6 May, 2023 by Shahriar Lahouti.
CONTENTS
- Preface
- Physiologic background
- Epidemiology and causes
- Clinical presentation
- Differential diagnosis
- Evaluation
- Management
- Prevention
- Going further
- References
Preface
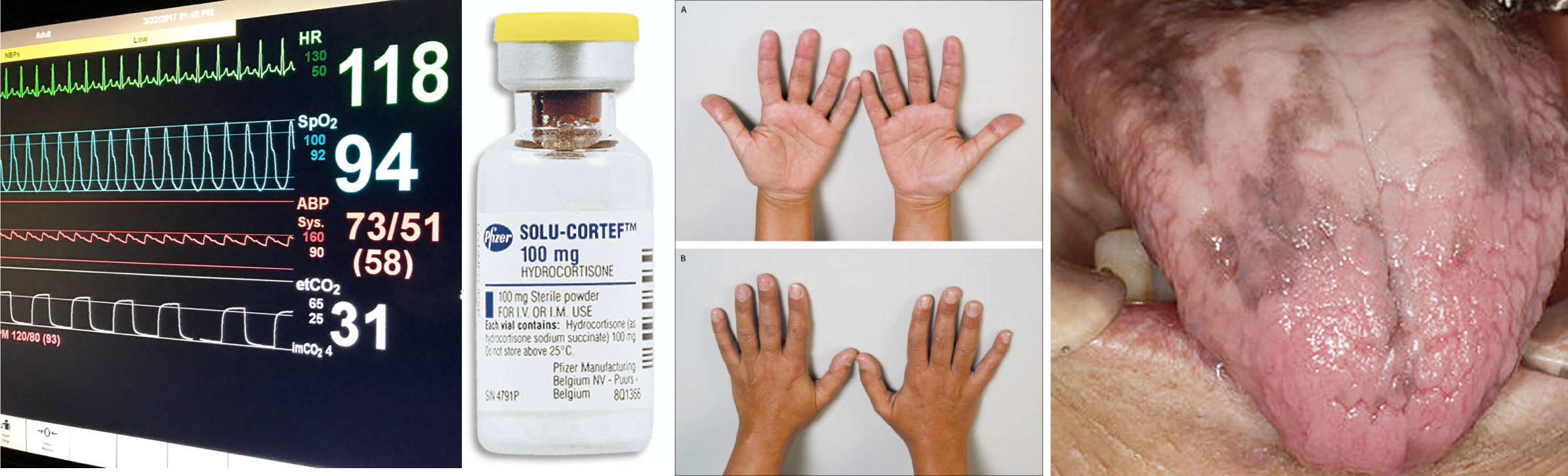
Adrenal crisis refers to the acute decompensation of a patient with adrenal insufficiency resulting in hemodynamic deterioration and refractory shock state. This is a medical emergency which requires immediate intravenous glucocorticoid administration and fluid resuscitation. Adrenal crisis should be included in the differential diagnosis of distributive shock, particularly for patients with risk factors for adrenal insufficiency.
Physiologic background
Adrenal gland function
- The adrenal gland is made up of 2 parts, the cortex and medulla.
- The adrenal cortex consists of 3 zones with each zone contributing the following hormone production as:
- Zona glomerulosa →mineralocorticoid aldosterone
- Zona fasciculata →glucocorticoid cortisol
- Zona reticularis →androgens
- The medulla produces catecholamines, including epinephrine, norepinephrine.
- The adrenal cortex consists of 3 zones with each zone contributing the following hormone production as:
Clucocorticoid
- Physiologic effects: Cortisol normally facilitates the body’s response to stress (trauma, infection) via following mechanisms:
- Suppression of inflammation: Cortisol inhibits pro-inflammatory cytokines (via mechanisms including depression of nuclear factor kappa b, i.e. NF-kB) *.
- Vascular tone: Cortisol normally functions synergistically with catecholamines to cause vasoconstriction.
- Metabolic effects: Cortisol is generally a catabolic hormone, which stimulates the production of numerous fuels in times of stress:
- Regulation: Cortisol is regulated by the hypothalamic-pituitary-adrenal (HPA) axis.
