9 Jan 2022 via Shahriar Lahouti. Last Update 30 August 2025.
CONTENTS
- Preface
- Anatomy of infarction
- Definition
- Diagnostic Evaluation
- Differential diagnosis of MI
- ECG findings in acute coronary occlusion
- ED Evaluation of Suspected ACS (Risk Stratification)
- Management of ACS
- Special circumstances
- Guideline links
- Further going
- References
Preface
The current practice and guidelines for ACS follow the STEMI vs. NSTEMI paradigm. However, this paradigm is suboptimal and should be updated to improve outcomes by identification of acute coronary occlusion (for which STEMI is a poor surrogate). Therefore, it is challenging to provide a comprehensive overview of ACS management that enables the reader to acquire the necessary skills to operate within the current standard of care while also recognizing the potential for improvement beyond that standard. This post serves as a guide for emergency physicians on myocardial infarction. It is assumed that cardiology will be consulted in patients with more severe myocardial infarction (e.g., occlusive MI and more severe nonocclusive MI). Likewise, it is assumed that cardiothoracic surgery will be consulted regarding patients who are potential candidates for CABG surgery.
Anatomy of infarction
◾️The right coronary artery (RCA) and the left main coronary artery (LMCA) extend from the aortic root 1. The left aortic sinus gives rise to the LMCA. The right aortic sinus gives rise to the RCA.
◾️Left main coronary artery (LMCA). It branches into:
- Circumflex artery (Cx)- Gives off obtuse marginal branches (OM). The Cx artery supplies blood to the left atrium, lateral and posterior to the left ventricle.
- Left anterior descending artery (LAD)- It gives off two branches: diagonal branches (D) and septal branches (S).
- LAD supplies the anterior and lateral of the left ventricle (via the diagonal branches) and the anterior of the septum (via septal branches). The apex of the heart is generally supplied by the “LAD”.
- The left anterior descending artery may extend even further in the case of a type 3 “wraparound left anterior descending artery,” which describes the left anterior descending artery wrapping around the apex and supplying the apex and inferior wall.
- LAD supplies the anterior and lateral of the left ventricle (via the diagonal branches) and the anterior of the septum (via septal branches). The apex of the heart is generally supplied by the “LAD”.
◾️Right Coronary Artery (RCA)
- The right coronary artery branches into:
- Sinoatrial nodal branch supplying the sinoatrial node.
- Right marginal artery
- Posterior descending artery (PDA)
- The right coronary artery supplies the right atrium, right ventricle, and the Inferior portion of both ventricles and the posterior part of the septum.
◾️PDA: is the artery that supplies the posterior interventricular septum and atrioventricular node.
- It is supplied by RCA in 70% of the general population (called right dominant).
- In 10% of the population, it is supplied by Cx (left dominant).
- In 20% of the population, it is supplied by both RCA and Cx (called co-dominant).
⚡️The coronary blockade can cause conduction block at the AV nodal, His, or bundle branch level (Figure right below).
- The sinus node is supplied by the RCA.
- The atrioventricular node is supplied by the RCA in right-dominant individuals.
- The right bundle branch is supplied via LAD.
- Anterior fascicule (AF) is supplied by LAD, and posterior fascicule (PF) has dual blood supply via both PDA and LAD.
🩸Coronary arterial supply to mitral papillary muscle (PM). Figure left below.
- Anterolateral PM: It receives dual blood supply (LAD and Cx) and, therefore, is relatively resistant to ischemia.
- Posteromedial PM: It receives a single blood supply (PDA) and is more prone to injury from ischemia.
Echocardiography
🔎The 16-segment model for the determination of regional LV function and the usual coronary artery distribution of segments is shown below.
ECG leads
◾️Each ECG lead records the underlying cardiac surface (Figure left below).
- The ECG changes in MI are helpful to localize the involved coronary artery (Figure right below). Keep in mind that there are certain overlaps between the coronary arteries’ territorial supply, such as:
- Anterolateral LV: supplied by both LAD and Cx
- Inferolateral LV: supplied by both Cx and RCA
Definitions
Myocardial Injury vs. Myocardial Infarction
◾️Should all patients with cTn elevation be labeled as myocardial infarctions?
- Not every patient with cTn elevation has a myocardial infarction.
◾️Definition of Myocardial Injury
- Detection of an elevated cardiac troponin value above the 99th percentile URL is defined as myocardial injury.
- The injury is considered acute if there is a rise and/or fall of cardiac troponin values.
◾️Clinical criteria for Myocardial Infarction (MI)
- The term acute myocardial infarction should be used when there is a dynamic rise and fall in troponin (with at least one value above the 99th percentile of normal) and at least one of the following *:
- Ischemic symptoms OR
- New ischemic ECG changes OR
- New regional wall motion abnormality (RWMA) in a pattern consistent with an ischemic etiology.
⎮📈 The New ischemic ECG changes here is referred to:
- ST-segment changes
- New ST elevation at the J-point in ≥2 contiguous leads {≥1 mm in limb leads, ≥2 mm in precordial leads (sex- and age-specific cutoffs may apply)}
- New horizontal or downsloping ST depression ≥0.5 mm
- Dynamic ST changes during symptoms are particularly diagnostic.
- T-wave abnormalities: New inversion ≥1 mm in ≥2 contiguous leads <OR> Hyperacute T-waves (early STEMI sign).
- Pathological Q waves: ≥0.03 sec and ≥1 mm deep in ≥2 contiguous leads.
- New conduction abnormalities, e.g., LBBB, RBBB (with ischemic symptoms)
Pathophysiologic Classification of Myocardial Infarction
◾️Does every patient who meets the diagnosis of AMI have coronary arterial disease (CAD) with superimposed plaque rupture/erosion?
◾️Pathophysiologic classification of MI:
- Type 1 MI = Acute atherothrombosis (plaque rupture/erosion → thrombus).
- Patients meet the AMI definition ( discussed above), AND
- Identification of a coronary thrombus by angiography, including intracoronary imaging, or by autopsy.
- Type 2 MI = Supply–demand mismatch without acute thrombosis.
- Patients meet the AMI definition, BUT without evidence of coronary thrombus (if angiography was done).
- Mechanism: T2MI 🔄 Supply-demand mismatch. Possible causes for the mismatch are:
- Systemic / non-cardiac causes
- Severe anemia (↓ oxygen carrying capacity)
- Severe hypoxemia (respiratory failure, pneumonia, ARDS, CO poisoning)
- Sepsis / critical illness (cytokine-driven ↑ demand + ↓ supply)
- Pulmonary embolism (↑ right ventricular strain, ↓ LV filling, hypoxemia)
- Stroke, SAH, stress states (catecholamine surge, ↑ demand)
- Renal failure (often chronic injury, but can exacerbate ischemia acutely)
- Cardiac causes
- Tachyarrhythmias (atrial fibrillation with rapid ventricular response, VT, SVT)
- Bradyarrhythmias (high-grade AV block, severe sinus bradycardia)
- Severe hypertension (↑ afterload, ↑ demand)
- Severe hypotension / shock (↓ coronary perfusion pressure)
- Coronary spasm (vasospasm)
- Coronary embolism (e.g., from AF, endocarditis, prosthetic valves)
- Spontaneous coronary artery dissection (SCAD)
- Endothelial dysfunction or microvascular dysfunction
- Systemic / non-cardiac causes
- Type 3 MI = Cardiac death before biomarkers are measured, but presumed ischemic mechanism.
- Types 4 & 5 = Procedural pathophysiology (PCI-related, CABG-related).
◾️Why Pathophysiologic classification matters?
- It links troponin elevation to mechanism (atherothrombosis vs. supply–demand mismatch vs. procedural injury).
- Guides management decisions:
- Type 1 MI → usually needs urgent reperfusion (PCI, lytics).
- Type 2 MI → treat underlying cause (e.g., tachyarrhythmia, anemia, sepsis) rather than routine PCI.
- Type 4/5 MI → procedural context, thresholds for troponin rise are different.
- Improves research, epidemiology, and coding (registries, trials, outcomes).
Type 1 vs. Type 2 MI
⏐💡Differentiating Type 1 vs. Type 2 MI is often challenging and subjective. In the following table some helpful clues are summarized (see figure below).
| Feature | Type 1 MI | Type 2 MI |
|---|---|---|
| Underlying Mechanism | Acute atherothrombosis from plaque rupture, erosion, fissure, or dissection → thrombus formation | Oxygen supply–demand mismatch without acute plaque rupture (e.g., anemia, sepsis, tachyarrhythmia, hypoxemia) |
| Amount of Physiologic Stress | Low stress required to provoke ischemia (culprit lesion drives event) | High stress state (anemia, hypoxemia, hypoperfusion, severe tachy/bradyarrhythmia, shock) |
| Clinical Context | Initial presentation consistent with acute coronary syndrome (sudden chest pain/angina) | Patients present with a primary non-cardiac problem (sepsis, hemorrhage, respiratory failure); ischemia is secondary |
| Symptoms | Chest pain or anginal equivalent usually present | Often absent; ischemia may be silent, overshadowed by systemic illness |
| ECG Findings | May show diagnostic STEMI/NSTEMI changes; strongly suggestive of occlusive event | Minimal or nonspecific ST–T changes; abnormalities resolve with correction of stressor |
| Troponin Elevation | Usually larger and more pronounced rise/fall pattern | Typically moderate rise; but may be large if severe fixed CAD (e.g., left main stenosis) is unmasked by stress |
| Echocardiography | New regional wall motion abnormality (RWMA) or reduced ejection fraction | May show hyperkinetic, underfilled LV; often no new RWMA |
| Angiography | Culprit lesion with thrombus usually seen | No acute thrombus; may show normal coronaries or stable CAD |
| Management | Urgent reperfusion (PCI/fibrinolysis) + antithrombotic therapy | Treat underlying cause (correct anemia, sepsis, arrhythmia, blood pressure); PCI only if culprit lesion identified |
◾️Evolving Frameworks of Myocardial Infarction Classification
- Troponin elevation is the starting point for identifying myocardial injury, but further classification depends on clinical context, ischemic evidence, and coronary anatomy. The Universal Definition of MI distinguishes Type 1 (plaque rupture/atherothrombosis) from Type 2 (supply–demand mismatch), while subsequent frameworks—STEMI/NSTEMI and, more recently, OMI/NOMI—further refine risk and treatment strategies. The following schematic illustrates this progression.
Classification of troponin elevation and types of myocardial infarction, highlighting the evolution from the Universal Definition (Type 1 vs. Type 2 MI) through the historical STEMI/NSTEMI framework to the OMI/NOMI paradigm.
STEMI vs. NSTEMI
◾️Background
- The STEMI/NSTEMI dichotomy emerged in the thrombolytic era, when angiography was not routinely available. Clinicians observed that patients with ST-segment elevation on ECG derived the greatest survival benefit from fibrinolytics and assumed this reflected acute coronary occlusion, while those without ST elevation were classified as NSTEMI and managed less urgently *.
⚠️ Caveats
- Despite its historical utility, the STEMI versus NSTEMI classification has important limitations.
- A substantial proportion of NSTEMI patients harbor acute coronary occlusions (ACO) that go unrecognized, resulting in harmful delays to reperfusion, while some patients with apparent STEMI have no true occlusion and undergo unnecessary invasive procedures.
- This reliance on ST-segment elevation as a surrogate marker for occlusion fails to capture the true pathophysiology of myocardial infarction.
- To compensate, risk scores such as GRACE were developed to triage high-risk NSTEMI patients, underscoring the inadequacy of the dichotomy itself.
- A substantial proportion of NSTEMI patients harbor acute coronary occlusions (ACO) that go unrecognized, resulting in harmful delays to reperfusion, while some patients with apparent STEMI have no true occlusion and undergo unnecessary invasive procedures.
- These pitfalls have prompted a shift toward a more accurate paradigm—occlusion myocardial infarction (OMI) versus non-occlusion myocardial infarction (NOMI)—which classifies patients by the presence or absence of acute coronary occlusion, the actual target of reperfusion therapy.
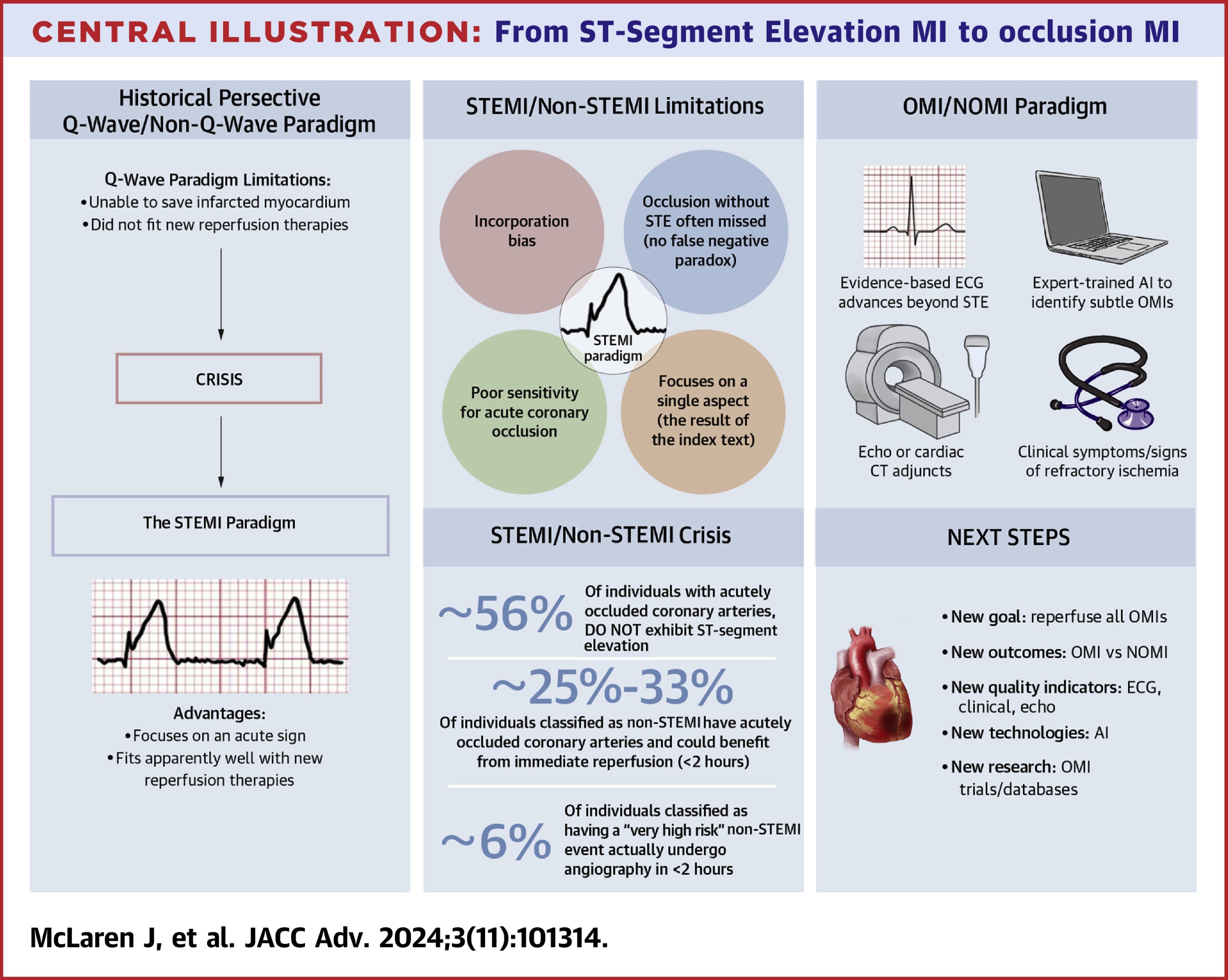
The OMI vs. NOMI paradigm
◾️Essentials
- The OMI versus NOMI paradigm reframes myocardial infarction classification around the presence or absence of acute coronary occlusion, rather than the ECG millimeter criteria of ST elevation.
- OMI is defined as an acute culprit lesion with TIMI 0–2 flow, or TIMI 3 flow accompanied by markedly elevated troponin, reflecting an artery that requires immediate reperfusion to salvage myocardium.
- NOMI, in contrast, represents infarction without acute occlusion, where invasive therapy can be safely deferred.
- This pathophysiology-based framework directly identifies the patients who truly benefit from emergent reperfusion, providing a more accurate and clinically meaningful approach than the traditional STEMI/NSTEMI dichotomy.
⎮Although the 2025 ACC/AHA guideline still classifies MI as STEMI or NSTEMI, the OMI/NOMI framework better reflects the underlying pathophysiology in emergency and critical care practice.
◾️Practical bedside algorithm
- The transition from the traditional STEMI/NSTEMI paradigm to the OMI/NOMI framework can be visualized in a practical bedside algorithm.
Figure: Proposed bedside algorithm integrating traditional STEMI criteria with the OMI/NOMI paradigm. Designed to complement the 2025 AHA/ACC ACS guideline, not to replace it.”
