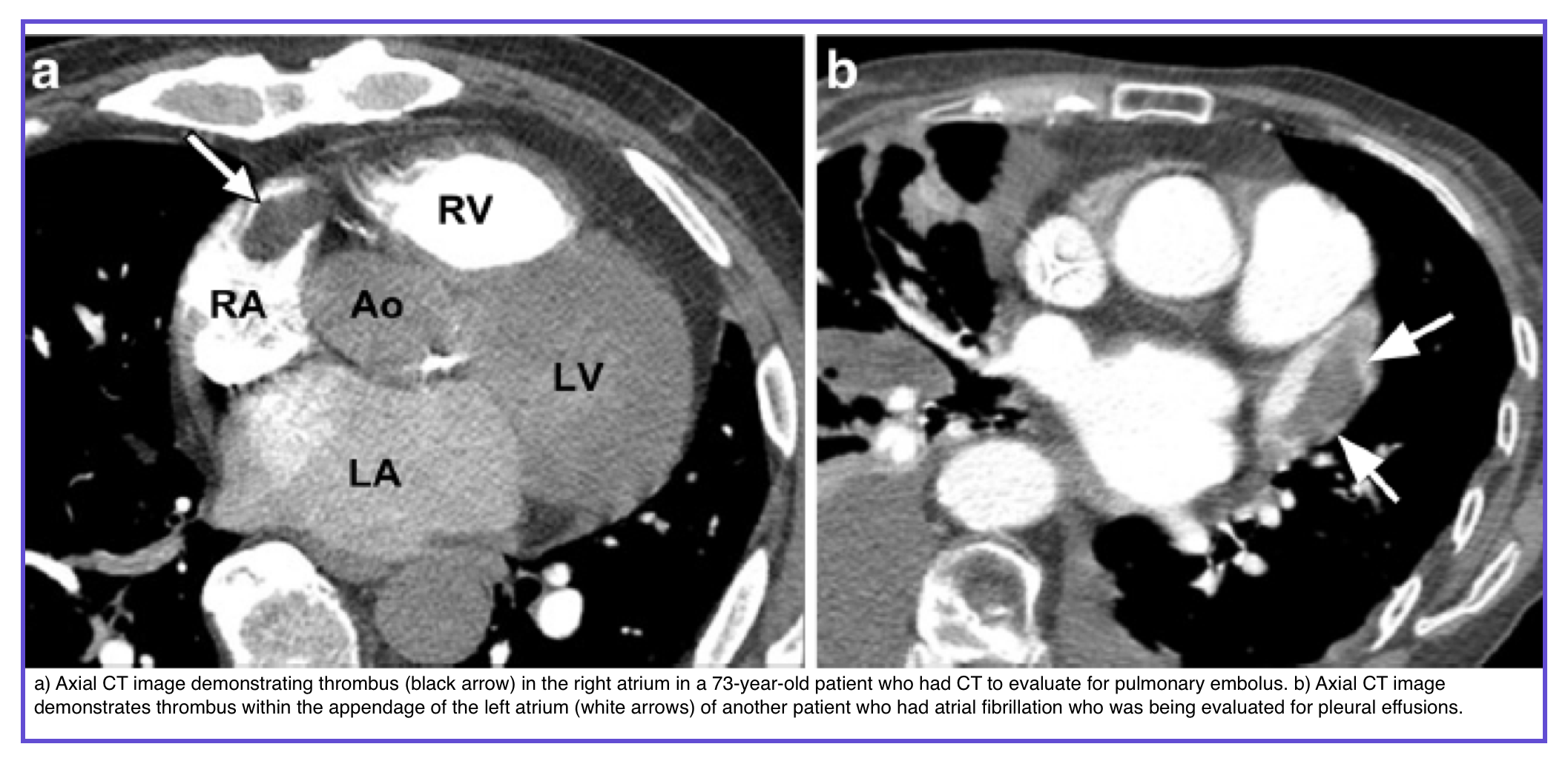
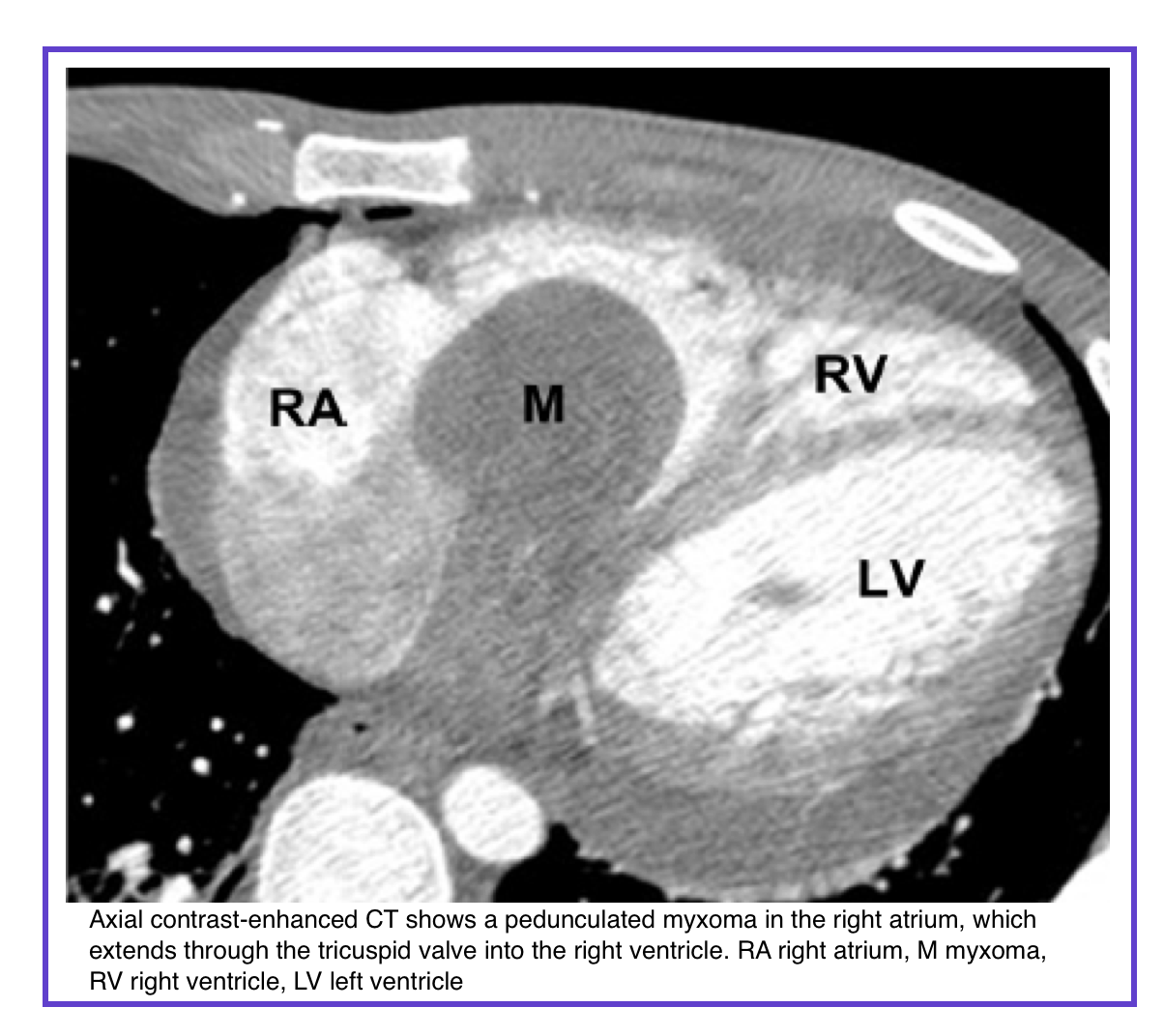
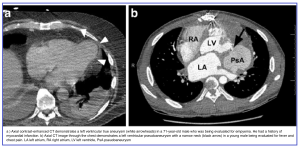