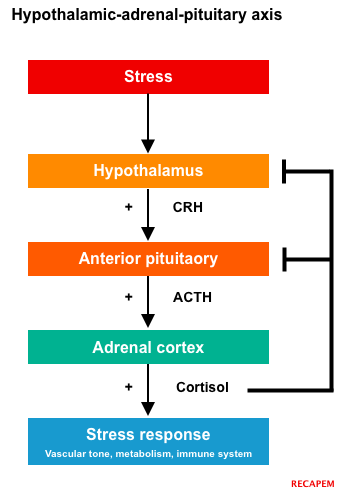
Mineralocorticoid (aldosterone)
- Physiologic effects: Aldosterone normally maintains the body’s salt balance and blood pressure.
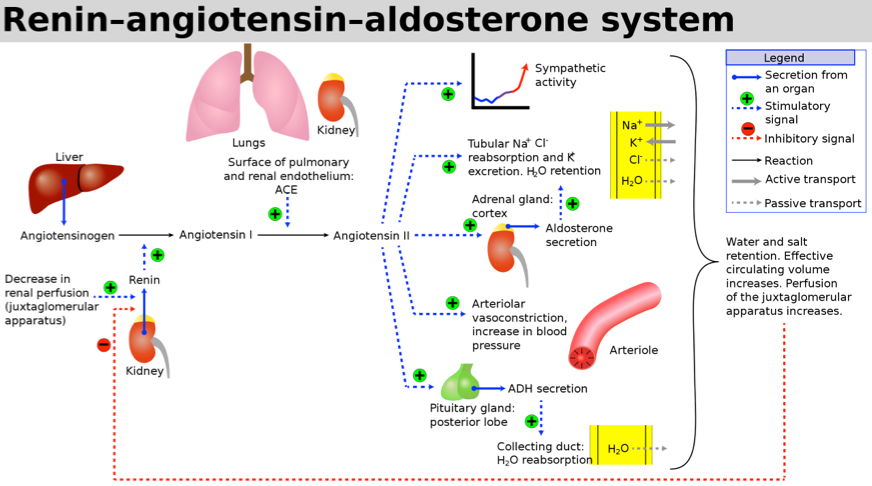
- Regulation: Aldosterone is regulated by the renin-angiotensin-aldosterone system (RAAS), shown below.
- In the setting of decreased blood flow to the kidney (as in hypovolemia), aldosterone causes the renal nephrons to conserve sodium (and water) and to secrete potassium and hydrogen ions.
Epidemiology and causes
Epidemiology
- The lifetime risk of adrenal crisis in patients with adrenal insufficiency is approximately 50% *.
- Risk factors for adrenal crisis in patients with adrenal insufficiency include *
- Advanced age
- Known patient with adrenal insufficiency or risk factors for developing AI (e.g. long–term exogenous glucocorticoid use)
- History of prior adrenal crises
- Medical conditions
- Autoimmune polyglandular syndromes
- Diabetes mellitus
- Thyrotoxicosis
- Diabetes insipidus
- Premature ovarian failure
- Adrenal crisis is associated with imminent death without prompt recognition and appropriate treatment.
- The adrenal crisis–associated rate of death may reach 6% of crisis events *.
Causes
- Adrenal crisis can be caused by an acute stressor in those with baseline adrenal insufficiency (most common) or by sudden damage to the hypothalamic-pituitary-adrenal axis (HPA axis).
- Chronic adrenal insufficiency plus acute stressor
- Chronic adrenal insufficiency (common causes) *
- Chronic steroid therapy (the most frequent cause of adrenal insufficiency)
- Chronic adrenal gland dysfunction:
- Addison’s disease (may be associated with other autoimmune disorders or endocrinopathies).
- Malignancy.
- Infection (esp. tuberculosis or fungal infection).
- Chronic anterior pituitary dysfunction:
- Granulomatous disease (e.g. tuberculosis, sarcoidosis).
- Neoplasms (e.g. craniopharyngiomas, adenomas).
- Iatrogenic (e.g. surgery, radiotherapy).
- Acute stressor
- Infection (the most common trigger)
- Reduced steroid dose: Non-adherence, steroid tapering, sudden withdrawal from exogenous steroids.