◾️Comparison of STEMI/NSTEMI vs. OMI/NOMI Paradigm
| Aspect | STEMI vs. NSTEMI (Old Paradigm) | OMI vs. NOMI (New Paradigm) |
|---|---|---|
| Historical Origin | Developed in the thrombolytic era (1980s–1990s), when angiography was not widely available. | Emerged from angiographic and ECG evidence (2000s onward) showing many NSTEMIs are true occlusions and many STEMIs are false positives. |
| Hypothesis | ST-elevation on ECG = acute coronary occlusion; no ST-elevation = no occlusion. | The presence or absence of acute coronary occlusion (or near-occlusion with poor collaterals) determines prognosis and therapy. |
| Definition | Based solely on ECG millimeter criteria for ST elevation (age- and sex-adjusted). | Based on angiography: culprit lesion with TIMI 0–2 flow, or TIMI 3 flow with markedly elevated troponin. |
| Theoretical Foundation | Surrogate marker (ST elevation) chosen because fibrinolytic trials showed benefit in this subgroup. | Pathophysiology-based: identifies the lesion that causes imminent myocardial necrosis unless reperfused. |
| Endpoint Used | Mortality reduction with fibrinolytics stratified by STE presence; angiographic occlusion not tested. | Actual coronary occlusion (OMI) as the endpoint; outcome = myocardial salvage and mortality reduction with reperfusion. |
| Surrogate Marker | ST-elevation (insensitive and nonspecific). | TIMI flow + troponin as gold standard; ECG, echo, and clinical findings as diagnostic tools. |
| Pitfalls | – Misses ~30–40% of true occlusions (STEMI– OMI). – False-positive STEMI activations. – Forced reliance on risk scores (e.g., GRACE, TIMI) to triage NSTEMI. |
– Requires integration of multiple tools at the bedside. – Still not universally adopted in guidelines. |
| Clinical Implication | Delayed reperfusion for many NSTEMI patients with OMI; unnecessary cath for some STEMI patients without occlusion. | More accurate triage: all OMIs → emergent reperfusion, NOMIs → safe for delayed or medical management. |
This table contrasts the historical STEMI/NSTEMI paradigm, built on ECG surrogates, with the OMI/NOMI framework, which focuses on the true pathophysiology of acute coronary occlusion and its management implications.
Traditional STEMI criteria rely on rigid millimeter-based ST-segment elevation thresholds, which frequently miss acute occlusions and generate false positives. The OMI paradigm broadens ECG interpretation by incorporating a spectrum of high-risk patterns and reciprocal changes that more accurately identify acute coronary occlusion at the bedside. The table below contrasts these two approaches, highlighting how OMI findings extend beyond the limitations of STEMI rules.
Electrocardiographic Features Distinguishing STEMI Criteria from OMI Patterns
| STEMI Criteria | OMI Findings |
|---|---|
| Must have ST-elevation in 2 contiguous leads | Broader patterns: hyperacute T waves, de Winter T waves, Wellens T waves, posterior STDmax V1–V4 |
| Fixed millimeter cut-offs (≥1–2 mm) | Pattern-based approach: South African flag sign, Aslanger pattern, reciprocal STD/T-wave inversion |
| Excludes STE in LBBB/paced rhythm | Smith-Modified Sgarbossa criteria identify OMI in LBBB or paced rhythms |
| Q waves considered old infarct | T/QRS ratio >0.36 in V1–V4 → acute LAD occlusion vs. LV aneurysm |
| Limited to ST elevation as surrogate | OMI integrates false positive/negative recognition (pericarditis, early repol, LV aneurysm) and multiple ischemic clues |
This table contrasts the rigid millimeter-based STEMI criteria with the broader OMI pattern-recognition approach, emphasizing how advanced ECG findings improve detection of acute coronary occlusion.
⎮📖 Limitations of the 2025 ACS Guideline in ECG-Based Diagnosis
- There is a growing recognition in the literature that the classification of MI should be based on the concept of occlusive myocardial infarction (OMI) versus non-occlusive myocardial infarction (NOMI), especially in light of emerging evidence on ECG patterns*.
- Despite these advancements, the 2025 AHA guideline partly incorporates OMI-aligned recognition (posterior STEMI evaluation, RV leads; de-emphasizing “new LBBB”) but does not adopt the OMI framework or its full ECG pattern profile. The guideline continues to use the traditional STEMI/NSTE-ACS approach, without including the full range of ECG features that could aid in identifying OMI *.
🧰 Clinical toolkit for bedside OMI identification
- ECG interpretation
- Skilled recognition of high-yield ischemic patterns beyond millimeter ST-elevation (e.g., hyperacute T waves).
- Serial ECGs
- Repeated tracings during symptoms or dynamic changes increase sensitivity for detecting evolving occlusion.
- Troponin kinetics
- Rising and falling troponin levels help distinguish OMI from non-occlusive causes of injury.
- Bedside echocardiography
- New regional wall-motion abnormalities suggest acute occlusion when ECG is inconclusive.
- Clinical context
- Refractory chest pain, hemodynamic or electrical instability, or high-risk ACS presentation should prompt urgent angiography, even without STEMI criteria.
Diagnostic Evaluation
Diagnosis of MI is complicated since no single risk factor, symptom, sign, or investigation finding in isolation has a high sensitivity or specificity for the diagnosis 3 4. Usual components of diagnosis are discussed here.
Clinical presentation
◾️Background
- Clinical presentation is highly variable.
- Clinical features associated with ACS vary based on the patient type, including sex, comorbid conditions, and age considerations.
- No single risk factor, symptom, or sign has high accuracy for the diagnosis of ACS *.
- Unfortunately, multiple systematic reviews have confirmed the poor accuracy of any single risk factor, symptom, or sign for the diagnosis of ACS
◾️Risk factors
- The most useful risk factors with positive likelihood ratios (LR+) in the acute setting are:
- PAD (LR+ 2.7)
- Abnormal previous stress test (LR+ 3.1)
- Established history of CAD (LR+ 2.0)
- The most useful symptoms in the acute setting include:
- Radiation to the shoulder, jaw, or arms (bilateral arms (LR + 2.6) > right >> left)
- Pain similar to prior ischemia (LR+ 2.2)
- Worse with exertion: LR + 2.5
- A change in the pattern of chest pain over the most recent 24 h (LR+ 2.0)
- Associated with diaphoresis: LR +2.
- Associated with vomiting: LR +1.9
- All of the following patient descriptions of chest discomfort are associated with the same incidence of AMI: Burning, pressure, squeezing, indigestion, crushing, tightness, numbness, and nondescript chest discomfort.
- Only “stabbing” is associated with a lower probability
- Conversely, helpful historical features that decrease the likelihood of ACS are pain that is
- Fully reproducible by palpation (LR+ 0.28)
- Fully pleuritic or fully positional
⚠️ Unusual presentations (e.g., MI without chest pain) are more likely in women, the elderly, and/or diabetic patients.
- Presentations can vary broadly (e.g., nausea/vomiting, dyspnea, syncope, fatigue, jaw pain, epigastric pain).
- Atypical presentations: especially in the elderly, women, and diabetics:
- Epigastric pain, indigestion
- Isolated dyspnea or fatigue
ECG
◾️Principles
- Do not merely rely on the presence or absence of ST elevation (STE) for the diagnosis of OMI.
- Diagnosis of AMI is, unfortunately, vastly more complex than simply counting the number of millimeters of ST elevation in various leads (thereby invalidating the traditional STEMI/NSTEMI dichotomy), as discussed above.
- This is evolving into the NOMI/OMI conceptualization, which emphasizes more holistic ECG interpretation to detect a variety of patterns suggestive of coronary occlusion.
- Diagnosis of AMI is, unfortunately, vastly more complex than simply counting the number of millimeters of ST elevation in various leads (thereby invalidating the traditional STEMI/NSTEMI dichotomy), as discussed above.
- Goal of ECG interpretation
- The goal of ECG interpretation is just to get a fairly reasonable concept of the diagnosis – to get pretty close.
- ⎮💡 The goal is not to secure a diagnosis with 100% certainty.
- ⎮⚠️ A normal ECG does not exclude ACS nor eliminate the need for further work-up if clinical suspicion of ACS remains.
- Definitive diagnosis will often require additional ECGs, echocardiography, and/or cardiac catheterization.
- The goal of ECG interpretation is just to get a fairly reasonable concept of the diagnosis – to get pretty close.
- How to interpret ECG in practice more efficiently?
- Context: Always interpret ECG in a clinical context. i.e., “Is ECG obtained as a routine screening or is it performed in a patient suspected of MI?”
- Comparison: Always compare STAT ECG to baseline ECG (if available) as well as to serial ECGs over time.
- Rule of Proportionality: Implies that the finding on ECG should be compared to the overall ECG (e.g., 0.5 mm STE may be significant if the QRS complex is tiny).
- Electrocardiographic occlusion myocardial infarction (OMI) patterns beyond classic STEMI changes are shown below.
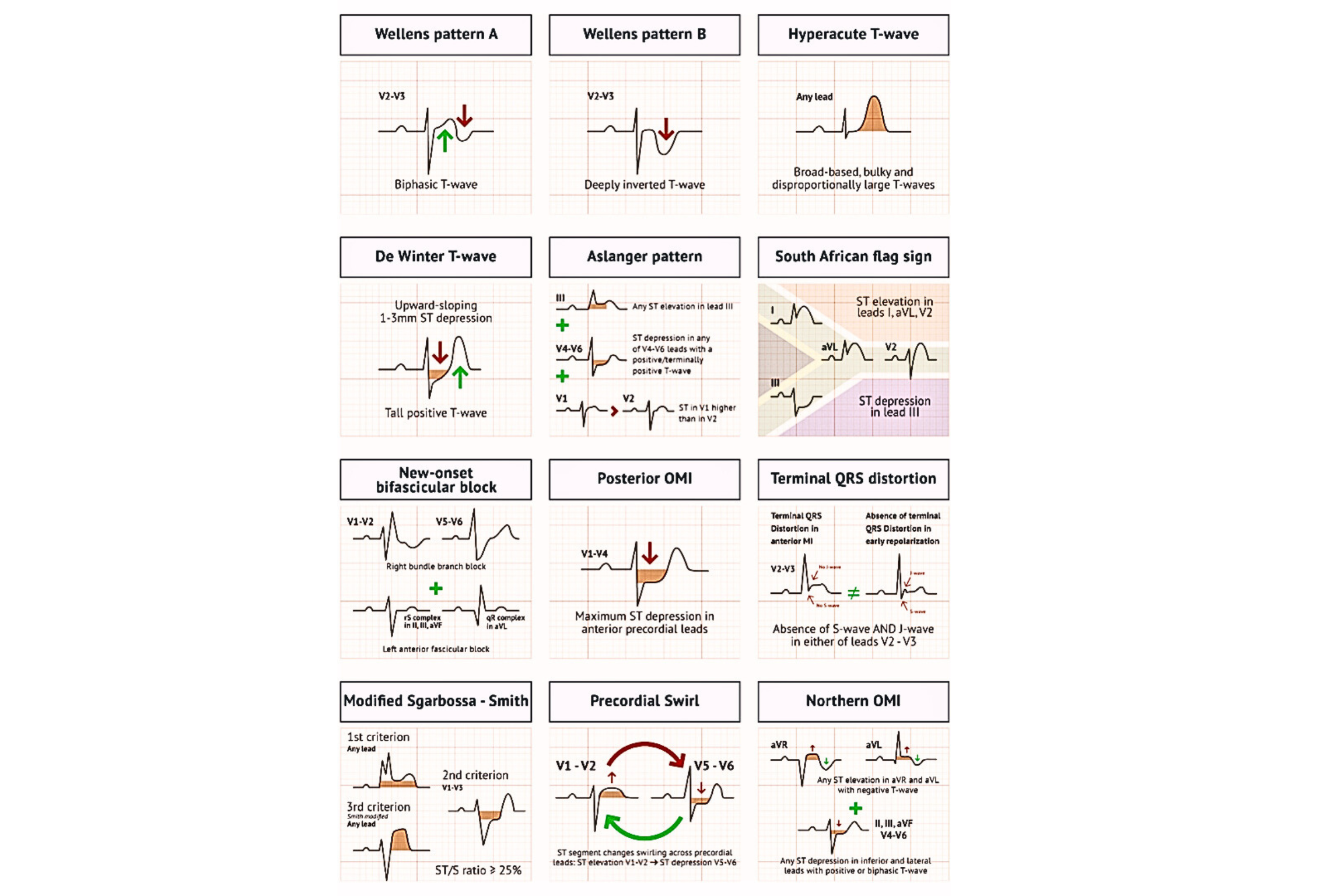
| OMI Patterns | ECG Features | Coronary Anatomy |
|---|---|---|
| Hyperacute T Waves |
• Broad-based T waves, disproportionately large compared with QRS • Bulky, convex, abnormally symmetric • T-wave amplitude/QRS ratio more clinically significant than T-wave amplitude alone |
Any artery, according to ischemia localization |
| De Winter pattern |
• ST-depression at J point in precordial leads, 1–3 mm, with tall symmetric T wave • 1–2 mm STE in aVR |
Proximal LAD occlusion |
| Aslanger pattern |
• STE isolated to lead III but not other inferior leads • ST-depression in V4–V6 (not in V2) with positive T wave • ST segment in lead V1 > V2 |
LCX or RCA occlusion in multivessel disease |
| South African flag sign |
• STE in leads I, aVL, V2 • ST-depression in lead III |
Occlusion of first diagonal branch of LAD |
| New-onset RBBB and LAFB |
• New RBBB • New LAFB • Concordant STE in anterior/lateral leads; concordant STD in inferior leads |
Proximal LAD occlusion |
| Posterior OMI | • STD max in V1–V4 not due to abnormal QRS (e.g., RBBB) | Occlusion of LCX, RCA, or branches |
| Terminal QRS distortion | • Absence of S wave and J wave in V2 or V3 | LAD occlusion |
| Modified Sgarbossa–Smith criteria |
• Concordant STE ≥1 mm in ≥1 lead • Concordant STD ≥1 mm in V1–V3 • STE/S-wave amplitude ≥0.25 (excessively discordant) |
Localization per ischemia site |
| Precordial Swirl |
• STE in aVR and V1–V2 • STD in V5–V6 |
LAD occlusion |
| Northern OMI |
• Any STE in aVR, aVL with T-wave inversion • STD in inferior/precordial leads with biphasic T waves |
LAD and first diagonal branch occlusion in multivessel disease |
| Reperfusion patterns (Wellens) |
• Terminal T-wave inversion progressing to full inversion (Wellens A→B) • Seen after reperfusion |
Any artery (proximal LAD in Wellens) |
ECG Evolution in Acute Coronary Occlusion
The progression of ECG findings seen during acute coronary occlusion and reperfusion is shown below (adapted from the OMI manifesto, Dr. Smith blog).
- Prototypical Evolution During Acute Occlusion
- The earliest ECG sign of occlusion is QT lengthening, followed by a hyperacute T wave, which signals a viable but ischemic myocardium.
- Then, as the ischemia progresses, the ST segment begins to elevate.
- As the amount of viable myocardium diminishes with infarction (irreversible cell death or myocardial stunning), the ST segment begins to fall.
-
As the myocardium irreversibly infarcts, Q waves form.
-
⎮💡Q waves may also form in salvageable myocardium. This is especially true in anterior MI, where Q waves may form early and are reversible with early reperfusion.
-
-
- This prototypical evolution depends on the myocardium at risk, the location of the myocardium relative to the leads, the collateral circulation, the completeness and timing of reperfusion, and the presence of no-reflow (when downstream perfusion is still blocked at the level of the microvasculature despite successful intervention on the epicardial vessel during PCI).
◾️ECG Evidence of Reperfusion
- The resolution of hyperacute T waves and ST segment elevation (>70%).
- The progression of terminal, then full, T-wave inversion over the course of hours to days.
Echocardiography
◾️The role of echo in the evaluation of patients with suspected MI includes:
- Diagnosis of MI: Regional wall motion abnormalities (RWMA) support the diagnosis.
- Sensitivity: While the presence of a “WMA” may help to increase the suspicion of MI, its absence should not necessarily be reassuring unless the echocardiogram is truly of perfect quality, performed with contrast, and read by a skilled echocardiographer.
- Specificity: The presence of “WMA” has a low specificity (since this may reflect a remote MI, myocarditis, or Takotsubo cardiomyopathy). Combining echo with ECG can improve specificity. To maximize efficiency, it’s often useful to start with ECG interpretation and then use echo to sort out different possibilities. For example:
- A patient presents with diffuse STE, concerning for either a very large occlusive MI or pericarditis. Echocardiography can readily differentiate these possibilities.
- A patient presents with right precordial T-wave inversion that is concerning for either Wellens pattern or a submassive PE. Echocardiography can rapidly evaluate the right ventricle to assess the likelihood of a submassive PE.
- It is also important to remember that the “WMA” may be transiently present, corresponding with the dynamic nature of coronary occlusion and reperfusion. After reperfusion, the “WMA” may remain for hours to days to weeks (myocardial stunning), or it may resolve rapidly if the ischemia intensity and duration are minimal.
- Complications of MI. For example, papillary muscle rupture, myocardial rupture.
- Hemodynamic status of the patient and risk stratification
- Alternative diagnosis?
- RV dilation should always prompt consideration of PE (although other possibilities include right ventricular MI or chronic pulmonary hypertension).
- Aortic dilation and/or aortic valve regurgitation suggest aortic dissection.
Troponin
◾️Background
- Cardiac troponins are regulatory proteins located in the contractile apparatus of cardiomyocytes. They consist of three subunits:
- Troponin C (TnC): binds calcium and initiates contraction
- Troponin I (cTnI): inhibits actomyosin ATPase
- Troponin T (cTnT): binds tropomyosin, anchoring the complex
- Importantly, cardiac-specific isoforms (cTnI and cTnT) are distinct from skeletal muscle forms, making them highly specific biomarkers of myocardial injury.
🔬Laboratory assays for cardiac troponin
- Over the past two decades, troponin assays have evolved significantly:
- Conventional assays detect troponin at relatively high levels, limiting sensitivity for early myocardial injury.
- High-sensitivity cardiac troponin (hs-cTn) assays can detect concentrations an order of magnitude lower, identifying myocardial injury earlier and in a greater proportion of patients. Of note, hs-cTn measures both cTnI and cTnT.