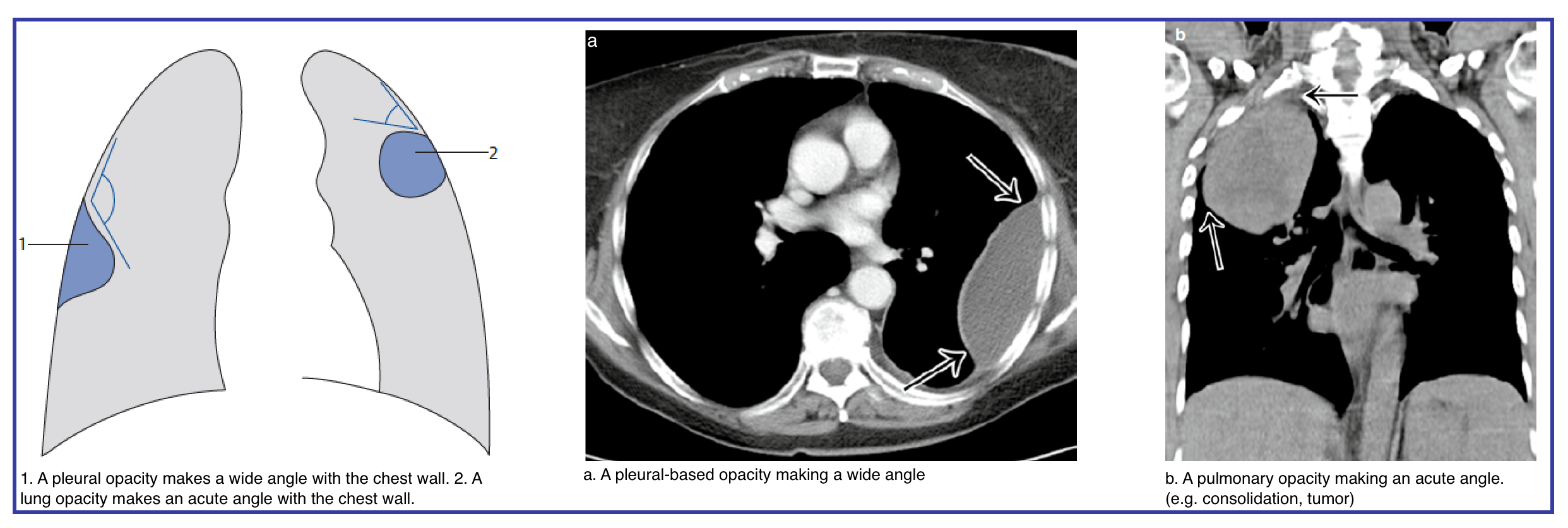
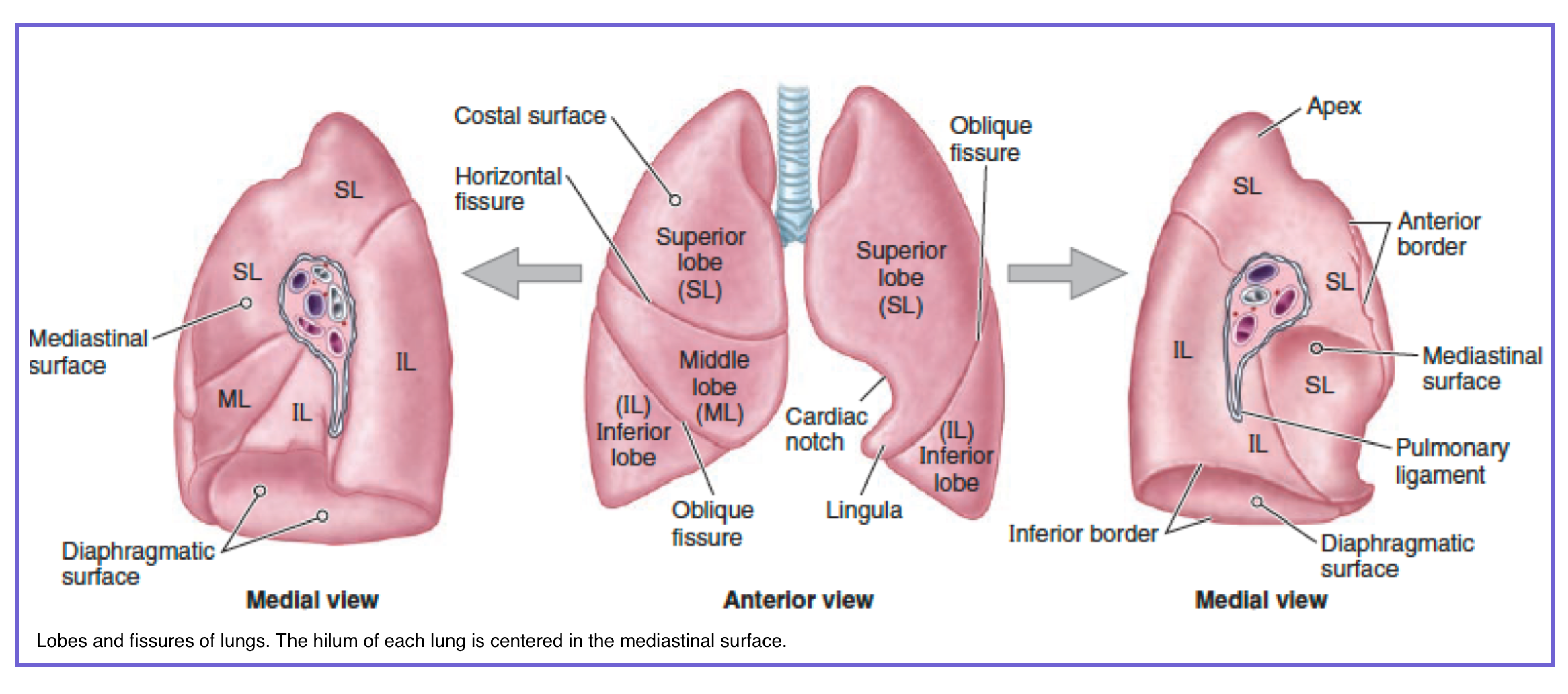
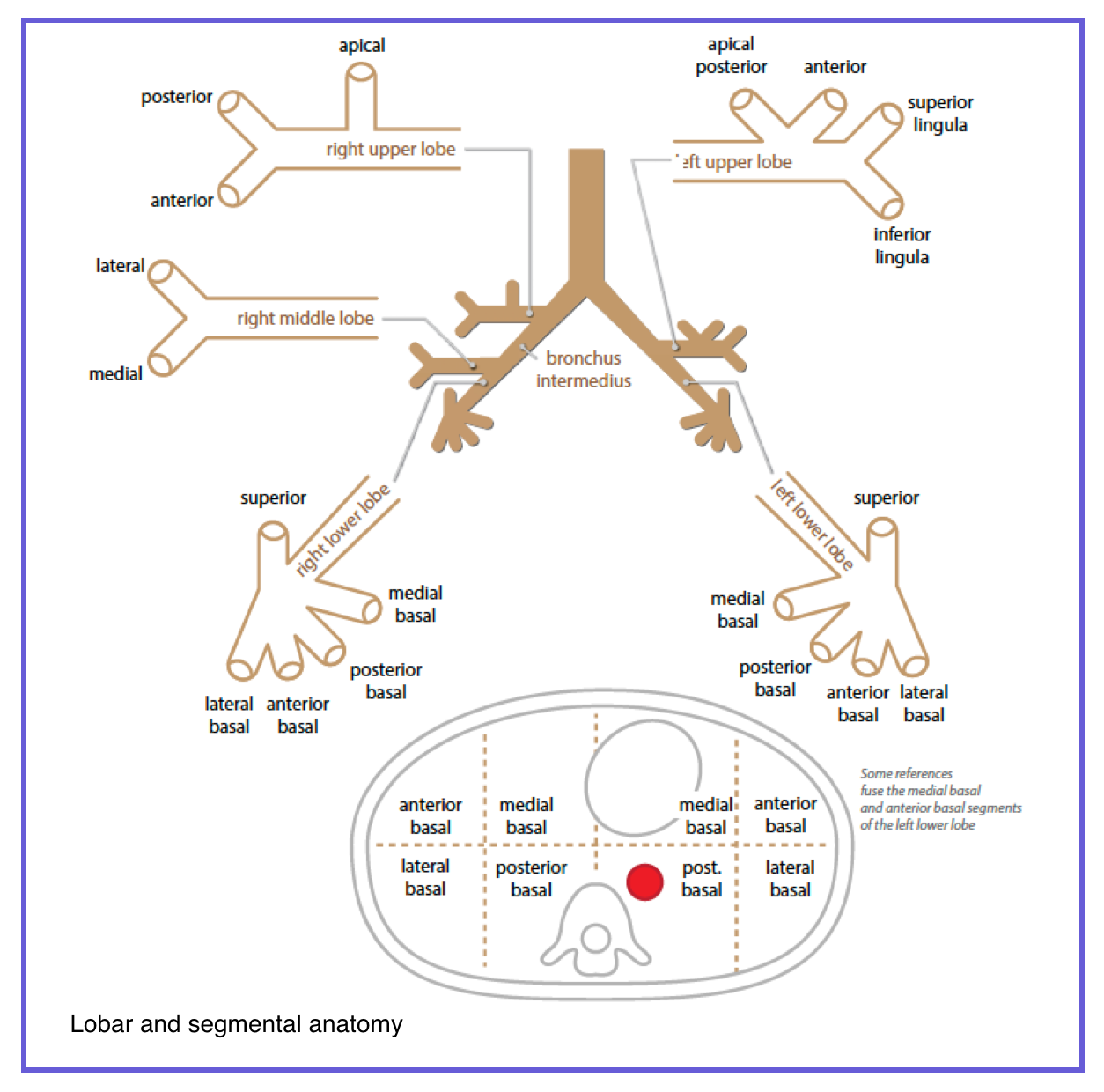