- Trauma/surgery.
- Strenuous activity, psychological stress
- Volume depletion
- Pregnancy
- Undiagnosed coexisting thyrotoxicosis
- Medications: Certain drugs may also contribute to adrenal insufficiency:
- Any medication that induces cytochrome P-450 3A4 (will increase hydrocortisone metabolism).
- Azoles (ketoconazole, fluconazole)
- Anti-epileptic drugs: Carbamazepine, phenobarbital, phenytoin
- Cholestyramine, etomidate, rifampin.
- Thyroid hormone (initiation of thyroxine therapy in a patient with undiagnosed hypoadrenalism)
- Infection (the most common trigger)
- Chronic adrenal insufficiency (common causes) *
- Acute failure of HPA axis
- Acute pituitary failure
- Pituitary apoplexy: It is an acute clinical condition caused by either hemorrhagic or non-hemorrhagic necrosis of the pituitary gland. Clinical presentations include thunderclap headache, visual deficit, ophthalmoplegia, altered mental status and hypopituitarism.
- Pituitary necrosis/bleeding (Sheehan syndrome) often postpartum.
- Tumors (pituitary adenoma or primary brain tumors with local invasion of pituitary)
- Trauma or complication of neurosurgery.
- Pituitary apoplexy: It is an acute clinical condition caused by either hemorrhagic or non-hemorrhagic necrosis of the pituitary gland. Clinical presentations include thunderclap headache, visual deficit, ophthalmoplegia, altered mental status and hypopituitarism.
- Acute adrenal gland failure
- Waterhouse-Friderichsen syndrome (adrenal infarction due to disseminated intravascular coagulation).
- Checkpoint-inhibitor immunotherapy for cancer may cause acute failure of the adrenal and/or pituitary gland *.
- Acute pituitary failure
- Chronic adrenal insufficiency plus acute stressor
Clinical presentation
Adrenal crisis is the most severe manifestation of adrenal insufficiency, but it may share signs and symptoms with milder hypoadrenal states (table below). Most patients with adrenal insufficiency are initially incorrectly diagnosed due to the non-specificity of their signs and symptoms.
The spectrum of severity of adrenal insufficiency is shown below.

Clinical definition of adrenal crisis
- There is no universally accepted definition of an adrenal crisis *. The pragmatic definitions of adrenal crisis have generally been adopted as *
- Acute deterioration in health status associated with absolute hypotension (SBP <100 mm Hg) or relative hypotension (SBP ≥20 mm Hg lower than usual), and
- Marked resolution within 1-2 hours of IV steroid administration.
- Acute deterioration in health status associated with absolute hypotension (SBP <100 mm Hg) or relative hypotension (SBP ≥20 mm Hg lower than usual), and
- 👉Note that steroid may play both a diagnostic and therapeutic role. When in doubt, steroid should be promptly started. If hemodynamic instability persists, then alternative etiologies should be sought.
Cardinal manifestations of adrenal crisis
- Hemodynamic compromise (90%)
- Hypotension: Adrenal crisis is characterized by persistent hypotension and vasodilatory shock despite fluid resuscitation and vasopressors.
- Note that 50% of patients will not have hypotension prior to developing frank shock.
- Hypotension is most likely contributed by aldosterone defficiency (leading to hypovolemia) as well as cortisol deficiency resulting to vasoplegia.
- When hypotension ascribed to an adrenal crisis does not adequately respond to glucocorticoid administration, the coexistence of other illnesses associated with hypotension, such as sepsis, should be considered.
- Tachycardia
- Shock index (HR/SBP) >1
- Hypotension: Adrenal crisis is characterized by persistent hypotension and vasodilatory shock despite fluid resuscitation and vasopressors.
- Fever (66%)
- Nausea/vomiting (47%)
- Abdominal pain and tenderness (86%)
- 🛑Adrenal crisis can mimic abdominal sepsis, gasteroentritis or severe pancreatitis.
Less common signs and symptoms
- Impaired consciousness: delirium, obtundation, coma (42%)
- Features of the trigger of the crisis (e.g. infection, surgery, trauma).