- According to the 2025 AHA ACS Guideline, hs-cTn assays are now the preferred biomarker for MI diagnosis, as they provide improved sensitivity while maintaining specificity when interpreted in context *.
| Feature | Conventional cTn | High-Sensitivity cTn |
|---|---|---|
| Analytical sensitivity | Detects moderate–high levels | Detects very low levels with precision |
| Early MI detection | Limited in first hours | Improved early rule-in/rule-out |
| Use in accelerated pathways | Less suited | Standard of care (0/1h, 0/2h) |
⚠️ Limitations of Troponin in Early Presenters
⎮Before applying the diagnostic threshold for myocardial injury, it is important to recognize that the sensitivity of hs-cTn depends heavily on the time from symptom onset. In early presenters, an initial negative value may be misleading.
- High-sensitivity cardiac troponin (hs-cTn) has transformed the diagnosis of myocardial infarction, but its performance depends strongly on the timing of presentation after symptom onset. Early presenters may still have negative troponin values despite ongoing occlusion.
- In a large cohort study of ~41,000 patients (31% presenting ≤3 h after onset), using the 99th percentile cutoff for hs-cTn had a sensitivity of only ~71% in early presenters, meaning nearly 30% of true MI cases were missed at initial measurement. In contrast, sensitivity improved to 92.5% in patients presenting >3 h after symptom onset *.
- Thresholds set at the limit of detection (LoD, e.g., ~2 ng/L) achieved >99% sensitivity and negative predictive value, but only about 40% of early presenters fell below this threshold — underscoring that most had detectable values but not yet diagnostic *.
⎮💡Clinical implication:
- A negative hs-cTn within the first 1–2 hours of chest pain does not exclude acute MI, especially occlusive MI.
- This justifies the use of serial hs-cTn testing (0/1h, 0/2h, or 0/3h algorithms) in all patients.
- ECG interpretation and bedside imaging remain critical in the early phase, as biomarkers may lag behind the pathology.
| Time from Symptom Onset | Sensitivity at 99th Percentile URL | % of MI Cases Missed | Notes |
|---|---|---|---|
| ≤ 3 hours | ~71% | ~29% | Early presenters often have sub-diagnostic hs-cTn at the first draw; serial testing (0/1–2 h) is essential. Using a very low “limit-of-detection” threshold yields >99% NPV, but only ~40% of early presenters fall below that LoD. |
| > 3 hours | ~92.5% | ~7.5% | Diagnostic performance improves as time elapses; still integrate ECG/clinical context. |
HS-troponin sensitivity at presentation depends on time from symptom onset. A negative early value does not exclude MI; use serial sampling and integrate ECG/clinical findings.
📍For this reason, serial hs-cTn measurements remain essential, and interpretation must always be integrated with ECG and clinical context before applying the universal threshold for myocardial injury.”
◾️Threshold for Myocardial Injury
- By international consensus, myocardial injury is defined as troponin >99th percentile upper reference limit (URL) for the assay used.
- Myocardial infarction requires an acute rise and/or fall of troponin with at least one value above the 99th percentile plus clinical evidence of ischemia (symptoms, ECG, imaging, or angiographic findings).
📈📉Dynamic Serial Evaluation
- A single troponin value is insufficient for diagnosis in most cases.
- Serial testing (0/1h, 0/2h, or 0/3h protocols depending on assay and setting) allows differentiation between acute MI and chronic myocardial injury.
- Rise and fall kinetics are key:
- Acute MI → significant delta (rise/fall)
- Chronic injury (e.g., renal failure, structural heart disease) → persistently elevated but stable values
- In ED populations, serial hs-cTn testing combined with ECG can rapidly rule out MI with very high negative predictive value.
- In ICU populations, frequent mild-moderate troponin elevations are common and often represent Type 2 MI or non-ischemic injury; thus, clinical context and supportive evidence are critical.
- Dynamic Change in Troponin (in Type 1 MI): Absolute vs Relative Delta
- Dynamic change in troponin levels can be expressed as either a relative (%) or absolute (ng/L) delta, and both approaches have advantages and limitations.
- Controversy remains around the optimal definition of a significant delta:
- Relative change (%)
- Traditionally, a ≥20% change has been used to define significant dynamics.
- However, relative change is less reliable when baseline troponin values are very low or very high.
- Absolute change (ng/L)
- Increasingly favored in the hs-cTn era.
- Even small absolute increases (e.g., 5–10 ng/L depending on the assay) can be clinically meaningful, especially near the decision threshold.
- Provides more consistent diagnostic accuracy across the spectrum of values.
- Relative change (%)
⎮📖 Guideline perspective:
- The 2025 AHA ACS guideline does not endorse a universal delta cutoff but emphasizes using assay-specific validated thresholds (absolute or relative).
- Serial measurement protocols (0/1h, 0/2h, or 0/3h depending on assay and population) are recommended to capture dynamic change.
⎮🔎 The table below summarizes these methods and emphasizes the importance of integrating troponin dynamics with clinical evidence of ischemia.
Interpreting Dynamic Troponin Change (Δ)
| Approach | Definition | Strengths & Limitations |
|---|---|---|
| Relative Change (%) | ≥20% rise or fall from baseline | Simple, familiar; less reliable at very low or very high troponin values |
| Absolute Change (ng/L) | Assay-specific cutoffs (e.g., 5–10 ng/L) | More consistent in hs-cTn era; small changes clinically meaningful near threshold |
| Clinical Integration | Δ troponin + ischemic evidence | Essential for distinguishing acute MI from chronic injury |
No universal delta cutoff exists; assay-specific thresholds and integration with clinical context, ECG, and imaging are required.
❤️🔥 Clinical Value of Troponin in ACS
- Diagnosis of MI: Cornerstone biomarker, used with ECG and clinical findings (see above).
- Risk stratification: Higher troponin levels correlate with larger infarct size, higher risk of heart failure, arrhythmias, and mortality.
- Decision-making: Informs urgency of invasive strategy (e.g., early angiography in high-risk NSTEMI).
💬 Pitfalls and Considerations
- Troponin elevation is a marker of myocardial injury, not synonymous with acute coronary syndrome.
- Elevated cTn, only when paired with ischemic symptoms, ECG changes, or imaging evidence can it be called myocardial infarction *.
- Numerous cardiac and non-cardiac conditions can cause myocardial injury and raise troponin levels, often without acute coronary occlusion. These are summarized in the following table.
- ICU setting: Elevated troponin is more prognostic than diagnostic — routine testing is not recommended without suspicion of ischemia.
- Marathon/athletes: Transient, benign troponin rise post-exercise highlights that elevation alone is not synonymous with infarction.
Non-ACS Causes of Troponin Elevation
| Condition | Mechanism | Clinical Notes |
|---|---|---|
| Sepsis / Septic Shock | Inflammatory myocardial injury, demand–supply mismatch | Common in ICU; prognostic marker of mortality |
| Hypotension / Shock (any cause) | Global hypoperfusion, ischemic injury | Seen in hemorrhage, hypovolemia, cardiogenic shock |
| Hypoxemia / Respiratory Failure | Oxygen supply–demand mismatch | ARDS, COPD exacerbation, severe pneumonia |
| Severe Anemia | Reduced oxygen carrying capacity | Can mimic ACS in presentation |
| Myocarditis / Pericarditis | Direct myocardial inflammation | Often associated with viral infection or autoimmune disease |
| Tachyarrhythmias / Bradyarrhythmias | Oxygen demand imbalance or poor perfusion | AF with RVR, SVT, VT, high-grade AV block |
| Acute Heart Failure | Wall stress, subendocardial ischemia | Troponin correlates with worse outcomes |
| Stress (Takotsubo) Cardiomyopathy | Transient LV dysfunction, catecholamine surge | Mimics ACS with modest troponin rise |
| Pulmonary Embolism | RV strain and ischemia | Troponin elevation predicts higher mortality risk |
| Renal Failure (CKD/ESRD/AKI) | Reduced clearance + chronic myocardial strain | Interpretation requires serial trends, not single value |
| Stroke / Intracranial Hemorrhage | Neurogenic stunned myocardium | Associated with poor neurologic prognosis |
| Aortic Dissection | Coronary malperfusion, secondary ischemia | Must not be mistaken for primary ACS |
| Catecholamine Excess | Direct myocardial toxicity | Pheochromocytoma, cocaine, methamphetamine |
| Cardiac Procedures / Trauma | Direct injury to myocardium | PCI, CABG, ablation, CPR, chest trauma |
| Endurance Exercise | Transient cytosolic troponin release | ~50% of marathon runners have elevated troponin; benign, resolves in 24h |
Troponin elevation reflects myocardial injury, not always acute coronary syndrome. Only when combined with ischemic evidence (symptoms, ECG, imaging) does it meet criteria for myocardial infarction.
◾️When to check troponin?
- Don’t check troponin for everyone!
- The clinical indications for troponin testing differ substantially between the emergency department and the intensive care unit.
- In the ED, troponin is primarily a diagnostic tool for suspected ACS and risk stratification.
- It is ordered when there is suspicion for ACS or ischemic injury, not for all-comers with chest discomfort. The main role is rule-in/rule-out MI and risk stratification in suspected ACS.
- In the ICU, troponin testing should be targeted, not routine.
- Every ICU patients need an ECG, not troponin: Troponin should only be obtained if there is genuine concern for myocardial ischemia based on history and ECG.
- Physician judgement is built into the EKG test itself. So you can order tons of EKGs without causing harm, whereas if you start shotgun ordering troponin this leads to iatrogenic chaos *.
- Studies show very high prevalence of troponin elevation in sepsis, renal failure, ARDS, and other critical illness — most of which is myocardial injury, not Type 1 MI *.
- 📍Takeaway: In the ICU troponin is ordered selectively, serving more often as a prognostic marker unless ischemic evidence is present.
- In the ED, troponin is primarily a diagnostic tool for suspected ACS and risk stratification.
📍The table below highlights these contrasting approaches.
When to Order Troponin: ED vs ICU
| Emergency Department (ED) | Intensive Care Unit (ICU) |
|---|---|
| Chest pain or ischemic equivalents (dyspnea, syncope, diaphoresis) | Unexplained hemodynamic instability or shock |
| Suspicious ECG findings (STE, STD, T-wave inversion, dynamic changes) | New arrhythmia (AF with RVR, VT, bradyarrhythmia with poor perfusion) |
| Intermediate/high pre-test probability for ACS | Unexplained acute pulmonary edema or new LV dysfunction |
| Use in accelerated diagnostic algorithms (0/1h or 0/2h hs-cTn) | Concern for myocardial ischemia in sepsis, PE, or severe hypoxemia |
| Risk stratification in suspected ACS | Targeted testing only; not recommended as routine screening |
Troponin testing in the ED is primarily diagnostic and risk-stratifying for suspected ACS, whereas in the ICU it is selective and usually prognostic, requiring ischemic evidence for invasive action.
CK-MB
- CK-MB is an isoenzyme of creatine kinase found primarily in cardiac muscle, with smaller amounts in skeletal muscle.
- Current Utility
- CK-MB is no longer recommended as the primary biomarker for MI.
- Possible uses: Reinfarction.
- CK-MB rises and falls more rapidly than troponin. In theory, a “second peak” can suggest reinfarction if troponin remains elevated from the index event.
Differential Diagnosis
◾️Aortic dissection (AD)
- Symptoms: Chest pain (Sudden onset with rapid acceleration to maximal intensity, sharp/tearing/ripping, radiation to the shoulder, may be migratory). May be associated with a variety of symptoms (e.g., abdominal pain, neurologic, syncope).
- ECG: Usually nonspecific. Aortic dissection may rarely cause occlusion of a coronary artery, producing an OMI-pattern ECG.
- AD is notorious for causing right-sided STEMI; However, it’s not true, as it has been shown that left-sided events can happen 5.
- POCUS: Aortic root dilation, aortic regurgitation, pericardial effusion. Ultrasound of the abdominal aorta, carotids, or femoral arteries may show a dissection flap.
- Other tests: CT angiography of the aorta is diagnostic.
-
More on this here
◾️PE causing pleural infarct with chest pain
- Symptoms: Sharp, pleuritic chest pain, hemoptysis
- ECG: Nonspecific
- POCUS: May show pleural effusion, small area of lung consolidation, and possibly DVT.
- Other test: D-dimer elevation. CT angiography is diagnostic.
◾️PE causing RV strain with myocardial ischemia
- Symptoms: May cause ischemic-quality chest pain due to hypoperfusion of the right ventricular free wall. It’s often associated with syncope or dyspnea.
- ECG may show:
- Tachycardia
- T-wave inversion involving the inferior leads and/or right precordial leads (V1-V4).
- 👉The presence of TWI in both leads III and V1 allows PE to be differentiated simply but accurately from ACS in patients with negative T waves in the precordial leads.
- Right axis deviation.
- Complete or partial right bundle branch block (RBBB).
- SI-QIII-TIII pattern.
- ST elevation in aVR and ST depression in lead I
- RV injury pattern with STE in V1-V3 and/or STD in V4-V6. More on this here.
- POCUS: RV dilation and RV systolic dysfunction. The DVT study may be positive.
- Other tests: D-dimer and troponin is positive. CT angiography should be diagnostic.
- (sub)Massive PE can mimic acute MI.
- STEMI should cause RWMA involving the left ventricle and dysfunction of the left ventricle.
- Massive PE will cause RV dilation and usually an underfilled left ventricle (which is vigorously contracting).
◾️Pericarditis
- Symptoms: Chest pain is often pleuritic, positional (worse lying flat), and may be sharp. May be associated with febrile illness.
- ECG: May show diffuse concave STE and PR depression throughout most of the limb leads (I, II, III, aVL, aVF) and precordial leads (V2-6). STD and PR elevation in lead aVR (± V1).
- POCUS: pericardial effusion may be present.
- Pericarditis is a diagnosis of exclusion. See more here.
◾️Pneumothorax
- Symptoms: Dyspnea, sharp pleuritic chest pain
- ECG: Nonspecific
- POCUS: “Presence” of lung sliding and/or B-lines virtually excludes pneumothorax in that lung field (however, absence of lung sliding and/or B-line is not specific for pneumothorax).
- Visualizing the junction between sliding lung and absent sliding is known as the “lung point sign” and is nearly 100% specific for pneumothorax
◾️Takotsubo cardiomyopathy (aka apical ballooning syndrome, or broken heart syndrome)
- Symptoms: Ischemic-quality chest pain is often preceded by emotional or physical stress. Dyspnea, arrhythmia, and cardiogenic shock can be seen.
- ECG: STE is generally the first finding (usually greatest V2-V5). TWI may follow.
- POCUS: Typically shows apical hypokinesis. However, some patients may display a pattern of circumferential mid- or basal hypokinesis 21 (beyond a single epicardial vascular distribution).
- Other test: Elevated troponin. Cardiac catheterization is needed to exclude MI.
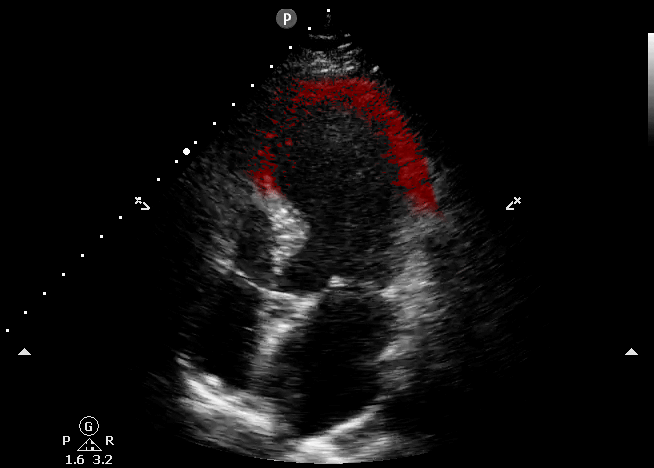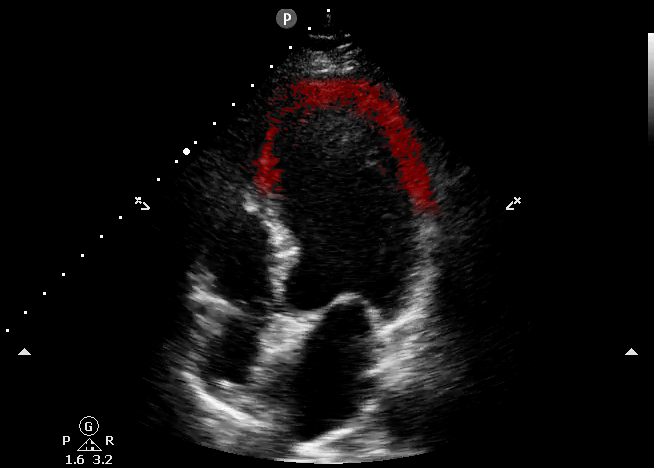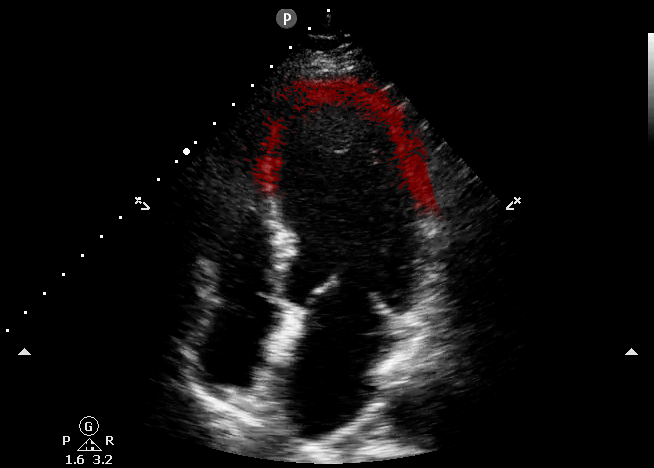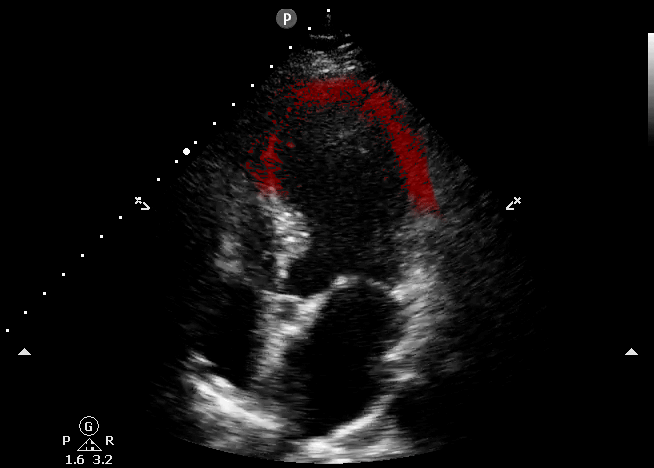
This echocardiography represents the classic appearance for Takotsubo Cardiomyopathy; akinetic apical septum and LV apex in the vascular territory of the LAD. This can be described as “ballooning” of the LV apex. The basilar septal segment is spared from CORE ULTRASOUND.