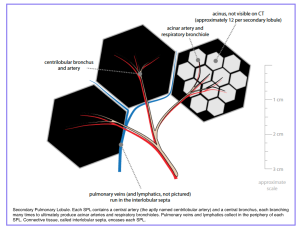
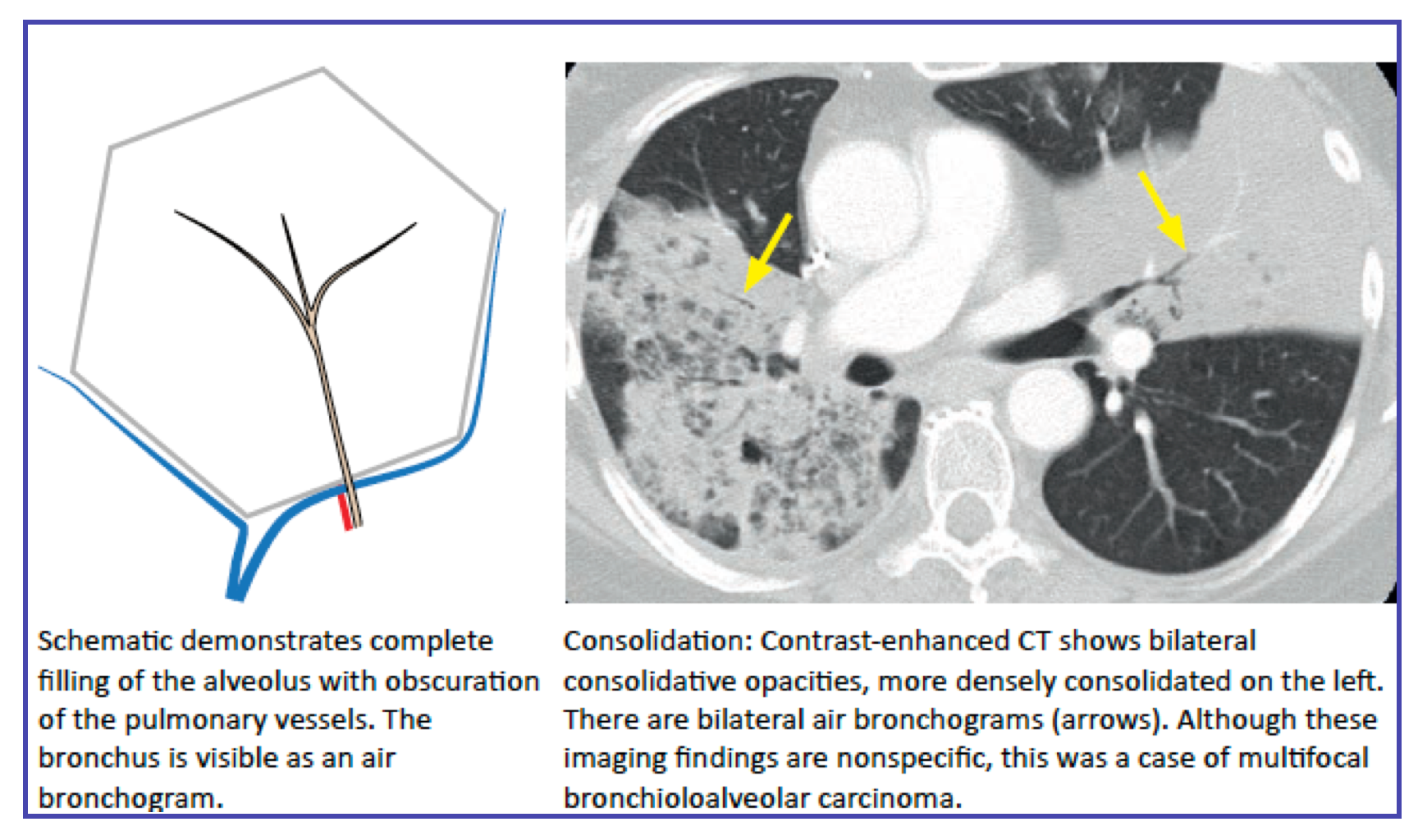
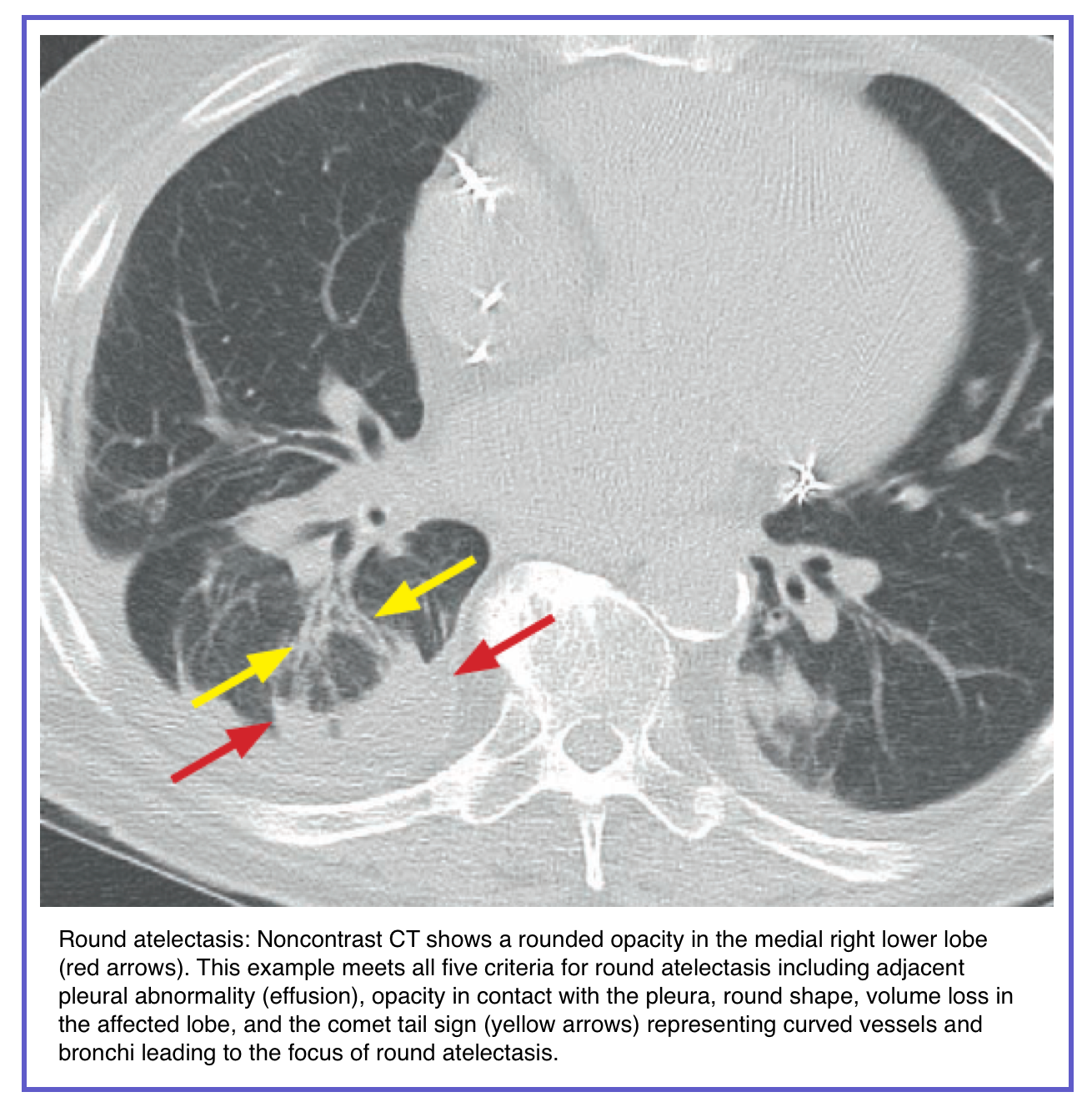