Helpful clinical clues to the presence of chronic adrenal insufficiency
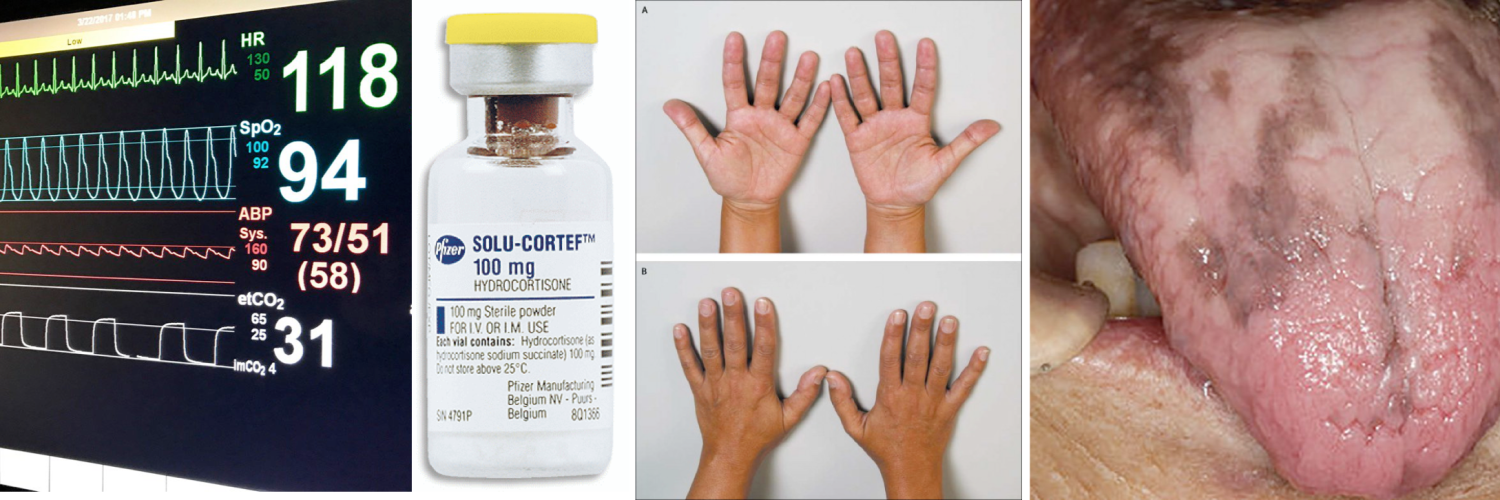
- Cutaneous hyperpigmentation
- This develops slowly and is seen only in primary adrenal insufficiency *.
- Low cortisol levels in primary adrenal insufficiency drives more ACTH secretion (HPA axis) in an attempt to stimulate the adrenal gland to make more cortisol (↓Cortisol, ↑ACTH levels)
- Notably, ACTH is a precursor in the production of melanin. The body therefore converts the excess ACTH into melanin, causing the hyperpigmented skin.
- Hyperpigmentation specially involves the sun-exposed area and flexural regions (palmar creases, knuckles, elbows). Patients often have patchy hyperpigmentation of oral mucosa (figure below)
- This develops slowly and is seen only in primary adrenal insufficiency *.
- History of chronic fatigue, anorexia, vomiting, postural dizziness, and weight loss.