ECG Findings in Acute Coronary Occlusion
Hyperacute T-Wave
Hyperacute TW suggests ischemia, which is either transmural or nearly transmural. Isolated hyperacute TW (without ST deviation) is often a harbinger of impending ST elevation. However, isolated hyperacute TW may also be seen following spontaneous thrombolysis that has recently led to the resolution of ST elevation.
◾️Characteristics of hyperacute TW
- Relatively symmetric, broad-based, with a relatively blunt peak. Straightened upslope (they should not be deeply concave). Tall in comparison to the RW.
- Involving multiple leads in an anatomic pattern:
- Hyperacute TW typically involves several contiguous leads.
- If only one lead looks hyperacute, this may be an anomaly.
- If all of the ECG leads look similar, this argues against hyperacute TW (suggesting hyperkalemia or early repolarization).
- May be associated with other ischemia features, for example:
- STE or STD (de Winter pattern).
- Loss of RW progression.
- Changed compared to baseline ECG.
◾️Differential diagnosis of hyperacute TW
- Prominent T-waves as a reciprocal change to TWI
- Reperfused posterior MI may cause prominent right precordial TW.
- Reperfused high-lateral MI may cause a large inferior TW.
- Reperfused inferior MI may cause a large TW in aVL.
- Early repolarization
- TWs are tall and asymmetric.
- RW is also tall.
- Hyperkalemia
- TWs are narrow-based, pointy, symmetric, and tall.
- TWs are most marked in V2-V3, but the overall pattern is one of diffuse TW abnormality.
- May see other features of hyperkalemia (e.g., diminished P-waves, QRS widening). More on this here
ST Elevation
◾️Background
- OMI may produce ST elevation (STE) with either concave, convex, or obliquely straight morphology.
- The current formal “STEMI ECG criteria” include a new ST elevation measured at the J-point relative to the PQ junction, in two contiguous leads, with the following cutoffs:
- V2-V3: ≥2 mm in men ≥40 y old; ≥2.5 mm in men <40 y old, or ≥1.5 mm in women regardless of age.
- Other leads: ≥1 mm.
◾️DDX: STE may be seen in other conditions
- LBBB: ST segments and TW show “appropriate discordance” (i.e., they are directed opposite to the main vector of the QRS complex). This produces STE and upright TW in leads with a negative QRS complex.
- LVH: Causes a similar pattern of repolarization abnormalities as LBBB
- Ventricular paced rhythm
- LV aneurysm
- Pericarditis: widespread concave (“saddleback”) ST segment elevation with PR segment depression in multiple leads, typically involving I, II, III, aVF, aVL, and V2-6.
- Early repolarization: The ER pattern is a normal variant commonly seen in young, healthy patients. Most literature defines ER pattern as being present on the ECG when there is J-point elevation of ≥1 mm in ≥2 contiguous inferior and/or lateral leads 6. ECG features include:
- Widespread concave STE, most prominent in the mid-to-left precordial leads (V2-5)
- Remember that the STE of ER is not constant. In particular, it may be diminished with exercise, sympathetic tone, and heart rate, and may be increased when the heart rate is slower.
- There is often notching of the J-point; the “fish-hook” pattern.
- Prominent, slightly asymmetrical T waves that are concordant with the QRS complex
- Widespread concave STE, most prominent in the mid-to-left precordial leads (V2-5)
Early repolarization (ER) vs. Pericarditis
- Pericarditis can be difficult to differentiate from “ER”, as both conditions are associated with concave ST elevation.
- One useful clue to distinguish between these two entities is to look at the ST segment / T wave ratio and the Fish Hook Pattern.
- In ER, the TW amplitude is more prominent than in pericarditis. Calculate the height of ST segment elevation and compare to the TW height in lead V6. STE to TW height ratio in V6 < 0.25 (see below)
STE: South African flag pattern
◾️High lateral STEMI is associated with a pattern of ST elevation caused by acute occlusion of the first diagonal branch of the left anterior descending coronary artery (LAD-D1) *.
- ECG Features
- STE in noncontiguous leads; V2 and aVL with inferior reciprocal ST depression
- Coronary Anatomy
- Occlusion of the first diagonal branch of LAD
- Clinical Implication
- Requires prompt recognition and intervention
STE: Aslanger pattern
Aslanger et al identified a specific ECG pattern concerning acute inferior OMI in patients with concomitant multi-vessel disease that does not display contiguous ST-segment elevation or fulfill STEMI criteria.
⎮⚠️ 13.3% of inferior MIs may present with Aslanger Pattern, and due to being incorrectly labelled as an NSTEMI, may be deprived of emergent revascularisation therapy *.
◾️ECG Features (criteria)
- STE in lead III (but not any other inferior leads), as well as STE in V1, aVR
- ST-segment in V1 > V2
- Concomitant ST depression in any of V4-V6, with a positive/terminally positive T-wave (but no STD in V2).
◾️Coronary Anatomy
- Aslanger’s pattern is due to a combination of OMI involving the LCX or RCA plus simultaneous diffuse subendocardial ischemia (e.g., due to triple vessel disease) *. Diffuse subendocardial ischemia causes ST depression in leads II and aVF that camouflages the inferior OMI.
◾️Clinical Implication
- Suggests high-risk inferior-territory occlusion, warranting urgent intervention.
- Concurrent multi-vessel disease predisposes these patients to poor outcomes if there is a delayed time to emergent reperfusion, and prompt recognition of this potential OMI should improve outcomes
- Identification of the culprit lesion at the time of angiography may be difficult if there are multiple critical stenoses, and this pattern would guide lesions supplying the inferior wall to be opened first.
Shark Fins MI Pattern
◾️Background
- Shark Fin MI patterns refer to ECGs with broad QRST complexes that resemble fins.
- These very broad QRS complexes on the ECG are caused by:
- Massive STE merging with hyperacute TWs. This makes QRS complexes look superficially very broad.
- Concomitant bundle branch block.
- Often, there’s a bundle branch block, which widens the QRS complexes.
- These complexes manifest in contiguous ECG leads corresponding to the coronary anatomy and represent transmural ischemia (a sign of acute coronary occlusion).
⎮⚠️Pitfall: Be cautious, ECG can be mistaken for hyperkalemia, metabolic/toxicologic, bundle branch block, and wide complex tachycardia *.
⎮💡Diagnostic clue
- Identifying the unique morphology of QRS complexes (shark fins).
- The key to seeing and understanding how these complexes represent profound ST-Deviation lies in delineating the end of the QRS.
- The problem is that with this unique morphology, the QRS complex and T-wave merge as a result of extreme ST-Deviation, and the two become indistinguishable.
- But remember this: If you can find the end of the QRS in one lead, you can find the end of the QRS in any lead.
- Examine all 12 leads and identify one that clearly indicates the end of the QRS. In the following figure B, lead V5 happens to show the beginning and end of the QRS very nicely. Now all you have to do is simply draw a line straight up from this point (J-point) in V5, and you can find the same point in any lead.
- If the end of the QRS complex can be identified on one lead, it can be used to time-stamp the end of the QRS and find other leads using vertical lines
◾️Clinical significance: The presence of this sign appears to be associated with a significant mortality (due to cardiogenic shock and ventricular fibrillation), underscoring the critical importance of prompt recognition and emergency reperfusion.
Anterior OMI
- STE
- Most often seen in V2-V4
- Coved STE favors ischemia if seen (but anterior OMI can also cause concave STE).
- TW changes
- Hyperacute T-waves may be seen.
- Terminal QRS distortion in V2-V3 may occur (results from broad, malignant T-waves that invade the QRS complex and obliterate any S- or J-waves). See the right figure below.
- Loss of RW progression
- QW changes: may emerge rapidly (these are NOT a contraindication to revascularization).
- Reciprocal changes: STD anywhere other than in aVR or V1 favors OMI.
⚠️ Terminal QRS distortion: Defined as the emergence of the J-point > 50% of the RWs in leads with qR configuration or the disappearance of the S-wave in leads with Rs configuration.
- Terminal QRS distortion is identified as the absence of both the S-wave and J-wave in either lead V2-V3. (Figure below).
- Terminal QRS distortion is very specific for “LAD occlusion”. When differential diagnosis is acute LAD occlusion vs. early repolarization, terminal QRS distortion is 100% specific for LAD occlusion.
Specific Considerations & DDx
◾️Anterior OMI vs. Early Repolarization
- In the presence of anterior STE of at least >1 mm, the following approach may help differentiate anterior OMI versus early repolarization. Presence of “ANY” of the following features suggests anterior OMI:
- STE >5 mm.
- Any convex STE in V2-V6.
- Inferior reciprocal changes.
- Anterior ST depression.
- Any Q-wave in V2-V4.
- Any TWI in V2-V6, and/or aVL.
- Terminal QRS distortion in V2 or V3 (distortion is defined as a lack of either an S-wave or a J-wave in that lead. See above).
- If none of the above features are present, Smith’s formula can be used to differentiate OMI versus early repolarization.
- 👉 Caution: This formula is designed only to sort out OMI vs. early repolarization. It will not differentiate OMI from other MI mimics (e.g., LVH or pericarditis).
- 👉Avoid diagnosis of “ER” in patients older than 50 y/o or patients with cardiovascular risk factors.
◾️Anterior OMI vs. LV aneurysm
The features below may help sort out acute anterior OMI versus chronic aneurysm. For patients presenting late in the course of MI (e.g.,>6 hours after chest pain onset), this distinction will become blurred.
- Typical findings in LV aneurysm
- Deep Q-waves in V1-V4 (usually with at least one QS wave in these leads).
- Coved STE in V1-V4.
- Often causes some TWI, which is typically shallow.
- Features that suggest acute anterior MI (rather than solely an aneurysm)
- Any tall upright T-wave in V1-V4, with T-wave >36% the size of the QRS complex 8.
- Choose one lead with positive TW in V1-4. Calculate the height of the TW and compare it to the QRS height. A TW: QRS ratio of >0.36 is in favor of new anterior OMI.
- Other ischemic findings on the ECG, for example:
- STE in leads without deep Q-waves (especially aVL).
- Reciprocal STD.
- Any tall upright T-wave in V1-V4, with T-wave >36% the size of the QRS complex 8.
High Lateral OMI
◾️ECG findings of lateral OMI
- STE in aVL +/- Lead I.
- STE is often <1 mm (especially with low QRS voltages).
- Inferior reciprocal changes.
- STD in lead III (+/- STD in II and aVF) – may be the most obvious abnormality.
- TWI may occur as a reciprocal of hyperacute T-waves in lateral leads (more worrisome if this occurs in leads with a positive QRS complex).
◾️Culprit vessels
- Occlusion of the first diagonal branch (D1) of the LAD may produce isolated STE in I and aVL
- Occlusion of the left circumflex artery may cause STE in I, aVL, along with leads V5-6.
◾️DDx of high lateral OMI
- Anterior aneurysm involving the lateral wall.
- Pericarditis (suggested by more diffuse STE and lack of reciprocal STD).
- Early repolarization (suggested by large complexes with proportionally unimpressive ST deviation, lack of hyperacute T-waves, and benign early repolarization morphology in anterior leads).
- Right ventricular hypertrophy (suggested by a tall R-wave in V1, a terminal S-wave in lead I).
Posterior OMI
Posterior OMI is usually associated with inferior OMI and/or lateral OMI, though it can happen alone (5-10% of OMIs).
◾️ECG findings
- Precordial STD >1 mm that is maximal in V1-V4.
- Normally, there is a slight STE in V2-V3, so any STD is worrisome.
- This differentiates posterior OMI from subendocardial ischemia (wherein STD is usually maximal ~V4-V6).
- Tall R-waves (>30msec) in the right precordial leads.
- This represents a Q-wave equivalent.
- It is not invariably present, nor is it necessarily an early finding.
- Abnormal T-waves in the right precordium.
- TWI may occur as a mirror-image of hyperacute posterior T-waves.
- Prominent upright T-waves can occur as a mirror-image of posterior TWI (due to reperfusion).
- Ischemia in the inferior or lateral leads.
- Posterior ischemia often occurs together with inferior or lateral ischemia.
- Ischemic morphology using the mirror test.
- The mirror test inverts the precordial ECG to mimic what you would see using posterior leads. This is probably about as good as getting an ECG with posterior leads (V7-V9), and it’s much faster.
- Using paper: flip the ECG over and look through it as shown below.
◾️DDx of posterior OMI
- RV hypertrophy may cause a tall R-wave in the right precordial leads and a strain pattern in V1-V3 with STD.
- Other features of RV hypertrophy may suggest this diagnosis (e.g., terminal S-wave in lead I, TWI in lead III).
- Digoxin or hypokalemia may cause scooped STD in the right precordial leads. Clues to suggest digoxin or hypokalemia:
- STD is diffusely present throughout the ECG (including the inferior leads).
- Other features:
- Hypokalemia: Prominent U-wave.
- Digoxin: short QTc, unusual rhythms.
- Subendocardial ischemia (NOMI):
- Maximal STD in ~V2-V4 favors posterior OMI, whereas maximal STD in ~V4-V6 favors subendocardial ischemia.
- If seen, posterior OMI is suggested by:
- Tall R-waves in the right precordium.
- Upright T-waves in the right precordium (however, TWI in the right precordium can be consistent with either posterior OMI or subendocardial ischemia).
- Anterior de Winter T-wave pattern
- Suggested by the lack of a tall R-wave and broad/hyperacute T-waves.
Inferior OMI
◾️Core features of inferior OMI
- STE in inferior leads (👉caution: STE can be subtle).
- Should generally see STE in two inferior leads.
- STD in aVL (+/- Lead I).
- If no STD in aVL, the diagnosis should be questioned. STD in aVL may theoretically be obscured by simultaneous lateral ischemia (with STE in V5 or V6). However, evidence shows that STD occurs in 99% of inferior OMI.
- TWI in aVL may also support the diagnosis (this is the mirror image of hyperacute T-waves in the inferior leads).
◾️Culprit lesion in the inferior OMI
- Inferior OMI can happen due to RCA occlusion (80%) or, less commonly, occluded Cx (10%). See below.
◾️Additional features that can be seen in the inferior OMI
- Right precordium:
- RV MI may cause STE in V1.
- Manifestations of posterior ischemia (~V2-V4).
- ST depression or flattening.
- T-wave abnormalities.
- STE in V5, V6 may occur in left circumflex occlusions.
- In inferior MI, always systematically evaluate for possible RV myocardial infarction.
◾️ECG mimics of IMI
- LVH.
- It can be very difficult to sort out.
- Features that may support inferior MI:
- STE is disproportionately large compared to preceding S-waves.
- Ischemic features in other territories (e.g., right precordial STD).
- Old inferior MI
- Early repolarization. Clues that suggest benign early repolarization:
- Benign early repolarization may also be present in precordial leads.
- Benign-appearing morphology (e.g., concave STE, J-waves).
- Absence of STD in aVL.
- PE can cause STE in inferior leads and V1, with reciprocal changes in aVL.
- Diffuse and prominent TWI in the inferior and anterior leads may help suggest PE.
Right Ventricular OMI (RVMI)
- Diagnosis of RVMI is important since the supportive management is different (more on this here).
- RVMI most commonly happens in the context of inferior OMI, but it can rarely happen alone.
- In both conditions (either in inferior MI or isolated RVMI), it almost always occurs due to a proximal RCA occlusion.
- Stepward approach to the diagnosis of RVMI on ECG is shown in the figure below.
◾️RVMI in the context of inferior MI
- Diagnose an inferior MI.
- Look for signs of an RCA culprit lesion (table below👇).
- Look for evidence of RV ischemia
- STE of at least 0.5 mm in V1 suggests RVMI (STE may extend to V2-V4, but it should be maximal in V1-V2).
- Right-sided ECG showing >0.5-1 mm STE in V4R may be the most accurate ECG finding
👉When in doubt, POCUS may be the fastest way to evaluate RV function.
◾️Isolated RVMI. This may occur in two situations:
- Isolated RV marginal branch occlusion.
- Old inferior MI with a new proximal occlusion of the right coronary artery (the inferior wall is already scarred, so only the RV MI causes ST elevation).
- ECG: The main finding is STE in V1-V3 (which will tend to be misdiagnosed as anterior MI).
OMI in the Context of LBBB or Paced Rhythm
A new LBBB in the context of acute MI implies a large infarction, which is potentially an indication for catheterization. However, in practice, many patients present with a chronic LBBB, so the finding of LBBB alone is nonspecific. For patients with either LBBB or an RV-paced rhythm, ischemia may be diagnosed using the Smith-modified Sgarbossa criteria as listed below.