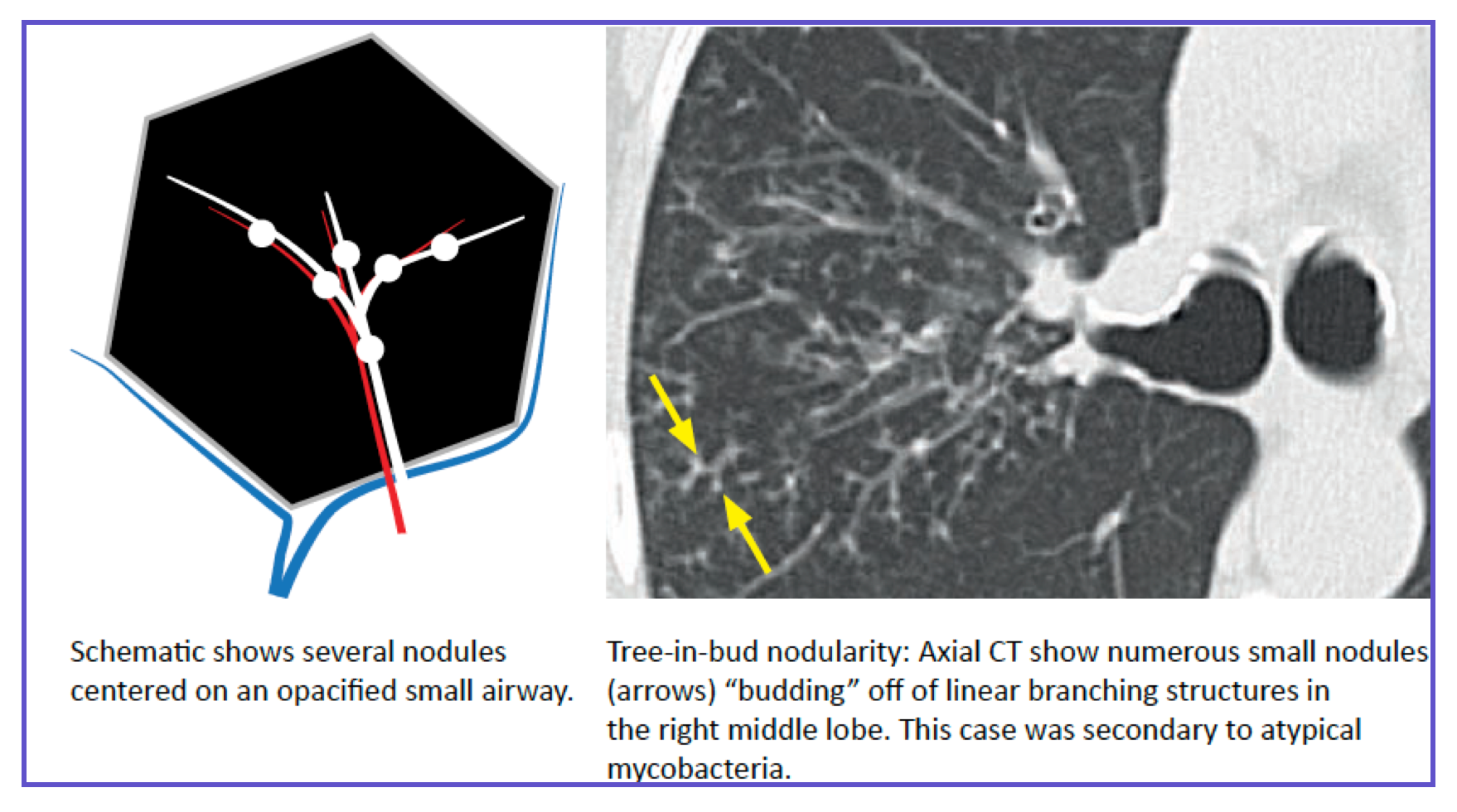
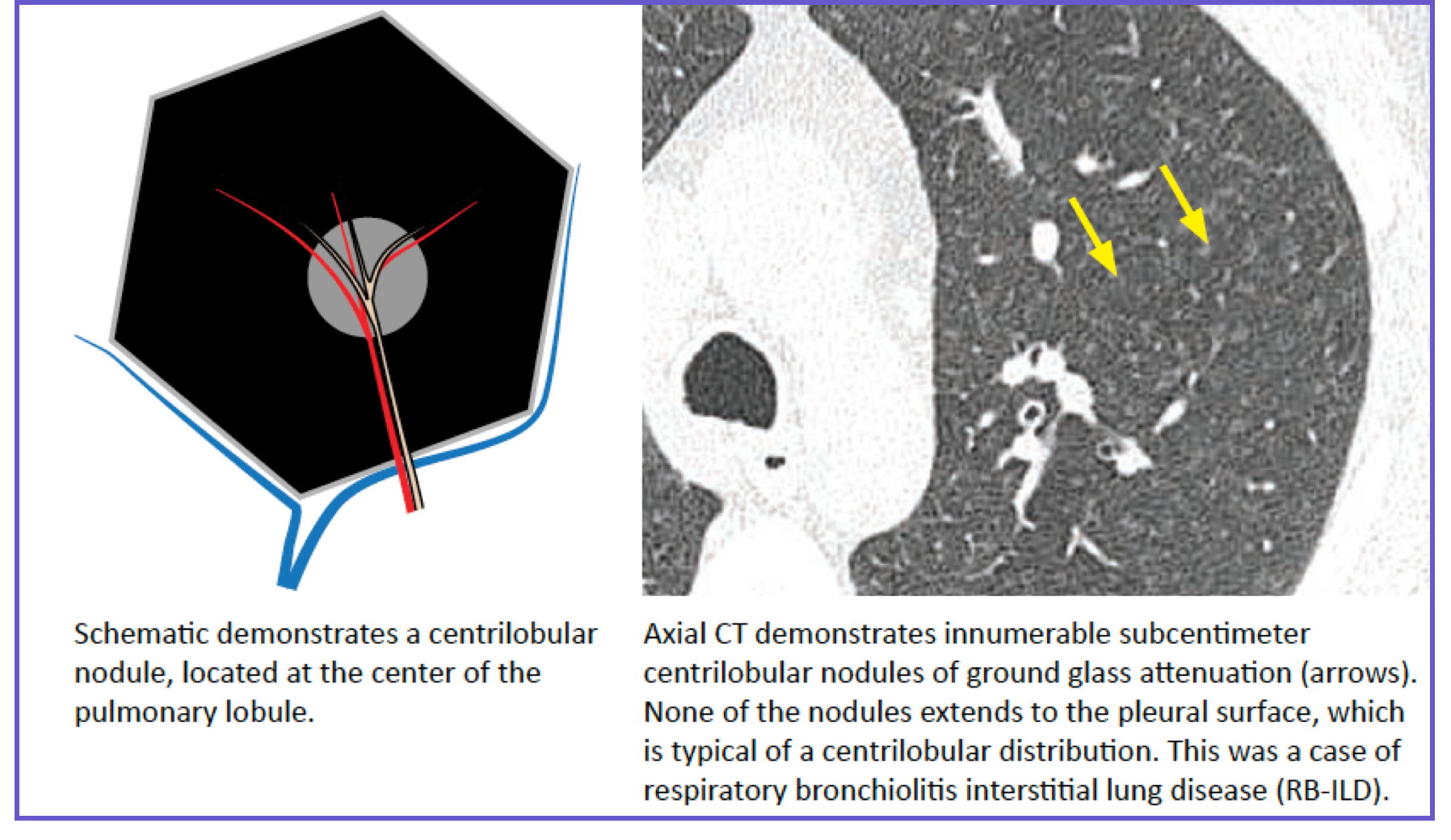
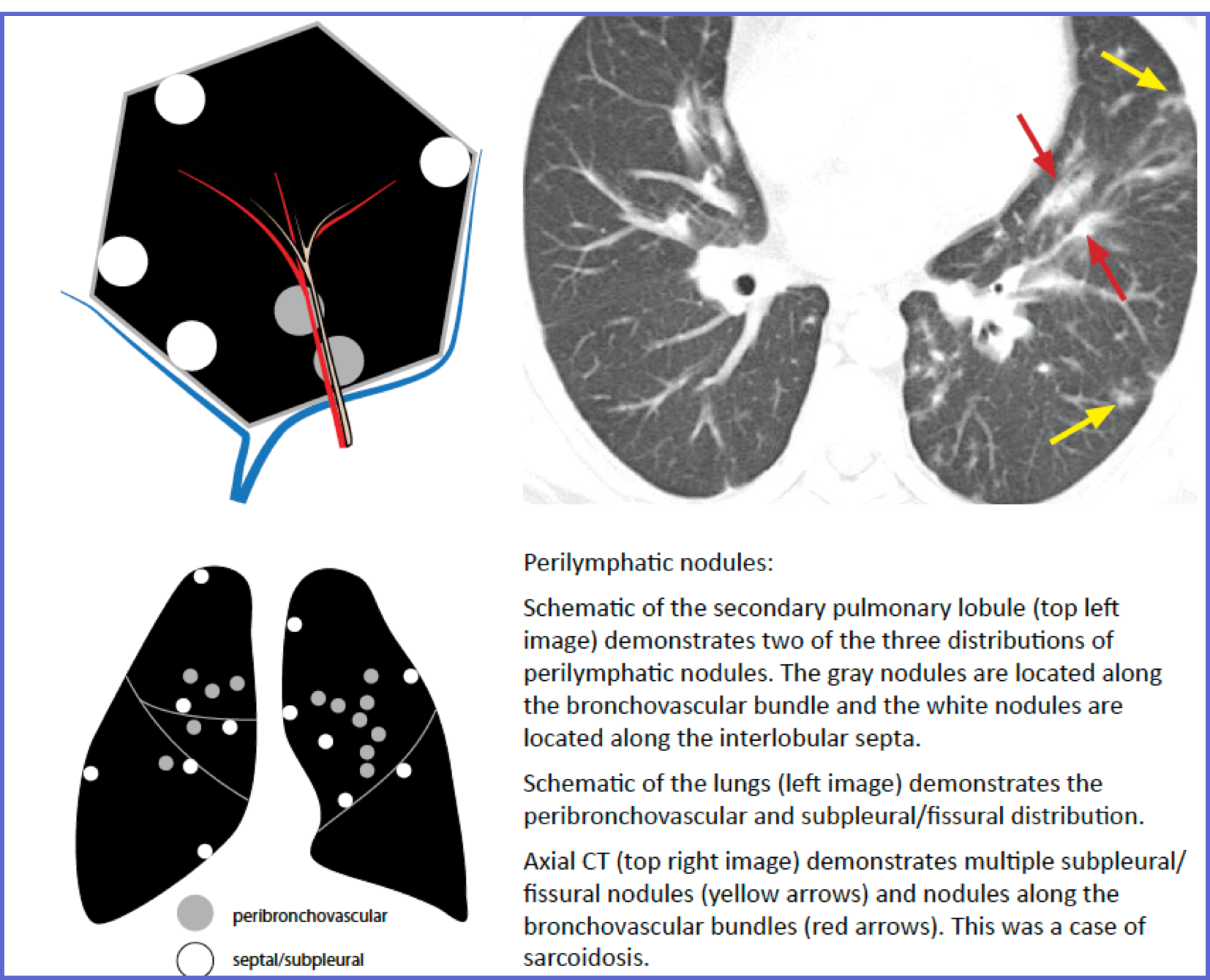