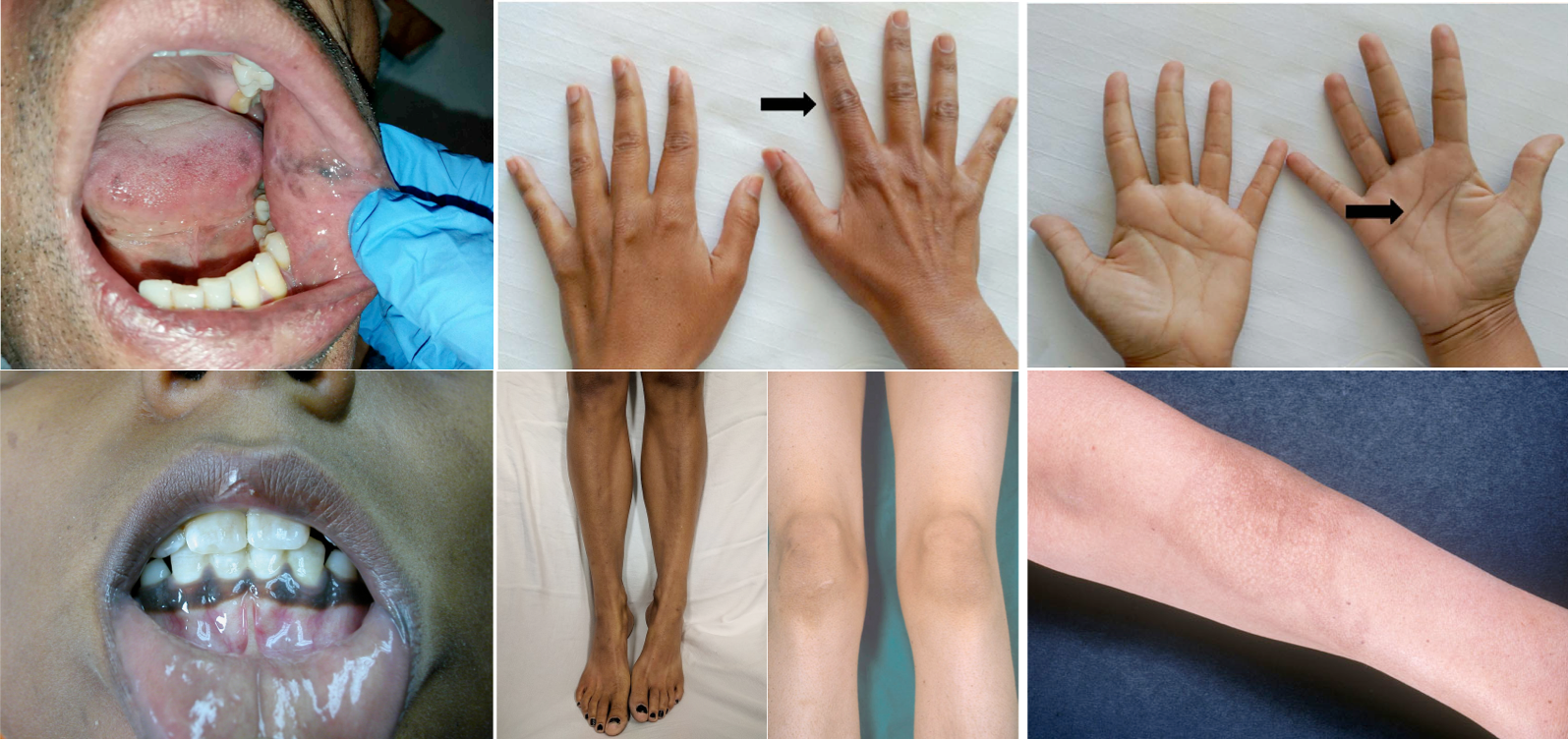
Hypopigmentation of Addison’s disease
Laboratory findings
The following laboratory abnormalities may serve as useful clues, but they cannot be used to exclude adrenal insufficiency, especially in critically ill patients with multiple coexisting electrolytic disorders.
- Cell count
- Physiologic stress normally stimulates cortisol production, which decreases the eosinophil and lymphocyte counts. In a patient under physiologic stress (e.g. clinical shock), adrenal insufficiency is suggested by either of the following:
- Normal or ↑eosinophil counts (e.g. an absolute eosinophil count >300 uL) *.
- Relative or absolute lymphocytosis.
- A relative lymphocytosis is suggested by finding that the neutrophil/lymphocyte ratio (NLR) is lower than would be expected for a critically ill patient (e.g. NLR < ~ 6).
- Physiologic stress normally stimulates cortisol production, which decreases the eosinophil and lymphocyte counts. In a patient under physiologic stress (e.g. clinical shock), adrenal insufficiency is suggested by either of the following:
- Metabolic / electrolytes
- Glucocorticoid deficiency → Hypoglycemia, hyponatremia, and rarely hypercalcemia
- Mineralocorticoid deficiency → Hyperkalemia, hyponatremia and non-anion gap metabolic acidosis
- Aldosterone defficiency only occurs in primary adrenal insufficiency (e.g. Addison’s disease), but not in secondary insufficiency (e.g. pituitary dysfunction).