Smith-Modified Sgarbossa Criteria (OMI is revealed if ANY criteria is met, even in just a SINGLE lead) 7:
- Concordant STE of at least 1 mm in any lead.
- Concordant STD of at least 1 mm in V1-V3.
- Excessive discordant STE or discordant STD in any lead that is >1 mm and also >25% of the depth of the preceding R-wave or S-wave, respectively (figure below). However, this doesn’t apply in extreme tachycardia, pulmonary edema, hyperkalemia, or hypertension (if these conditions are present, then treat them and repeat the ECG)
◾️Other features that may support the presence of ischemia:
- Marked convexity of the ST segment (especially in the precordial leads).
- T-wave inversion in leads with a negative QRS (concordant TWI).
- Substantial qualitative changes compared to the baseline ECG.
De Winter Pattern
This pattern reflects nearly complete LAD occlusion and should be treated as an occlusive MI (e.g. with cath lab activation). However, a de Winter pattern can also occur in the inferior or lateral territories, with similar implications.
Classically defined as a combination of the following:
- Tall, hyperacute T-waves in the precordial leads.
- Up-sloping STD >1 mm in the precordial leads.
- Absence of STE in the precordial leads.
- Normal QRS duration.
- Often, mild STE is seen in aVR
Abnormalities are often maximal around V3. 9
Wellens Pattern
- Wellens patterns reflect transient occlusion of the “LAD” with spontaneous reperfusion. Clinically, this is often associated with a history of chest pain, which has currently resolved.
- Wellens pattern A refers to biphasic TW, suggestive of very recent occlusion (figure above). Over time, Wellens pattern A will evolve into deep, symmetric TW inversion (pattern B).
- Wellens pattern B is less immediately worrisome because it implies that the vessel has managed to remain open for a bit longer, without re-occluding.
- Wellens patterns do not indicate an active occlusive MI, but they imply the presence of a high-grade proximal LAD lesion that needs to be taken very seriously. One series found that most patients with Wellens syndrome who didn’t undergo revascularization went on to develop an anterior OMI within a few weeks. Patients with Wellens pattern should not undergo stress testing, as this may trigger an acute MI. 9
◾️ECG criteria for Wellens pattern
- TWI in V2-V3 (and possibly extending out as far as V5).
- Preservation of the RW (if the RW is replaced by deep Q-waves, this suggests an anterior aneurysm).
- Evolution over time (in particular, Wellens A is never a static pattern).
- Normal finding in children
- TWI in the right precordial leads (V1-3) is a normal finding in children, representing the dominance of right ventricular forces.
- TWI in the right precordial leads may persist into adulthood (called “persistent juvenile T-wave pattern”). TWIs are asymmetric, shallow (<3mm), and usually limited to leads V1-3
- LBBB: Produces TWIs in the lateral leads I, aVL, and V5-6.
- LVH: Produces TWIs in the lateral leads I, aVL, V5-6 (left ventricular ‘strain’ pattern), with a similar morphology to that seen in LBBB.
- RBBB: Produces TWIs in the right precordial leads V1-3.
- RVH: Produces TWI in the right precordial leads V1-3 (right ventricular ‘strain’ pattern) and also the inferior leads (II, III, aVF).
- PE: Acute right heart strain (e.g., secondary to massive PE) produces a similar pattern to RVH, with TWIs in V1-3 and inferior leads.
- Raised intracranial pressure (e.g., via SAH): Produces widespread deep TWIs with a bizarre morphology.
- Hypertrophic cardiomyopathy (HCM):
- The chief abnormality associated with HCM is left ventricular hypertrophy (LVH), of variable degree from mild hypertrophy (13-15 mm) to extreme myocardial thickening (30-60 mm).
- The most commonly observed pattern is asymmetrical thickening of the anterior interventricular septum (aka asymmetrical septal hypertrophy). Typical ECG findings include: deep, narrow (“dagger-like”) Q waves in lateral (I, aVL, V5-6) +/- inferior (II, III, aVF) leads.
- Other less common patterns of LVH include apical hypertrophy (10%). The classic ECG finding in apical HCM is giant T-wave inversion in the precordial leads.
- Wellens syndrome Type 1: T waves go up (initial deflection positive) then down (terminal deflection is negative).
- Hypokalemia: T waves go down, then up.
Subendocardial Ischemia Pattern
This pattern reveals diffuse, severe subendocardial ischemia of the entire myocardium, and it does not indicate active coronary vessel occlusion.
◾️ECG features of subendocardial ischemia
- Diffuse STD, which is maximal in V4-V6 and lead II.
- No evidence of focal OMI (e.g., no STE in leads other than aVR and V1).
- If very severe, this may be accompanied by STE in aVR (and sometimes V1 as well). Cause of STE in AVR: Two possible mechanisms:
- Diffuse subendocardial ischemia, with STD in the lateral leads producing reciprocal change in aVR (most common)
- Infarction of the basal septum, i.e., a STEMI involving aVR
- The basal septum is supplied by the first septal perforator artery (a very proximal branch of the LAD), so ischemia/infarction of the basal septum would imply involvement of the proximal LAD.
◾️Clinical significance of severe subendocardial ischemia with STE in aVR
- This pattern signifies diffuse, severe subendocardial ischemia of the entire myocardium.
- Clinical causes include
- Left main coronary artery (LMCA) stenosis
- Proximal left anterior descending artery (LAD) stenosis
- Severe triple vessel disease
- Hypoxia or hypotension, for example, after resuscitation from cardiac arrest.
- Diffuse ischemia may be caused in various ways:
- It can reflect acute plaque rupture (Type I MI) with partial occlusion of the vessel. This is a very scary scenario, because there is a lot of myocardium in jeopardy if the stenosis progresses to an acute occlusion.
- It can reflect stable coronary stenosis plus acute physiological stress (Type-2 MI).
- Management depends on clinical context:
- If context suggests a Type I MI (e.g., patient presenting with chest pain): treat medically for MI and pursue prompt catheterization. However, be cognizant that patients will often have severe coronary artery disease that cannot be treated by percutaneous coronary intervention. Therefore, consider holding P2Y12 inhibitors, since CABG may be needed.
-
If context suggests a Type-2 MI (e.g,. patient presenting with gastrointestinal hemorrhage): treat the underlying cause of stress and follow the ECGs carefully to ensure resolution of ischemia.
◾️Clinical significance of milder subendocardial ischemia without STE in aVR
- Nonocclusive MI of any vessel causing subendocardial ischemia will tend to create this same ECG pattern. (For some reason, subendocardial ischemia doesn’t localize on ECG.)
- This is perhaps the most classic ECG pattern associated with nonocclusive MI (NOMI).
ED Evaluation of Chest Pain Patient with Suspected ACS
◾️Phases of ED Evaluation
- Phase 1: STEMI (or STEMI-equivalent) Recognition
- Primary goal: rapid 12-lead ECG acquisition & interpretation.
- Early activation of reperfusion pathways and resource mobilization.
- Time-sensitive (<10 minutes).
- Disposition: Admission or immediate transfer to PCI-capable center.
- Phase 2: Rule-Out of ACS (OMI Evaluation)
- Serial ECGs and cardiac biomarkers (troponin-based strategies).
- Diagnoses established: evolving STEMI (STEMI-equivalent), NOMI, or alternate conditions.
- Disposition: admission/transfer for “rule-in” cases; continued evaluation if “rule-out.”
- Observation vs inpatient strategies depend on resources and protocols.
- Phase 3: Consideration of Significant CAD
- Focused on identifying obstructive CAD after exclusion of AMI.
- Disposition strategies:
- Safe ED discharge with no further evaluation.
- Outpatient follow-up (primary care or cardiology).
- Continued ED/observation evaluation ± cardiology consultation.
- Inpatient admission for extended diagnostic work-up.
◾️Risk Stratification
- Occurs during Phases 1 & 2.
- Tools: Physician Gestalt Plus:
- High-risk: STEMI, NSTEMI, abnormal ECG, elevated troponin, high scores → admit/transfer.
- Intermediate-risk: requires further in-hospital evaluation (ED/observation/inpatient).
- Low-risk: suitable for ED discharge with follow-up, often no immediate further testing required.
◾️Intermediate-Risk Pathway
- Managed via observation units (physical or “virtual” within ED).
- Use of accelerated diagnostic protocols (ADPs).
- Additional testing often employed: stress testing, echocardiography, coronary CTA, nuclear imaging.
- Evidence: observation-based strategies reduce unnecessary admissions without increasing adverse outcomes.
◾️Low-Risk Pathway
- Identified by low HEART, HEART Pathway, or EDACS-ADP scores.
- Safe for ED discharge with structured follow-up.
- Outpatient risk stratification may include stress testing, imaging, or CTA.
- Some require no further evaluation for CAD.
📍Key Points
- ~75–80% of chest pain patients can be safely evaluated and discharged.
- ED-based evaluation reduces costs (20–50% compared to inpatient admission) and avoids iatrogenic harms.
- Success depends on ED resourcing (staff, space, biomarkers, cardiology access).
Management of ACS
Universal Initial Measures
- Establish IV access, perform a 12-lead ECG, and initiate cardiac monitoring.
- Target hemoglobin >8-10 mg/dL. 10
- Generally, the optimal hemoglobin level should be tailored to individual patient specifics.
- ACC 2025: Transfusion to ≧10 g/dL may be reasonable to reduce cardiovascular events (Class IIb) *.
- Supplemental oxygen (ACS 2025):
- If oxygen saturation is <90%, supplemental oxygen to increase saturation to ≧90% is recommended (ACC 2025 Class I) *.
- If oxygen saturation is ≧90%, routine administration of supplemental oxygen isn’t recommended because it doesn’t increase cardiovascular outcomes (ACC 2025 Class III).
- Target potassium >3.5 mEq/L and magnesium >2 mEq/L (avoids arrhythmia).
- Maximum medical management of ACS refers to administration of aspirin + P2Y12 inhibitor +/- anticoagulant (as per cardiology discretion/institutional protocol) +/- nitroglycerin (if not contraindicated).
Lab Panel
◾️Recomended laboratory tests for hospitalized ACS patient *,*.
Laboratory Tests in Hospitalized ACS Patients (2025 AHA Guideline + Selected Considerations)
| Test | Purpose | Timing |
|---|---|---|
| Complete blood count (CBC) | Detect anemia, leukocytosis, or thrombocytopenia | On admission |
| Basic metabolic panel (BMP) | Assess electrolytes, renal function (creatinine, potassium, magnesium) | On admission and as clinically indicated |
| Coagulation profile (PT/INR, aPTT) | Baseline before fibrinolysis, anticoagulation, or invasive procedures | On admission |
| Lipid panel (LDL-C, HDL-C, triglycerides) | Guide secondary prevention and lipid-lowering therapy | Within 24 hours of admission |
| HbA1c | Screen for diabetes or assess long-term glycemic control | If not done in prior 3 months |
| Renal & hepatic function tests | Guide dosing of antithrombotics, contrast, and statins | On admission |
| BNP / NT-proBNP | Prognostic marker, assess heart failure risk | Optional, as clinically indicated |
| CRP / hs-CRP | Prognostic utility, not for acute decision-making | Optional |
| TSH | Identify thyroid disease as cause of arrhythmia (e.g., AF) | If arrhythmia present |
| Iron panel (ferritin, transferrin saturation) | Evaluate for iron deficiency anemia; emerging evidence suggests prognostic relevance in ACS | Selective: only if iron deficiency suspected |
Note: Iron studies are not part of 2025 AHA/ACC ACS guideline–directed care, but may be considered in patients with suspected iron deficiency anemia. Emerging ESC evidence suggests prognostic importance.
🔎 Considerations
- Lipid profile (recommended as soon as possible after presentation, because LDL-C levels decrease modestly beginning 24 hours from symptom onset) *.
- Iron studies (if iron deficiency is suspected).
- Myocardial iron deficiency may impair the ability to recover from MI *.
- IV iron repletion should be considered for patients with iron-deficiency anemia, especially for patients with reduced ejection fraction.
- Myocardial iron deficiency may impair the ability to recover from MI *.
Risk Stratification
◾️Risk stratification tools
- The GRACE risk model (Global Registry of Acute Coronary Events) and TIMI risk score (Thrombolysis in Myocardial Infarction) were derived for inpatients, examining the need for invasive therapy.
- Patients with a high GRACE score (e.g., > 140) may require immediate, aggressive interventions, such as a coronary angiogram within 24 hours of admission.
◾️Bedside evaluation
-
Risk stratification and hemodynamic status can be rapidly determined at the bedside. This may be very helpful regarding management decisions, resource allocation. For example, the presence of severely reduced LV systolic function suggests that the patient may respond poorly to beta-blockers (management of cardiogenic shock 👉 here).
- Vitals
- Shock index (HR/SBP) > ~ 0.8 suggests significant instability and possible shock.
- Narrow pulse pressure (less than 25% of SBP) may suggest low cardiac output.
- MAP <65 mmHg or a significant drop from baseline is concerning for hemodynamic instability.
- Hemodynamically significant bradycardia (e.g., HR< ~30) should always raise concern for shock (even if BP is maintained by compensatory systemic vasoconstriction).
- Skin perfusion
- Cool extremities, mottling, e.g., and prolonged capillary refill are suggestive of low cardiac output.
- Echo
- RV, LV size and function?
- Mitral and aortic valve function (exclude severe regurgitation)
- IVC size and collapsibility?
- Pericardial effusion?
- Lung ultrasound
- B-Lines? Bilateral and diffuse anterior B-lines suggest elevated pulmonary capillary wedge pressure (can be a sign of cardiogenic pulmonary edema).
- Vitals

Opioid
- Avoid opioids unless the decision to proceed with immediate reperfusion therapy has already been made. Then opioids may be administered to alleviate symptoms during the time between evaluation and definitive reperfusion.
Beta-blocker
-
STEMI: It is reasonable to administer IV beta blockers at the time of presentation to patients with STEMI and no contraindications to their use who are hypertensive or have ongoing ischemia.
-
NSTEMI: (Oral) beta blockers are recommended for all ACS within the first 24 h without contraindications, but there is no strong evidence or recommendation that beta blockers should be started in the ED.
◾️⚠️Contraindications
- Bradycardia (heart rate <60 bpm).
- Heart block (PR interval >240 ms, or second/third degree heart block); most commonly seen in RCA occlusions.
- Heart failure (e.g., cardiogenic pulmonary edema).
- Cardiogenic shock, or risk factors for developing shock (e.g., HR >110, SBP< 120, or Shock index > ~0.8).12
- Clinical and echocardiographic risk stratification may help predict which patients will tolerate beta-blockade.
Tachycardia is a relative contraindication to beta-blockade. That may be implausible, but compensatory tachycardia may reflect underlying instability, which indicates an inability to tolerate beta-blockers.
◾️Dosing
- IV beta-blockade is generally achieved with sequential 5-mg doses of metoprolol (up to a total of 15 mg, as tolerated/required).
- For most patients, starting an oral beta-blocker within 24 hours is adequate. Preferred agents are metoprolol or carvedilol.
Nitroglycerin
There’s no robust evidence supporting the use of nitroglycerine in STEMI in the reperfusion era. Nevertheless, nitroglycerine may be useful for symptom management (e.g., relief from anginal chest pain, management of hypertension, and treatment of pulmonary edema).
◾️⚠️Contraindications
- Hypotension.
- Aortic stenosis (relative contraindication).
- RV infarction.
- Recent use of phosphodiesterase inhibitors:
- Sildenafil or vardenafil within 24 hours.
- Tadalafil within 48 hours.
◾️Dosing
- Initially may be given as a sublingual tablet of 0.4 mg q5 minutes (up to three doses).
- If the patient responds favorably, may transition to a nitroglycerine infusion (e.g., 10-50mcg/min for ongoing chest pain)
PCI
◾️💡General indications for emergent PCI
- ECG Features:
- ✔️Occlusive MI.
- ✔️Recurrent dynamic ECG changes suggestive of ischemia (especially intermittent ST elevation).
- ✔️NOMI with a high risk of ischemic events (urgent PCI within 24 hours).
- 🧮 GRACE ACS Risk and Mortality: A score of > 140 suggests PCI
- Myocardial dysfunction
- ✔️Hemodynamic instability (including cardiogenic shock, or new/worsening heart failure attributable to the acute MI, e.g., pump failure, ischemic mitral regurgitation).
- ✔️Electrical instability (ventricular tachycardia or ventricular fibrillation).
- ✔️Recurrent or persistent chest pain that is refractory to medical therapy.
⏐If unsure about need for PCI: optimize medically, obtain serial ECGs & echocardiography.
◾️Relative contraindications to PCI
- Severe renal failure.
- Patients at high risk of bleeding (after stenting, the patient must remain on dual antiplatelet therapy).
- Patients who are unlikely to be compliant with dual antiplatelet therapy.
- Low life expectancy (e.g., severe dementia, advanced malignancy with poor oncologic prognosis, extremely elderly).
- Patients with a history of prior CABG may benefit less from PCI. More on this here.
◾️Non-occlusive MI (NOMI): early invasive strategy vs. conservative strategy
- Patients with NOMI without indications for emergent PCI (as listed above) may be treated either with an early invasive strategy (e.g., catheterization within 24 hours) or with a conservative strategy (including medical management followed by a stress test, with PCI performed only if there is recurrent spontaneous ischemia or a positive stress test).
- An early invasive strategy generally reduces the risk of recurrent myocardial ischemia or rehospitalization for acute coronary syndrome. However, a meta-analysis involving 10,150 patients found no difference in the risk of mortality after one year (4.3% vs. 4.4%). ESC 2020 guidelines likewise state that an early invasive strategy doesn’t reduce all-cause mortality in the overall population of patients with NSTEMI.