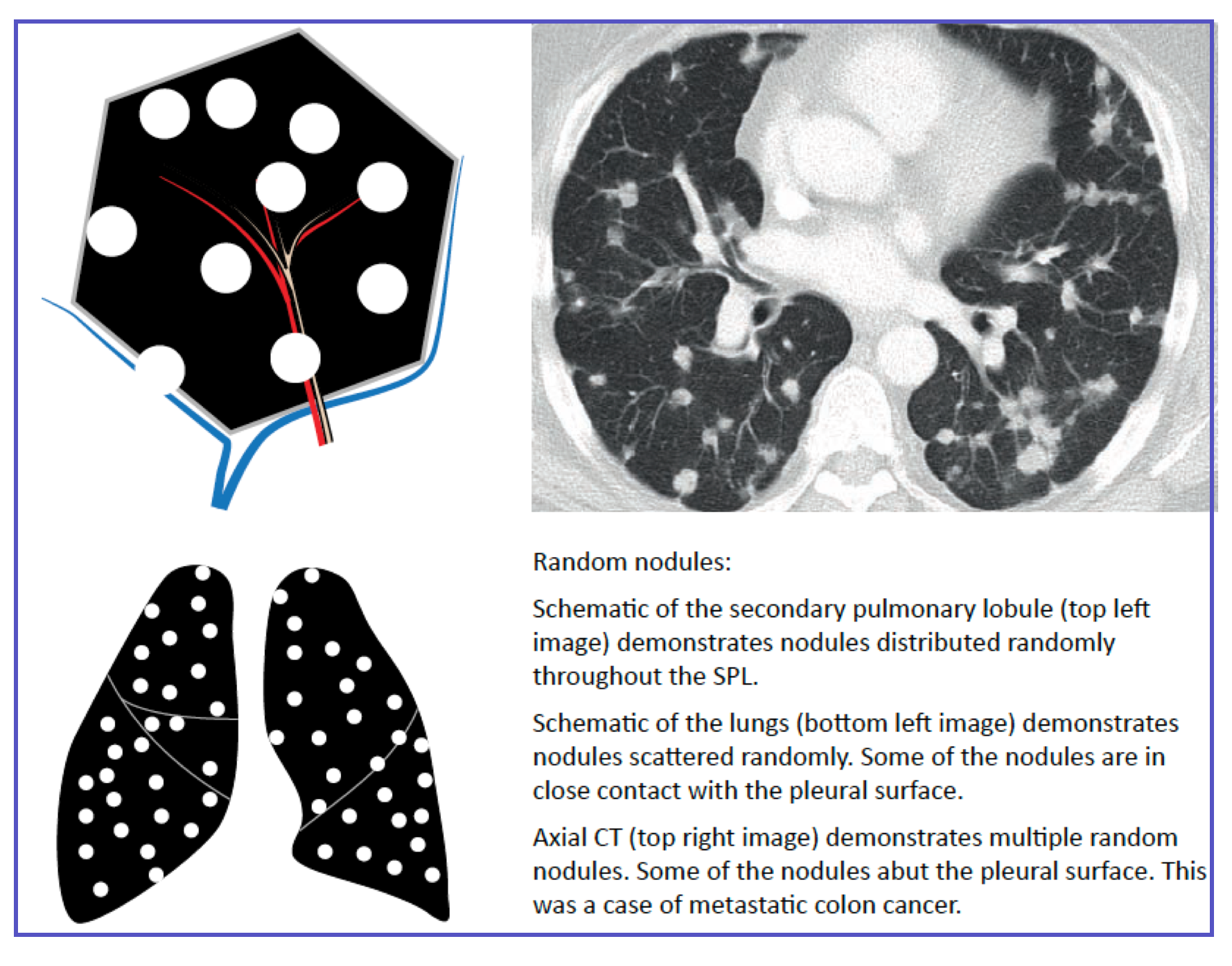
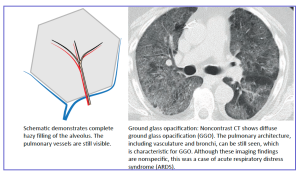
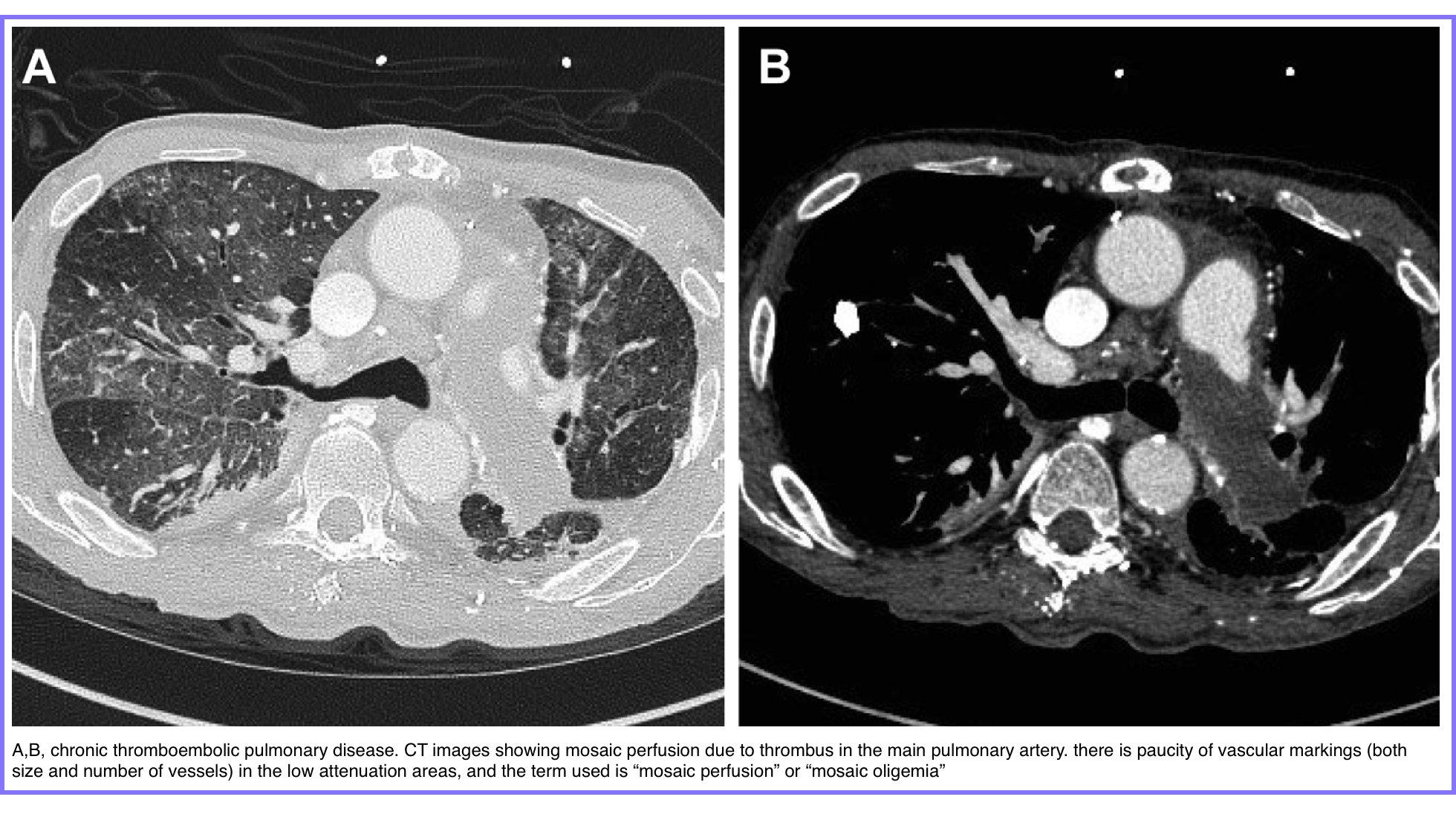
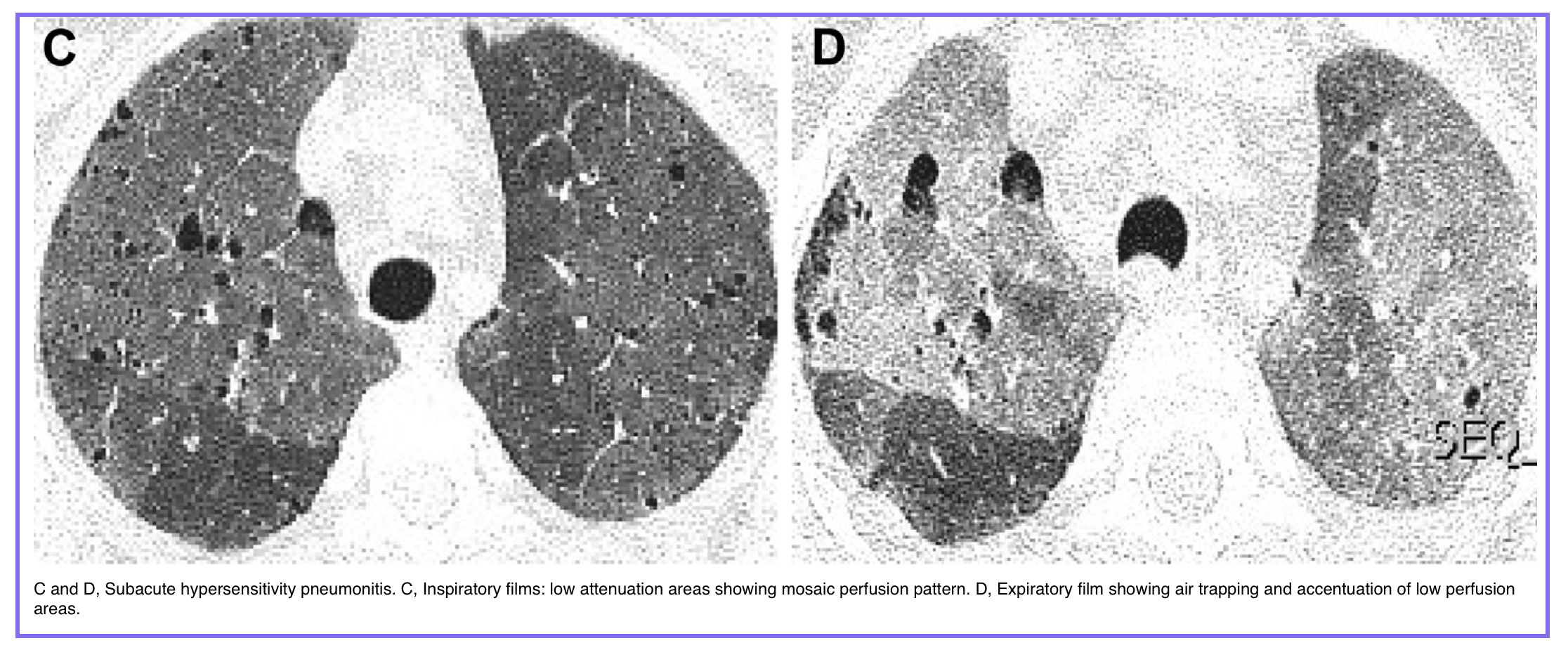
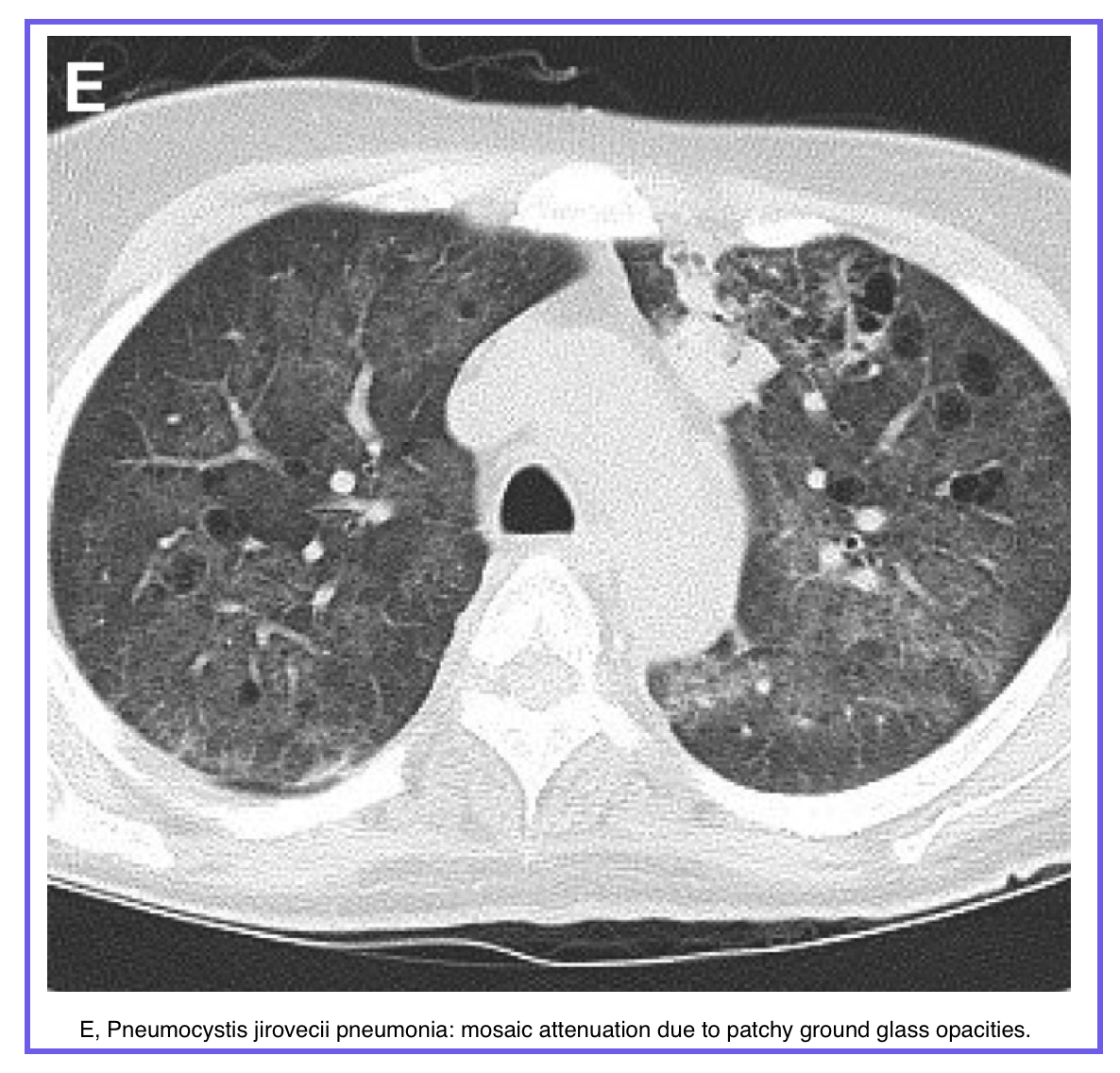