- When unclear, the decision to pursue PCI will be made in conjunction with cardiology consultation. This should take numerous factors into account (e.g., the specifics of ECG and echocardiography findings, renal function, indicators of risk due to myocardial ischemia, other active medical problems, and patient preferences).
◾️Maximal medical management for patients undergoing primary PCI
- This will be determined by cardiology. The typical regimen includes
- Aspirin: 162-325 mg PO (non-enteric-coated, chewable)
- P2Y12 Inhibitor: Ticagrelor 180mg or Clopidogrel 600 mg PO as per cardiology.
- Unfractionated heparin: 60-70 units/kg (max 4,000-5,000 units) IV push loading dose, then 12-15 units/kg/h (max 1,000 units/h) infusion. Following successful PCI, heparin is discontinued (leaving the patient on aspirin and P2Y12 inhibitor).
- Aspirin: 162-325 mg PO (non-enteric-coated, chewable)
- For patients with ECGs suggestive of surgical coronary disease (e.g., diffuse ST depression with STE in aVR), it may be wise to initially hold dual antiplatelet therapy in anticipation of the possibility of CABG surgery. A potent P2Y12 inhibitor may be initiated in the catheterization laboratory if coronary anatomy is amenable to PCI (either prasugrel or ticagrelor).
Thrombolytic For STEMI
-
If it will be >90-120 min total time from the first medical contact to PCI, or >90 min from STEMI diagnosis to PCI, and
-
Symptoms <12 h, or between 12-24 h if “a large area of the myocardium is at risk or hemodynamic instability is present”, or ECG shows high “acuteness” (large upright T waves, persistent R waves, persistent ST elevation).
-
Tenecteplase (TNK-tPA)
-
Reconstitute a 50-mg vial in 10 mL sterile water (5 mg/mL)
-
Single weight-based bolus, IV push over 5 sec
-
<60 kg: 30 mg
-
60-69 kg: 35 mg
-
70-79 kg: 40 mg
-
80-89 kg: 45 mg
-
>90 kg: 50 mg
-
-
Alteplase (tPA)
-
90-min weight-based infusion
-
>67 kg: Infuse 15 mg IV over 1-2 min; then, 50 mg over 30 min; then, 35 mg over the next 60 min (ie, 100 mg over 1.5 h)
-
≤67 kg: Infuse 15 mg IV over 1-2 min; then, 0.75 mg/kg (max 50 mg) over 30 min; then, 0.5 mg/kg (max 35 mg) over 60 min
-
◾️Maximal medical management for STEMI patients receiving thrombolytic
- The typical regimen includes
- Aspirin: 162-325 mg PO (non-enteric-coated, chewable)
- P2Y12 Inhibitor: Clopidogrel 300 mg PO
-
Enoxaparin:
-
<75 y old: 30 mg IV bolus, then by 1 mg/kg subcutaneous (SC) (max 100 mg for the first dose) in 15 min
-
≥75 y old: No bolus, 0.75 mg/kg SC (max 75 mg for the first dose)
-
For creatinine clearance <30, give 1 mg/kg q24h (not 12 h)
-
Continue throughout hospitalization
-
-
Transfer to a PCI-capable facility should be carried out after the administration of thrombolytics.
-
Immediate PCI is indicated if there is no ECG evidence of reperfusion after thrombolytics (Rescue PCI).
-
After thrombolytics, at the receiving institution:
-
Routine immediate angiography +/- PCI in high-risk patients
-
Extensive STE, new LBBB, previous MI, Killip class ≥2, LV ejection fraction <35%
-
-
Routine angiography +/- PCI for all others within 24 h.
-
Absolute Contraindications
1. Any prior ICH
2. Known structural cerebral vascular lesion (eg, arteriovenous malformation)
3. Known malignant intracranial neoplasm (primary or metastatic)
4. Ischemic stroke within 3 mo
5. EXCEPT acute ischemic stroke within 4.5 h
6. Suspected aortic dissection
7. Active bleeding or bleeding diathesis (excluding menses)
8. Significant closed-head or facial trauma within 3 mo
9. Intracranial or intraspinal surgery within 2 mo
10. Severe uncontrolled hypertension (unresponsive to emergency therapy)
11. For streptokinase, prior treatment within the previous 6 mo
Relative contraindications
1. History of chronic, severe, poorly controlled hypertension
2. Significant hypertension on presentation (SBP >180 mm Hg or DBP >110 mm Hg)
3. History of prior ischemic stroke >3 mo
4. Dementia
5. Known intracranial pathology not covered in absolute contraindications
6. Traumatic or prolonged (>10 min) CPR
7. Major surgery (<3 wk)
8. Recent (within 2 to 4 wk) internal bleeding
9. Noncompressible vascular punctures
10. Pregnancy
11. Active peptic ulcer
12. Oral anticoagulant therapy
Aspirin
◾️Aspirin has been robustly demonstrated to cause mortality benefit following MI, and is considered the most important antithrombotic agent in MI.
◾️Indications: Should be given immediately to any patient with definite or probable Type-I MI (unless contraindicated).
◾️Dosing
- Loading dose: 325 mg of non-enteric-coated aspirin (e.g., four uncoated 81-mg tablets).
- Maintenance dose: 81 mg enteric-coated aspirin daily (higher doses are not more effective, but carry greater bleeding risk).
◾️⚠️Contraindications
- Active bleeding or very high risk of bleeding.
- Aspirin allergy (in which case ticagrelor or clopidogrel may be used instead; if clopidogrel is utilized, then a 600-mg loading dose should be given to achieve therapeutic levels sooner).
P2Y12 Inhibitors
All patients undergoing PCI will require a P2Y12 inhibitor. These inhibitors must be given as soon as possible to achieve onset by the time of PCI (PCI results in heightened platelet reactivity). The following discussion is focused on patients undergoing medical management.
◾️Role of P2Y12 inhibitors in medical management
- Adding a P2Y12 inhibitor has benefits even in patients who aren’t undergoing PCI (in terms of fewer myocardial infarctions and potentially reduced mortality).
- Evidence supporting the use of P2Y12 inhibitors is more robust than evidence supporting the use of heparin anticoagulation.
-
General consideration
-
Thienopyridine, a prodrug, requires conversion to its active metabolite form and, therefore, has unpredictable effects.
- For patients on oral anticoagulation, clopidogrel may be favored over ticagrelor.
-
-
Dose
-
Loading dose of 300 mg or 600 mg, followed by 75 mg daily.
- The 600mg loading dose takes effect more rapidly (2 hours), compared to the 300 mg loading dose (~6 hours).
- The 300mg is often used for medical management. A 600-mg loading dose has been demonstrated to be superior to 300 mg before PCI.
-
-
General consideration
-
Does not require metabolic activation (unlike clopidogrel), thus has a faster onset.
-
Ticagrelor works more consistently than clopidogrel (which may have slow absorption and erratic metabolism, depending on individual CYP2C19 activity).
-
Reversible inhibition (unlike clopidogrel) enhances the rate of the offset of action but is also riskier in non-compliant patients.
- Ticagrelor has better ischemic outcomes than clopidogrel.
- Ticagrelor should be considered for MI patients who aren’t taking oral anticoagulation and aren’t at high risk of bleeding 13.
- Ticagrelor is safer if the patient ultimately needs CABG (shorter-acting).
-
- Dose: 180 mg loading dose, followed by 90 mg BID.
- Ticagrelor may cause nonexertional dyspnea as a side effect. Aminophylline may be utilized to transiently reverse dyspnea or bradycardia due to ticagrelor 14.
Anticoagulation
◾️Anticoagulation for patients undergoing PCI
-
- Angiography and PCI must always be in the setting of anticoagulation to avoid immediate procedural thrombosis.
- European Society of Cardiology lists unfractionated heparin as class I and enoxaparin as class IIa.
- Avoid fondaparinux due to the increased incidence of PCI catheter thrombosis.
- Following successful PCI, unfractionated heparin is discontinued (leaving the patient on aspirin and P2Y12 inhibitor).
◾️Anticoagulation in other patients
The rational use of heparin is as a temporary bridge to stabilize coronary stenosis before PCI. There is no evidence to support anticoagulation in patients who aren’t planned to undergo catheterization. However, short-term anticoagulation may be reasonable in patients who are evolving rapidly and might become candidates for catheterization.
- The traditional practice of providing heparin anticoagulation for 48 hours as “medical management” without subsequent PCI is not evidence-based and needs to be abandoned.
◾️1st-line anticoagulant: Fondaparinux 2.5 mg sq daily
- The ESC 2020 NSTEMI guidelines recommend fondaparinux as the first-line anticoagulant for patients not undergoing immediate catheterization (i.e., patients being medically managed, or receiving delayed PCI) 13.
- Compared to enoxaparin, fondaparinux caused less bleeding and reduced mortality in the OASIS-5 trial.
- Fondaparinux is contraindicated in renal dysfunction (GFR <20 ml/min).
- If fondaparinux is used alone, this will lead to an increase in catheter-related thrombi among patients undergoing PCI. However, this problem may be avoided by the addition of a single heparin bolus during the PCI procedure.
◾️2nd line anticoagulant: Low molecular-weight heparin (LMWH)
- It is useful when full anticoagulation is required for another reason (e.g., atrial fibrillation or pulmonary embolism).
- LMWH has more predictable pharmacology than unfractionated heparin (UFH), often making it safer and more effective. Indeed, meta-analyses have found that LMWH is superior to UFH in myocardial infarction.
- The usual dose is enoxaparin 1 mg/kg sq q12hr (contraindicated if GFR <30 ml/min).
◾️3rd line anticoagulant: Unfractionated heparin (UFH) infusion
- It is the only available option for patients with GFR<20 ml/min (who cannot be treated with fondaparinux or enoxaparin).
- UFH infusions have the highest risk of bleeding complications (compared to either fondaparinux or LMWH).
Special Circumstances
ACS in Cardiac Arrest
-
Attempt defibrillation for ventricular fibrillation (VF)/ventricular tachycardia (VT) and initiate high-quality cardiopulmonary resuscitation.
-
Consider transvenous pacemaker placement for significant bradycardia contributing to cardiogenic shock refractory to standard advanced cardiac life support (ACLS) and transcutaneous pacing when there will be a significant delay to the catheter lab.
-
VF/VT refractory to multiple defibrillations and standard ACLS:
-
Ensure optimal pad placement.
- Consider esmolol
- Consider veno-arterial extracorporeal membrane oxygenation (ECMO) followed by emergency PCI in patients deemed salvageable with a good baseline quality of life.
-
◾️Which Cardiac Arrest Patients with Return of Spontaneous Circulation Require the Catheterization Lab?
- All patients with STEMI on ECG require the catheterization lab.
- Patients with initial shockable rhythm who do not have diagnostic STE but occlusion MI is suspected (i.e., even in the absence of STEMI), catheterization lab activation is recommended. (see the following algorithm for risk stratification of comatose cardiac arrest patients). 15
ACS in Cardiogenic Shock
- The differential of cardiogenic shock in the setting of ACS is based on pump failure (LV and/or right ventricle), and mechanical complications (papillary rupture, free wall rupture, etc.) 18.
- ACS with resulting cardiogenic shock is an indication for emergency PCI, regardless of ECG findings or the time since the onset of symptoms.
-
The current guidelines give a class I, level B recommendation for emergency PCI of STEMI in cardiogenic shock, regardless of the time since onset.
-
The current guidelines recommend (I, B) fibrinolytics for STEMI patients with cardiogenic shock who are unsuitable candidates for either PCI or CABG.
- Emergency PCI is superior to thrombolytics in STEMI with cardiogenic shock, and emergency PCI is superior to “medical stabilization” with delayed PCI in this group.
-
Adjunct therapies to emergency reperfusion in ACS with cardiogenic shock 18:
-
Optimize the preload with fluids/blood as necessary based on hematocrit and fluid tolerance.
-
Consider intubation, mechanical ventilation, and paralysis to reduce the demand for cardiac output during the delay to definitive management.
-
Vasopressors in cardiogenic shock: Norepinephrine is first-line, then consider adding dobutamine for persistent low cardiac output after norepinephrine
-
Correct and prevent dysrhythmias (VT and VF).
-
- Consider veno-arterial ECMO to restore perfusion and prevent cardiac arrest while arranging for emergency PCI if the patient is worsening despite the above therapies.
Shifting from STEMI-NSTEMI to OMI-NOMI
📚Read more on this:
- 🔗 OMI/NOMI paradigm: OMI manifesto, Dr. Smith, and The OMI manifesto (EM:RAP). PDF Version of OMI Manifesto
- 📖 From ST-Segment Elevation MI to Occlusion MI. JACC Adv. 2024 Oct 8;3(11):101314.
- 📖 STEMI: A transitional fossil in MI classification? J Electrocardiol . 2021 Mar-Apr:65:163-169.
- 📖 Comparison of the ST-Elevation Myocardial Infarction (STEMI) vs. NSTEMI and Occlusion MI (OMI) vs. NOMI Paradigms of Acute MI, J Emerg Med. 2021 Mar;60(3):273-284
📚Guideline Link
- 2025 ACC/AHA/ACEP/NAEMSP/SCAI Guideline for the Management of Patients With Acute Coronary Syndromes
- 2023 ESC Guidelines for the management of acute coronary syndromes
- 2017 ESC Guidelines for STEMI
- 2020 ESC guidelines for NSTEMI
- 2018 ESC guidelines for myocardial revascularization
- 2020 NICE guidelines for ACS
🧑🏫Going further
📖References
1. Ogobuiro I, Wehrle CJ, Tuma F. Anatomy, Thorax, Heart, Coronary Arteries. [Updated 2021 Jul 28]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK534790/
2. Thygesen K, Alpert JS, Jaffe AS, Chaitman BR, Bax JJ, Morrow DA, White HD; Executive Group on behalf of the Joint European Society of Cardiology (ESC)/American College of Cardiology (ACC)/American Heart Association (AHA)/World Heart Federation (WHF) Task Force for the Universal Definition of Myocardial Infarction. Fourth Universal Definition of Myocardial Infarction (2018). Circulation. 2018 Nov 13;138(20):e618-e651. doi: 10.1161/CIR.0000000000000617. Erratum in: Circulation. 2018 Nov 13;138(20):e652. PMID: 30571511
3. Jung J, Bord S. Emergency department management of non-ST-segment elevation myocardial infarction. Emerg Med Pract. 2020 Jan;22(1):1-24. Epub 2020 Jan 1. PMID: 31855327
4. Fanaroff AC, Rymer JA, Goldstein SA, Simel DL, Newby LK. Does This Patient With Chest Pain Have Acute Coronary Syndrome?: The Rational Clinical Examination Systematic Review. JAMA. 2015 Nov 10;314(18):1955-65. doi: 10.1001/jama.2015.12735. PMID: 26547467
5. Kawahito K, Adachi H, Murata S, Yamaguchi A, Ino T. Coronary malperfusion due to type A aortic dissection: mechanism and surgical management. Ann Thorac Surg. 2003 Nov;76(5):1471-6; discussion 1476. doi: 10.1016/s0003-4975(03)00899-3. PMID: 14602269
6. Patton KK, Ellinor PT, Ezekowitz M, Kowey P, Lubitz SA, Perez M, Piccini J, Turakhia M, Wang P, Viskin S; American Heart Association Electrocardiography and Arrhythmias Committee of the Council on Clinical Cardiology and Council on Functional Genomics and Translational Biology. Electrocardiographic Early Repolarization: A Scientific Statement From the American Heart Association. Circulation. 2016 Apr 12;133(15):1520-9. doi: 10.1161/CIR.0000000000000388. Epub 2016 Mar 7. PMID: 27067089
7. Smith SW, Dodd KW, Henry TD, Dvorak DM, Pearce LA. Diagnosis of ST-elevation myocardial infarction in the presence of left bundle branch block with the ST-elevation to S-wave ratio in a modified Sgarbossa rule. Ann Emerg Med. 2012 Dec;60(6):766-76. doi: 10.1016/j.annemergmed.2012.07.119. Epub 2012 Aug 31. Erratum in: Ann Emerg Med. 2013 Oct;62(4):302. PMID: 22939607
8. Miranda DF, Lobo AS, Walsh B, Sandoval Y, Smith SW. New Insights Into the Use of the 12-Lead Electrocardiogram for Diagnosing Acute Myocardial Infarction in the Emergency Department. Can J Cardiol. 2018 Feb;34(2):132-145. doi: 10.1016/j.cjca.2017.11.011. Epub 2017 Nov 29. PMID: 29407007
9. Tzimas G, Antiochos P, Monney P, Eeckhout E, Meier D, Fournier S, Harbaoui B, Muller O, Schläpfer J. Atypical Electrocardiographic Presentations in Need of Primary Percutaneous Coronary Intervention. Am J Cardiol. 2019 Oct 15;124(8):1305-1314. doi: 10.1016/j.amjcard.2019.07.027. Epub 2019 Jul 27. PMID: 31455501
10. Ducrocq G, Gonzalez-Juanatey JR, Puymirat E, Lemesle G, Cachanado M, Durand-Zaleski I, Arnaiz JA, Martínez-Sellés M, Silvain J, Ariza-Solé A, Ferrari E, Calvo G, Danchin N, Avendaño-Solá C, Frenkiel J, Rousseau A, Vicaut E, Simon T, Steg PG; REALITY Investigators. Effect of a Restrictive vs Liberal Blood Transfusion Strategy on Major Cardiovascular Events Among Patients With Acute Myocardial Infarction and Anemia: The REALITY Randomized Clinical Trial. JAMA. 2021 Feb 9;325(6):552-560. doi: 10.1001/jama.2021.0135. PMID: 33560322; PMCID: PMC7873781
11. Stub D, Smith K, Bernard S, Nehme Z, Stephenson M, Bray JE, Cameron P, Barger B, Ellims AH, Taylor AJ, Meredith IT, Kaye DM; AVOID Investigators. Air Versus Oxygen in ST-Segment-Elevation Myocardial Infarction. Circulation. 2015 Jun 16;131(24):2143-50. doi: 10.1161/CIRCULATIONAHA.114.014494. Epub 2015 May 22. PMID: 26002889.