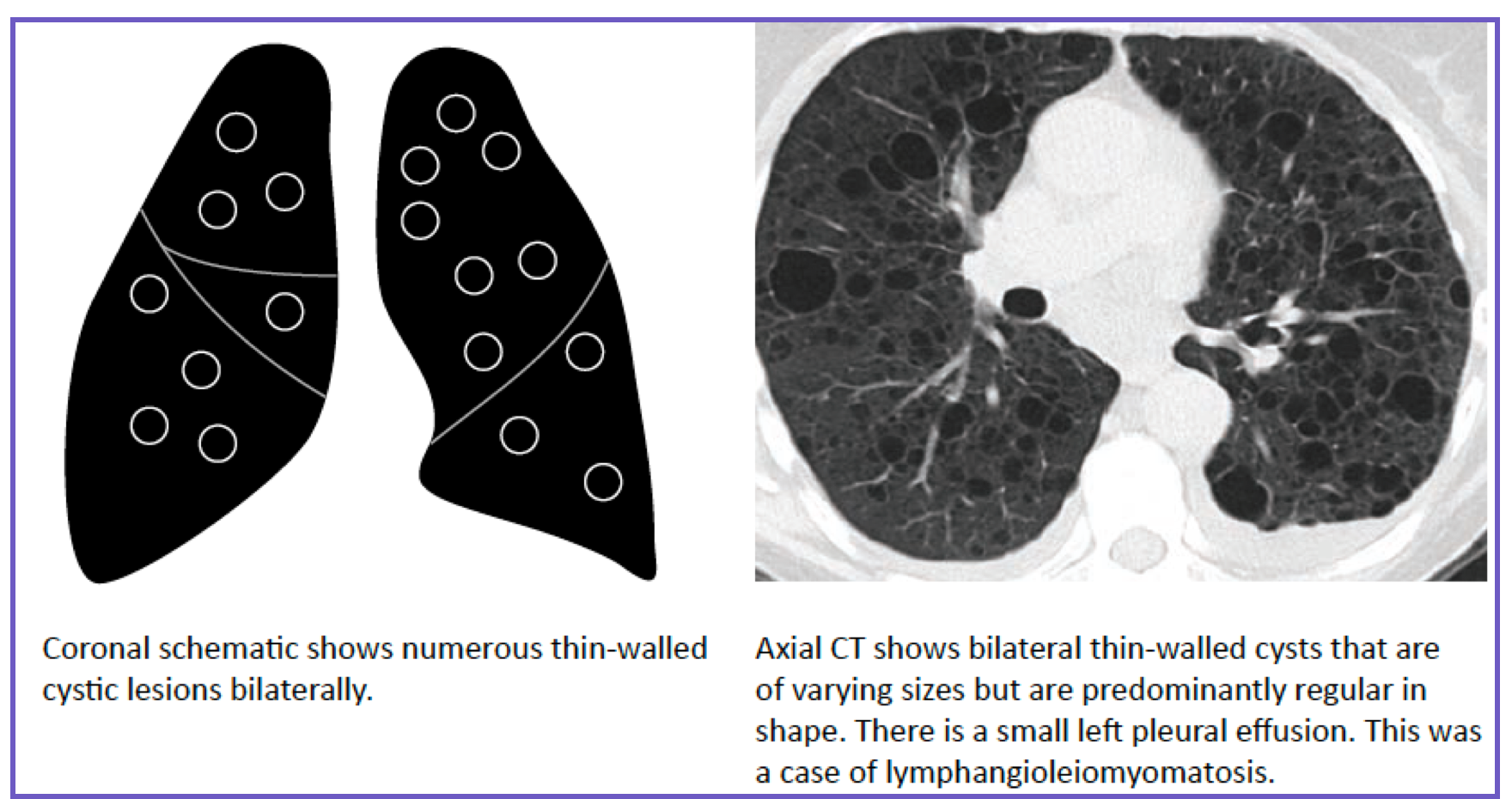
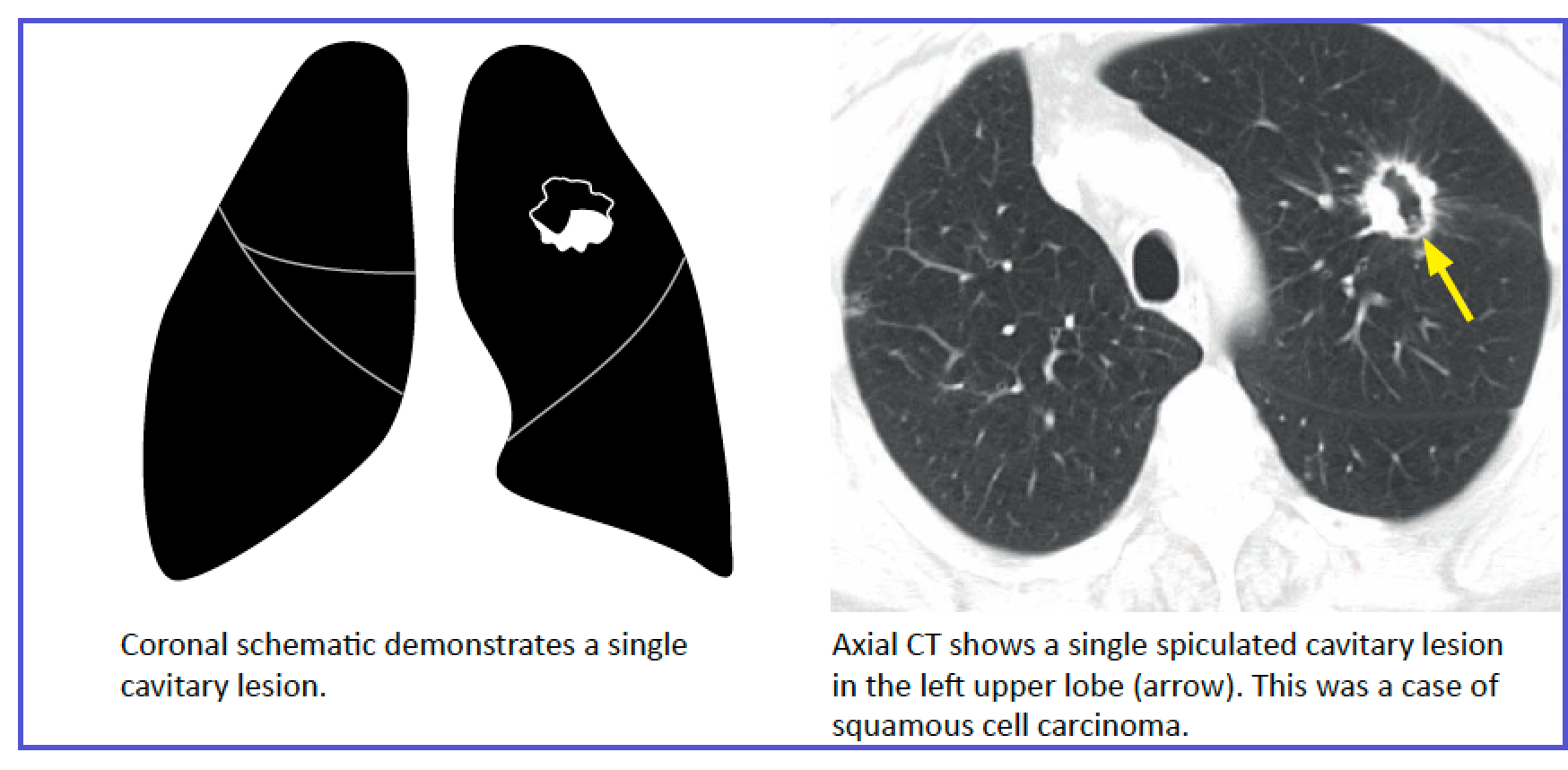
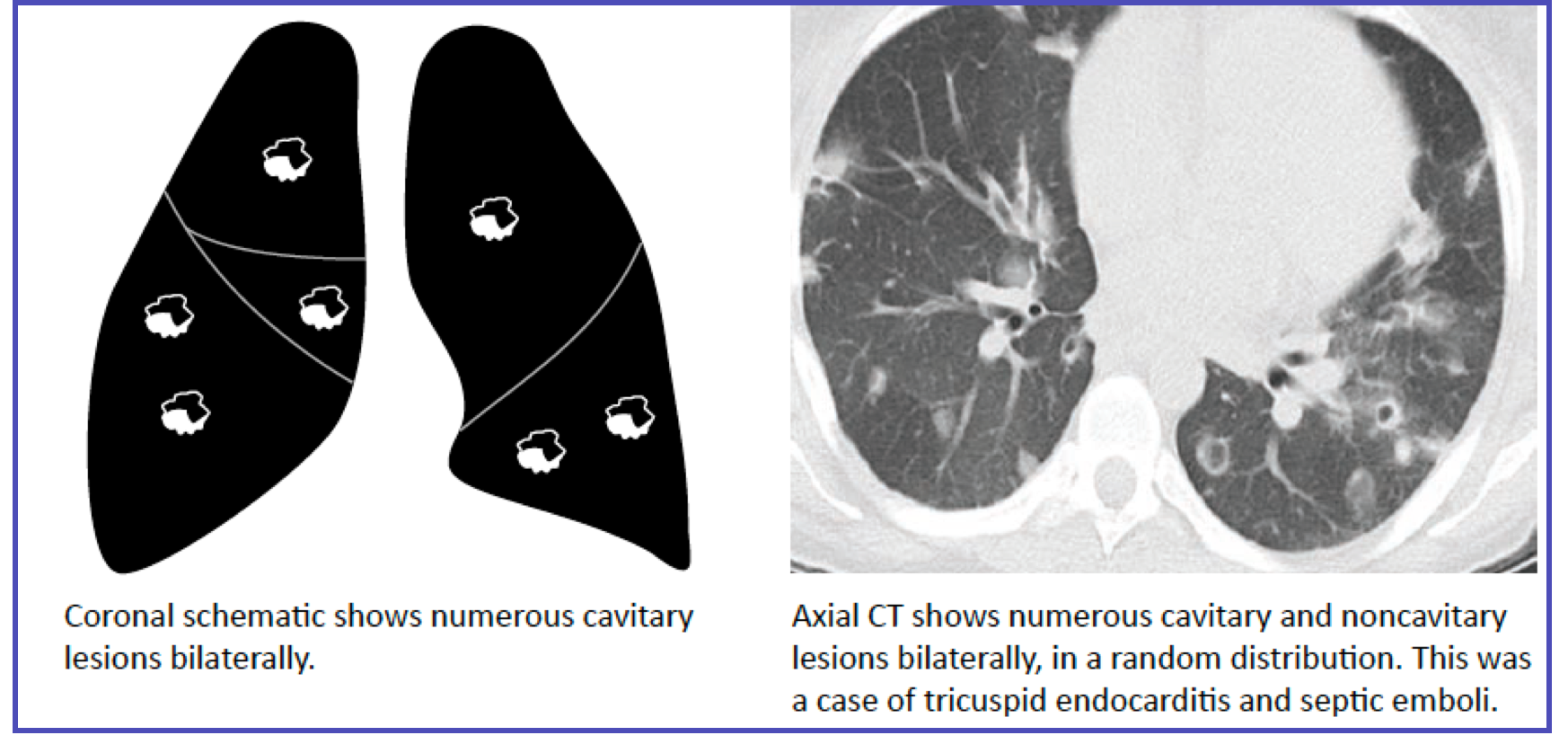
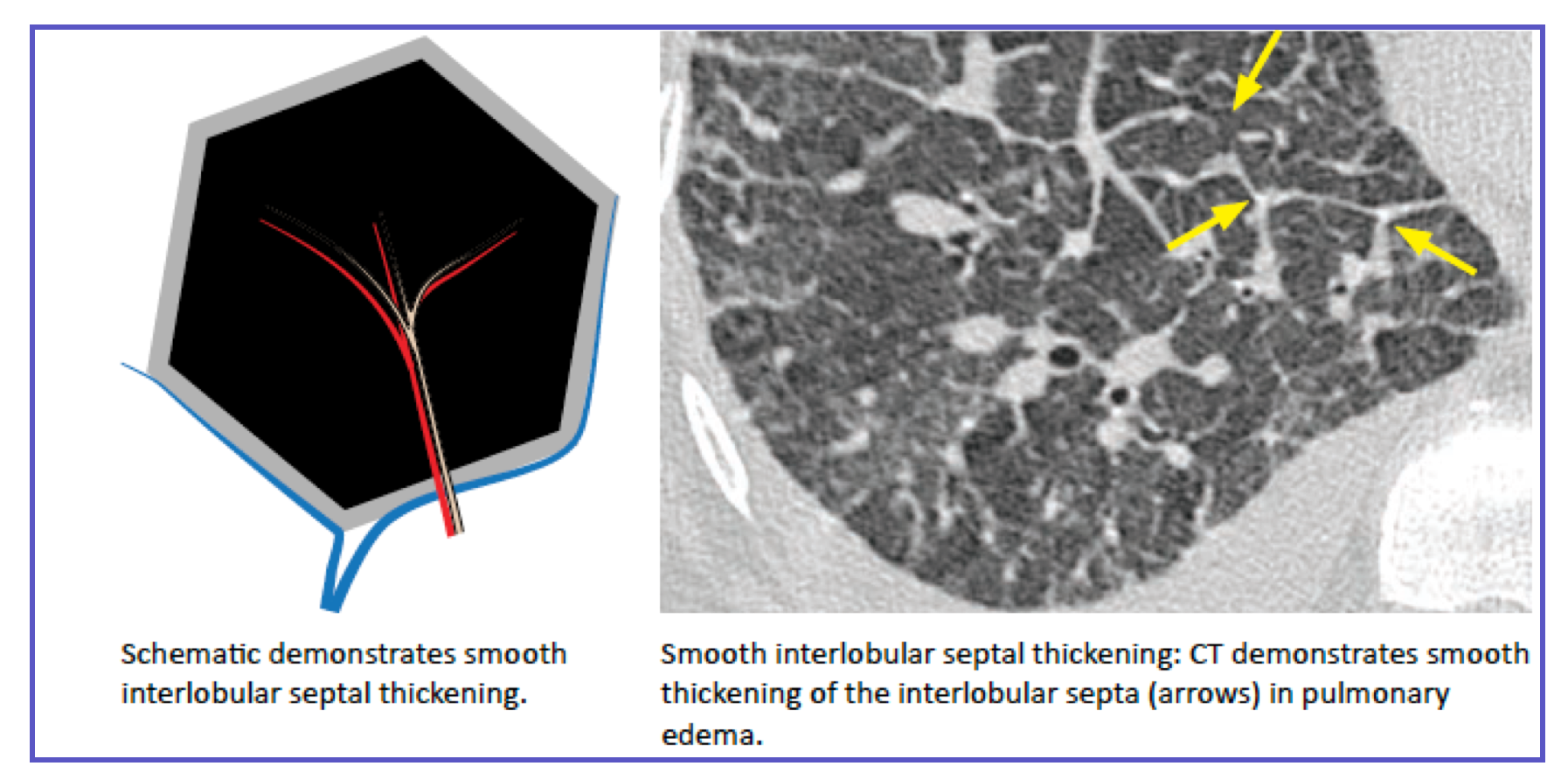
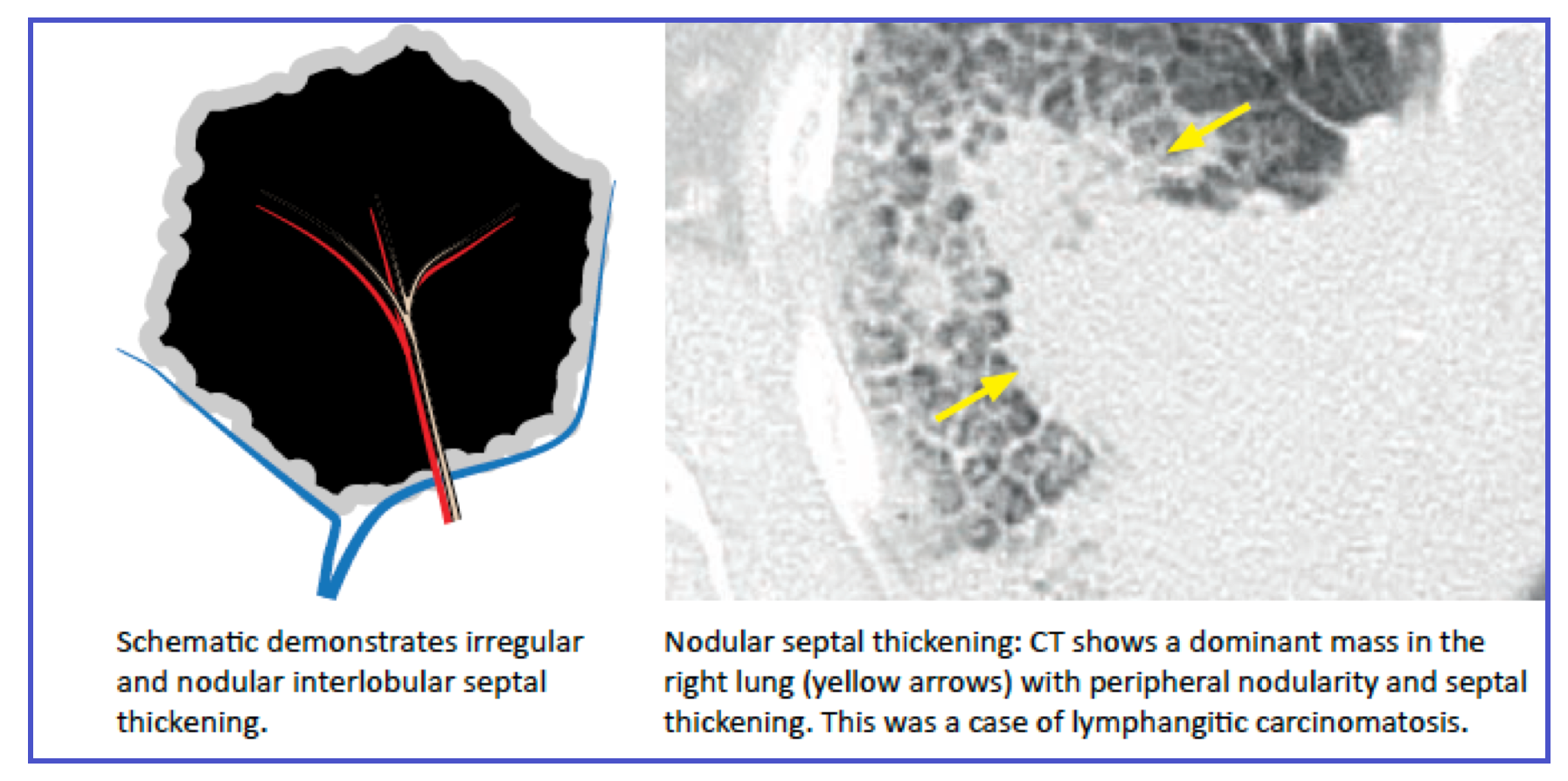
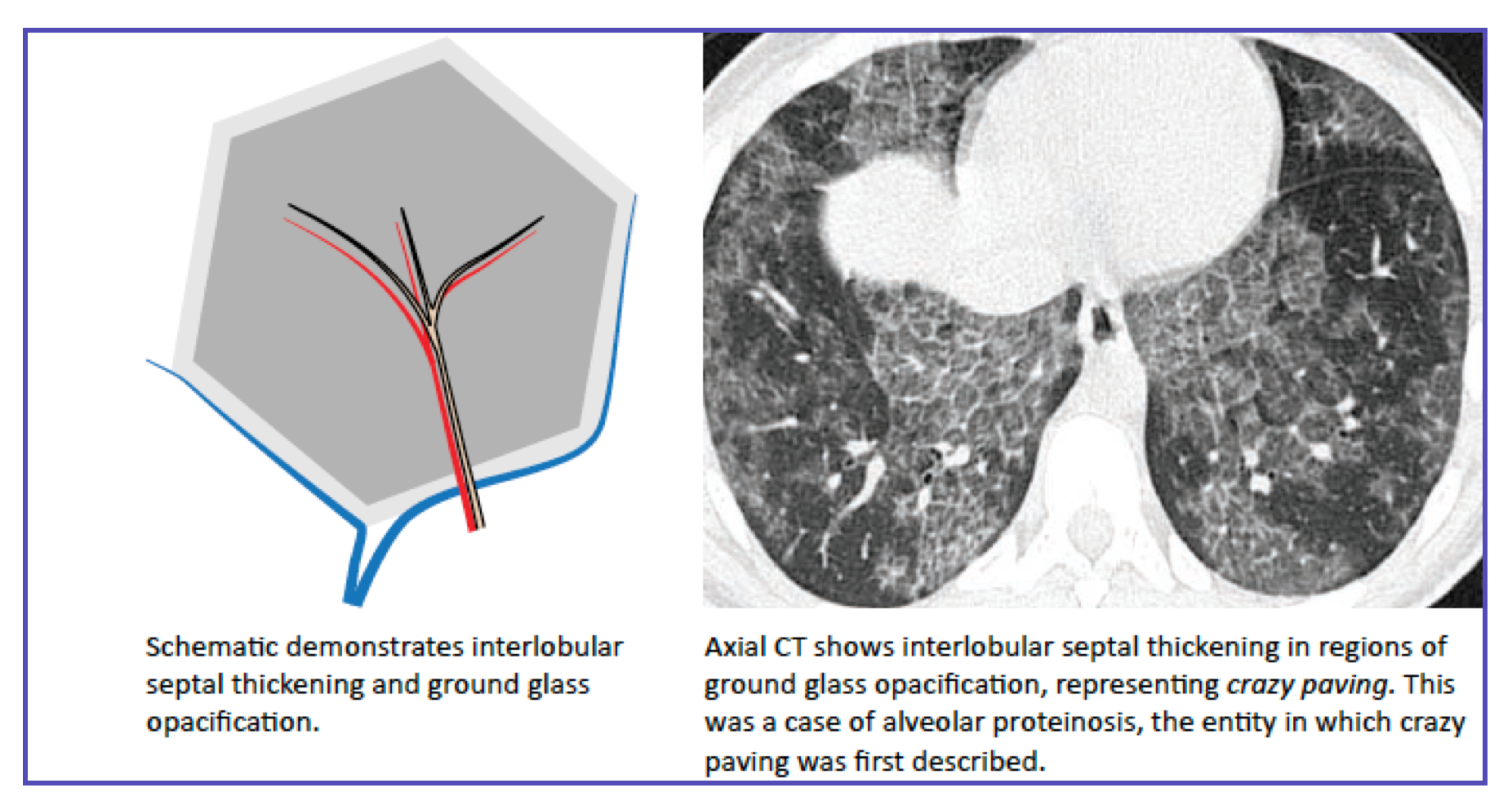


Add comment