12. Wei Z, Bai J, Dai Q, Wu H, Qiao S, Xu B, Wang L. The value of shock index in prediction of cardiogenic shock developed during primary percutaneous coronary intervention. BMC Cardiovasc Disord. 2018 Oct 1;18(1):188. doi: 10.1186/s12872-018-0924-z. PMID: 30285644; PMCID: PMC6167806
13. Collet JP, Thiele H, Barbato E, Barthélémy O, Bauersachs J, Bhatt DL, Dendale P, Dorobantu M, Edvardsen T, Folliguet T, Gale CP, Gilard M, Jobs A, Jüni P, Lambrinou E, Lewis BS, Mehilli J, Meliga E, Merkely B, Mueller C, Roffi M, Rutten FH, Sibbing D, Siontis GCM; ESC Scientific Document Group. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021 Apr 7;42(14):1289-1367. doi: 10.1093/eurheartj/ehaa575. Erratum in: Eur Heart J. 2021 May 14;42(19):1908. Erratum in: Eur Heart J. 2021 May 14;42(19):1925. Erratum in: Eur Heart J. 2021 May 13;: PMID: 32860058.
14. Minner SA, Simone P, Chung BB, Shah AP. Successful Reversal of Bradycardia and Dyspnea With Aminophylline After Ticagrelor Load. J Pharm Pract. 2018 Feb;31(1):112-114. doi: 10.1177/0897190016680978. Epub 2016 Dec 5. PMID: 27920235
15. Rab T, Kern KB, Tamis-Holland JE, Henry TD, McDaniel M, Dickert NW, Cigarroa JE, Keadey M, Ramee S; Interventional Council, American College of Cardiology. Cardiac Arrest: A Treatment Algorithm for Emergent Invasive Cardiac Procedures in the Resuscitated Comatose Patient. J Am Coll Cardiol. 2015 Jul 7;66(1):62-73. doi: 10.1016/j.jacc.2015.05.009. PMID: 26139060.
16. Lemkes JS, Janssens GN, van der Hoeven NW, Jewbali LSD, Dubois EA, Meuwissen M, Rijpstra TA, Bosker HA, Blans MJ, Bleeker GB, Baak R, Vlachojannis GJ, Eikemans BJW, van der Harst P, van der Horst ICC, Voskuil M, van der Heijden JJ, Beishuizen A, Stoel M, Camaro C, van der Hoeven H, Henriques JP, Vlaar APJ, Vink MA, van den Bogaard B, Heestermans TACM, de Ruijter W, Delnoij TSR, Crijns HJGM, Jessurun GAJ, Oemrawsingh PV, Gosselink MTM, Plomp K, Magro M, Elbers PWG, van de Ven PM, Oudemans-van Straaten HM, van Royen N. Coronary Angiography after Cardiac Arrest without ST-Segment Elevation. N Engl J Med. 2019 Apr 11;380(15):1397-1407. doi: 10.1056/NEJMoa1816897. Epub 2019 Mar 18. PMID: 30883057.
17. Millin MG, Comer AC, Nable JV, Johnston PV, Lawner BJ, Woltman N, Levy MJ, Seaman KG, Hirshon JM. Patients without ST elevation after return of spontaneous circulation may benefit from emergent percutaneous intervention: A systematic review and meta-analysis. Resuscitation. 2016 Nov;108:54-60. doi: 10.1016/j.resuscitation.2016.09.004. Epub 2016 Sep 15. PMID: 27640933.
18. Holger Thiele, E Magnus Ohman, Suzanne de Waha-Thiele, Uwe Zeymer, Steffen Desch, Management of cardiogenic shock complicating myocardial infarction: an update 2019, European Heart Journal, Volume 40, Issue 32, 21 August 2019, Pages 2671–2683, https://doi.org/10.1093/eurheartj/ehz363.
19. Xi H, Yu RH, Wang N, Chen XZ, Zhang WC, Hong T. Serum potassium levels and mortality of patients with acute myocardial infarction: A systematic review and meta-analysis of cohort studies. Eur J Prev Cardiol. 2019 Jan;26(2):145-156. doi: 10.1177/2047487318780466. PMID: 31060369.
20. Kieboom BC, Niemeijer MN, Leening MJ, van den Berg ME, Franco OH, Deckers JW, Hofman A, Zietse R, Stricker BH, Hoorn EJ. Serum Magnesium and the Risk of Death From Coronary Heart Disease and Sudden Cardiac Death. J Am Heart Assoc. 2016 Jan 22;5(1):e002707. doi: 10.1161/JAHA.115.002707. PMID: 26802105; PMCID: PMC4859391.
21. Templin C, Ghadri JR, Diekmann J, Napp LC, Bataiosu DR, Jaguszewski M, Cammann VL, Sarcon A, Geyer V, Neumann CA, Seifert B, Hellermann J, Schwyzer M, Eisenhardt K, Jenewein J, Franke J, Katus HA, Burgdorf C, Schunkert H, Moeller C, Thiele H, Bauersachs J, Tschöpe C, Schultheiss HP, Laney CA, Rajan L, Michels G, Pfister R, Ukena C, Böhm M, Erbel R, Cuneo A, Kuck KH, Jacobshagen C, Hasenfuss G, Karakas M, Koenig W, Rottbauer W, Said SM, Braun-Dullaeus RC, Cuculi F, Banning A, Fischer TA, Vasankari T, Airaksinen KE, Fijalkowski M, Rynkiewicz A, Pawlak M, Opolski G, Dworakowski R, MacCarthy P, Kaiser C, Osswald S, Galiuto L, Crea F, Dichtl W, Franz WM, Empen K, Felix SB, Delmas C, Lairez O, Erne P, Bax JJ, Ford I, Ruschitzka F, Prasad A, Lüscher TF. Clinical Features and Outcomes of Takotsubo (Stress) Cardiomyopathy. N Engl J Med. 2015 Sep 3;373(10):929-38. doi: 10.1056/NEJMoa1406761. PMID: 26332547.


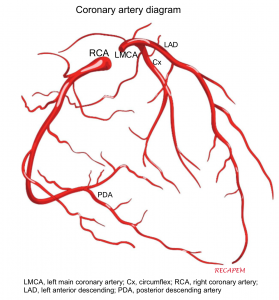

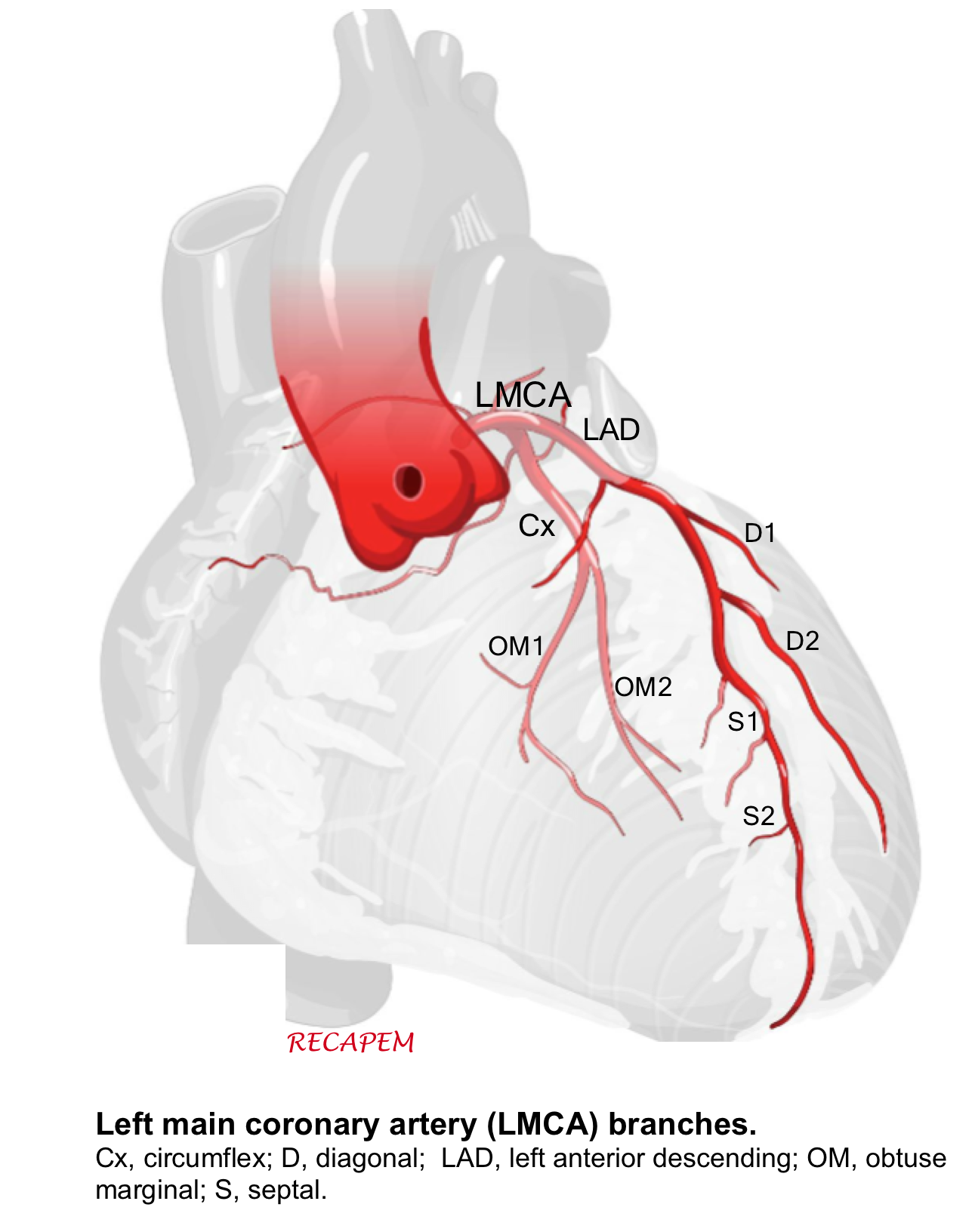
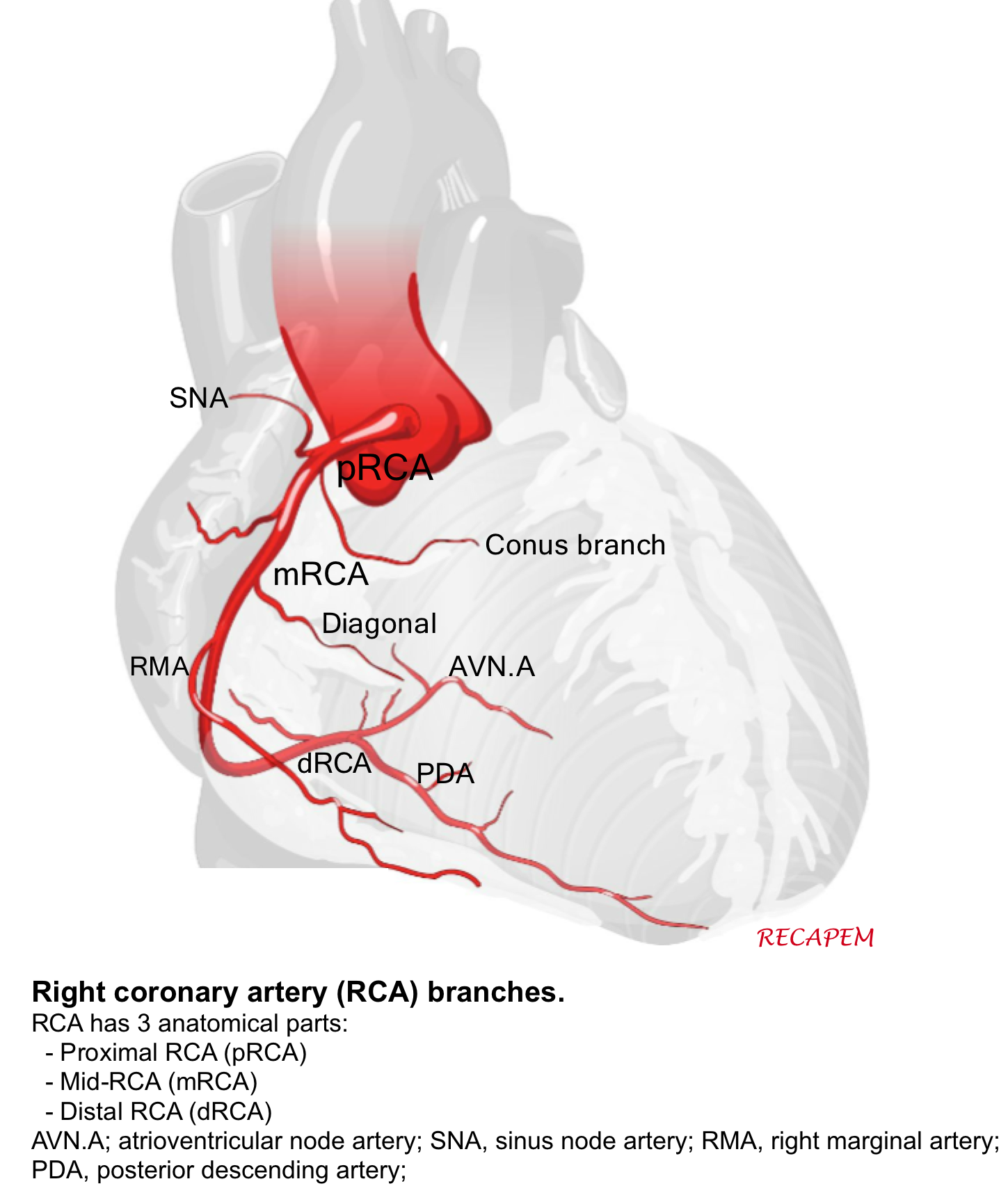

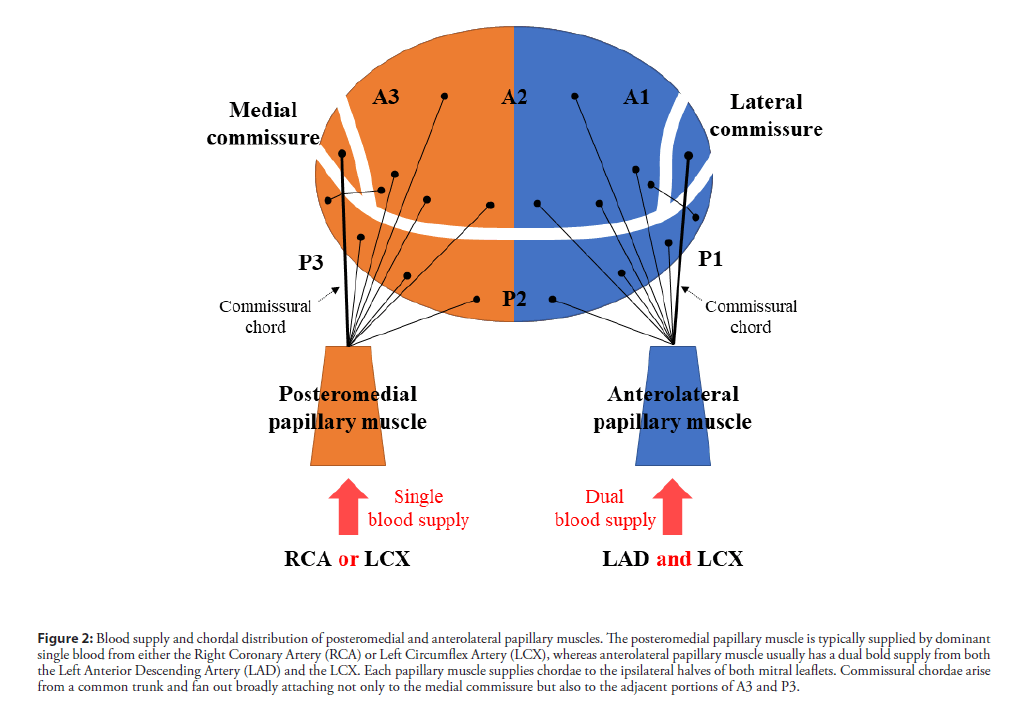
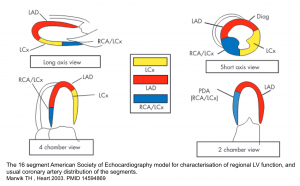
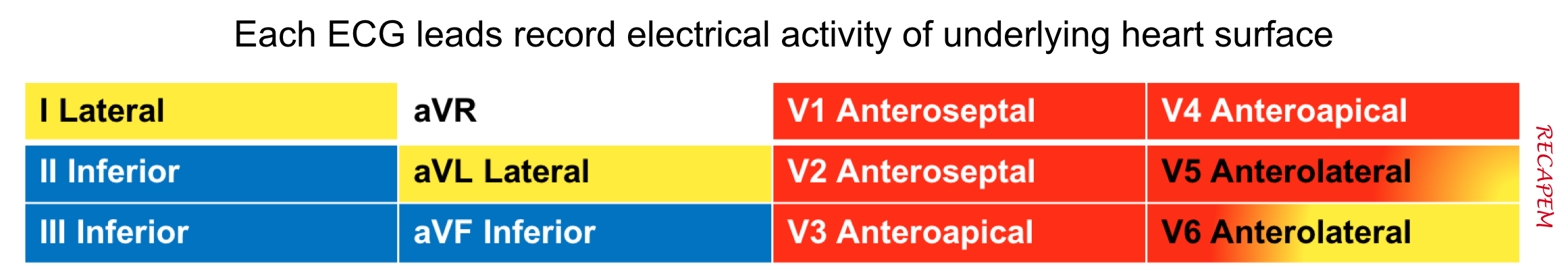
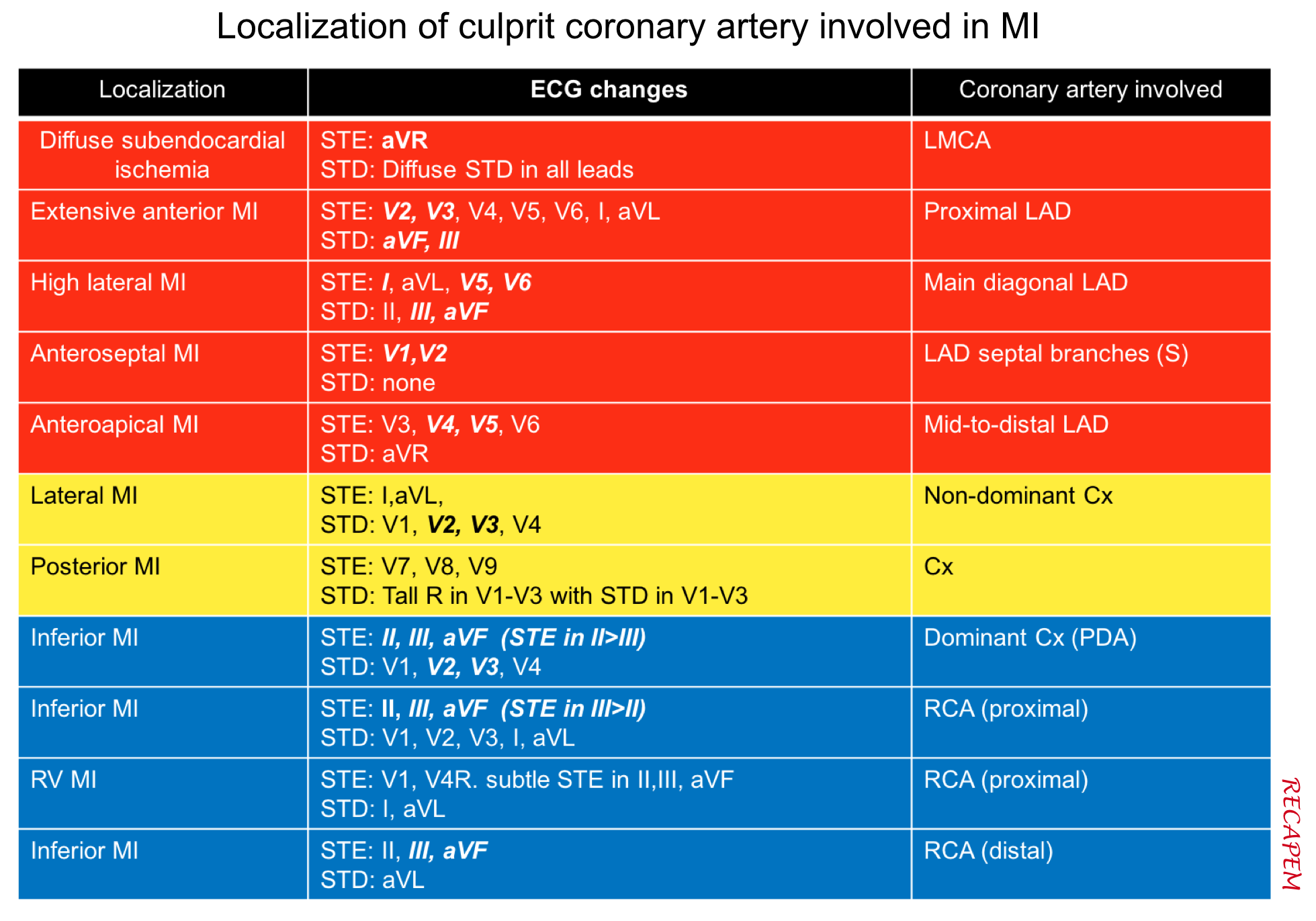
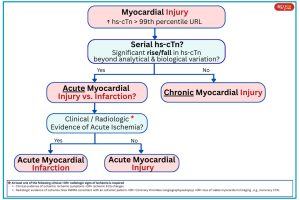
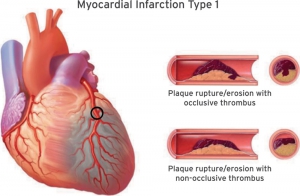
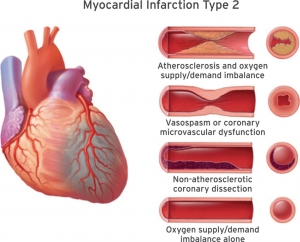
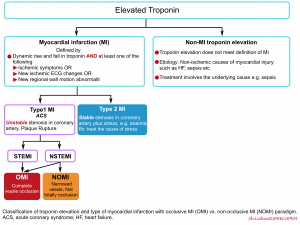
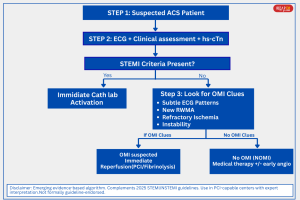
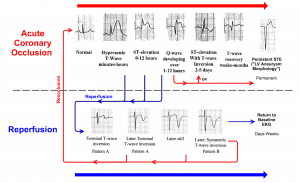
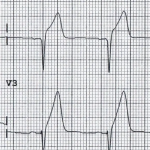
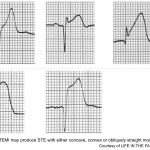
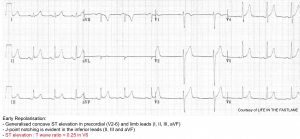
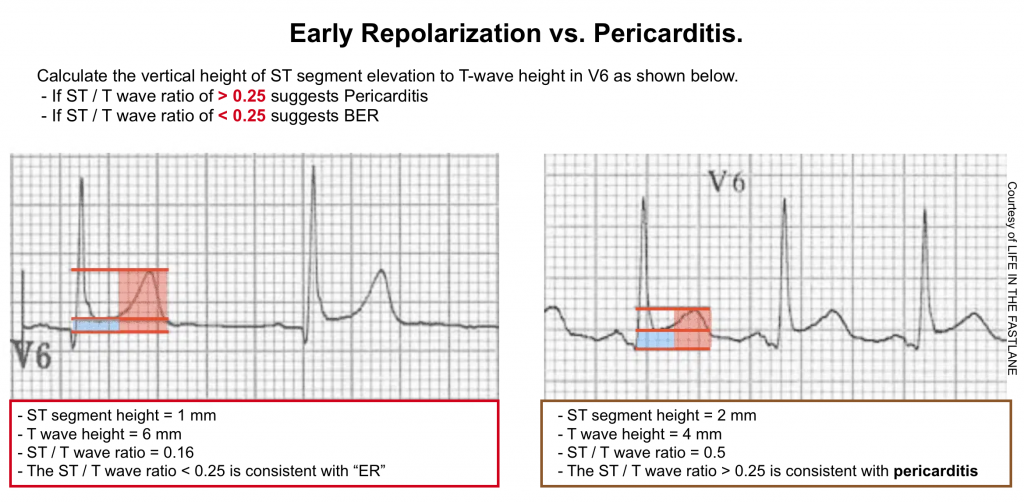
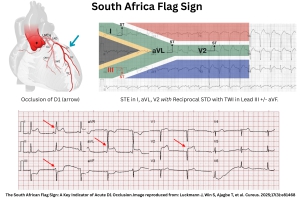
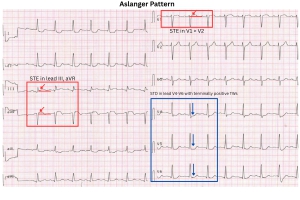
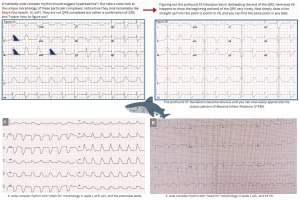
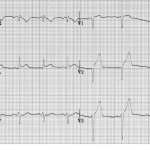
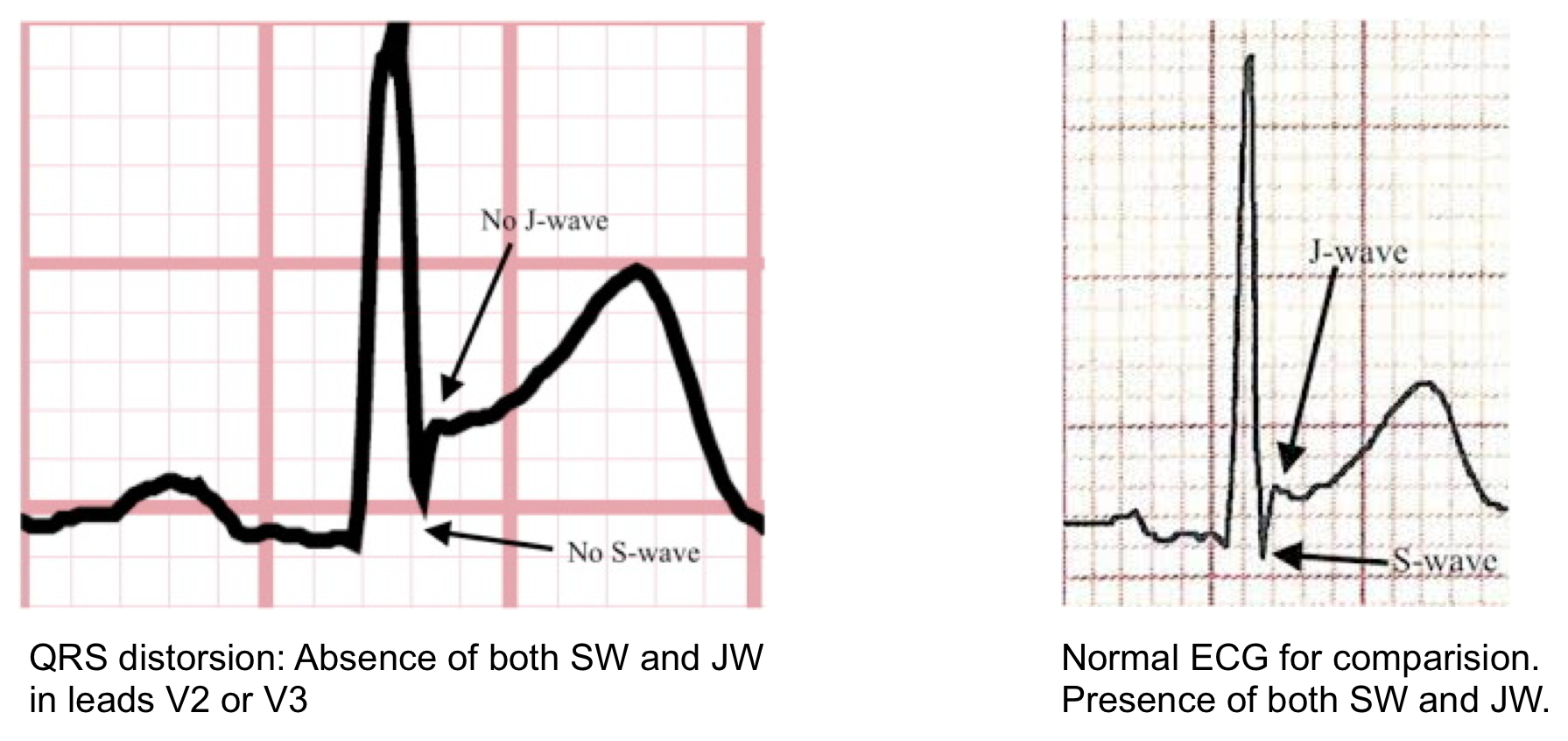
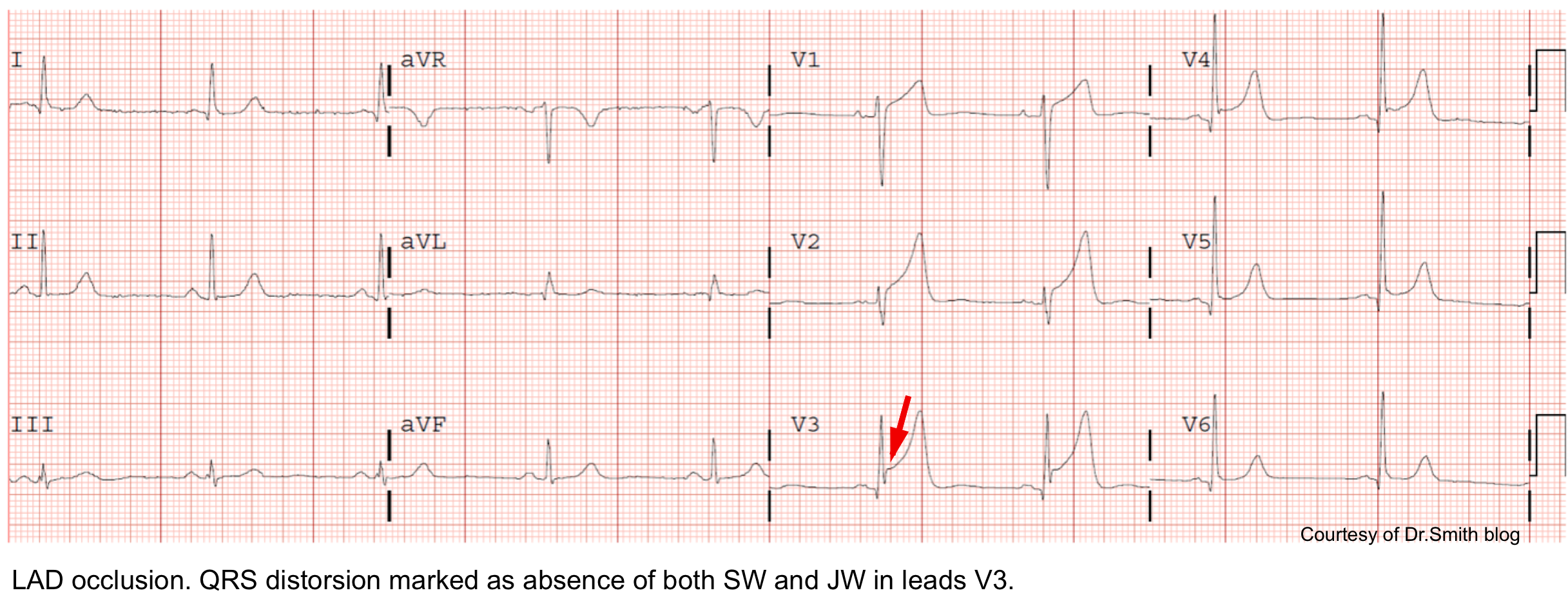
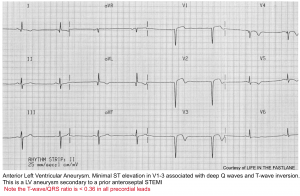
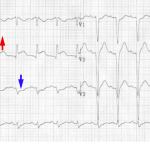
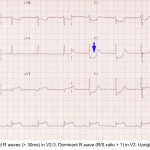
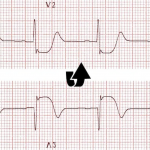
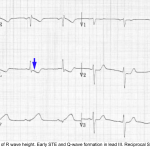
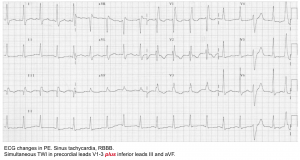
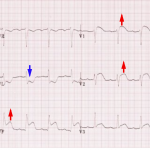
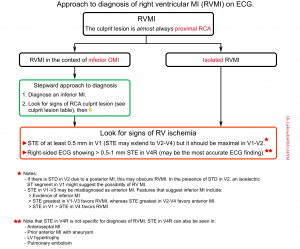

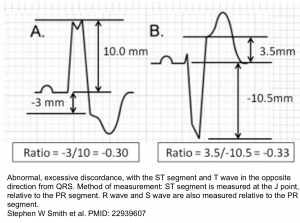
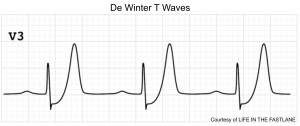
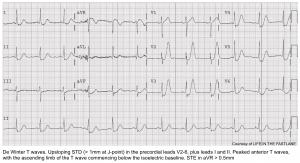
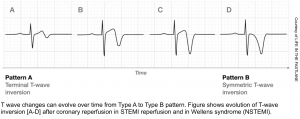
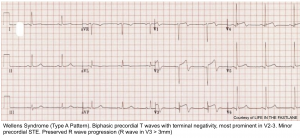
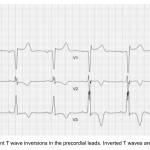
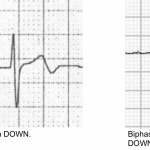
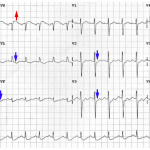
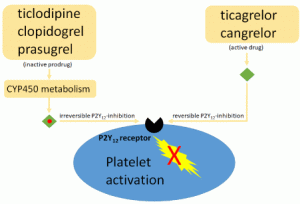
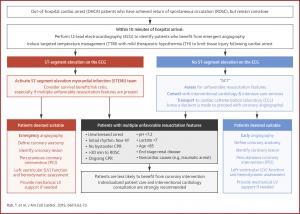
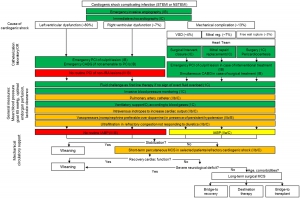



Add comment