Table. Clinical presentation of adrenal insufficiency and adrenal crisis.
| Characteristics | Adrenal insufficiency | Adrenal crisis |
| Symptoms | Fatigue, myalgia, arthralgia, postural dizziness Anorexia, nausea, vomiting, diarrhea Pain: abdominal, limb, back | Severe fatigue, delirium, obtundation, stupor Nausea, vomiting, diarrhea. Pain: abdominal, limb, back. |
| Signs | Fever Abdominal tenderness or guarding Orthostatic hypotension, syncope Hyperpigmentation of the skin (primary AI only) | Fever Abdominal tenderness or guarding 🛑End-organ hypoperfusion (shock state) Hyperpigmentation of the skin (primary AI only) |
| Labs | Serum level : ↓Glucose, ↓Na, ↑K, ↑Ca Neutropenia, eosinophilia, lymphocytosis | Serum level : ↓Glucose, ↓Na, ↑K, ↑Ca Neutropenia, eosinophilia, lymphocytosis |
Hint
| 🛑Acutely symptomatic disease without evidence of hemodynamic compromise or hypotension, should be classified as symptomatic adrenal insufficiency (a precursor of adrenal crisis, or an incipient adrenal crisis). This should be considered physiologically distinct from an adrenal crisis, and treatment with hydrocortisone and IV fluids may avert the development of an actual adrenal crisis. |
Differential diagnosis
- Shock: Any type of shock (esp. distributive shock)
- Hypovolemic: Dehydration, hemorrhagic shock
- Distributive: Septic shock, anaphylaxis, toxicologic
- Cardiogenic shock: ACS, acute valvular regurgitation, acute decompensated heart failure, dysrhythmia, drug toxicity (e.g. beta blocker, calcium channel blocker)
- Obstructive: Pulmonary embolism, tension pneumothorax, tamponade.
- Endocrinologic/metabolic
- Decompensated hypothyroidism
- Thyroid storms
- Hypoglycemia due to another etiology.
- GI
- Abdominal sepsis
- Severe pancreatitis
- Gastroenteritis
- Intestinal ischemia
- Psychiatric condition or anorexia nervosa
- Neurologic
- Meningitis
- CVA
Evaluation
Adrenal crisis is a life-threatening emergency, and treatment should not be delayed for any testing.
Universal panel
- CBC
- BMP
- Fingerstick glucose
- VBG
- Coagulation profile
- ECG
- Other tests depending on clinical presentation
- Sepsis work up: Cultures, CXR, inflammatory markers e.g. procalcitonin
- A low level of procalcitonin will lower the likelihhood of septic shock.
- Thyroid profile
- Neuroimaging (if neurological deficit present).
- CT or MRI of the head may be useful in identifying pituitary or hypothalamic disorders, including empty sella syndrome, hemorrhage, brain tumors, cerebral infiltrative, or infectious processes.
- Sepsis work up: Cultures, CXR, inflammatory markers e.g. procalcitonin
Diagnostic tests
- It’s important to determine whether or not the patient has underlying adrenal insufficiency, as this subsequent therapeutic implication.
- Random serum cortisol, ACTH, and renin level.
- Cortisol >20 ug/dL excludes adrenal insufficiency.
- Cortisol <20 ug/dL is nonspecific, requiring an ACTH stimulation test.
- <10 ug/dL in the context of physiologic stress strongly suggests adrenal insufficiency *.
- Cosyntropin test (ACTH stimulation test).
- Give 250 micrograms of IV ACTH (cosyntropin test).
- Check cortisol at baseline and 60 min after ACTH administration.
- If cortisol level doesn’t rise above >20-25 ug/dL, this confirms the diagnosis of adrenal insufficiency.
- This test can be done if the patient is receiving dexamethasone, but administration of other types of steroid renders the results meaningless.
- Random serum cortisol, ACTH, and renin level.
🛑Note that these diagnostic tests are not helpful in patients who are on chronic steroids as they likely have temporary suppression of the adrenal gland by exogenous steroids.
Management
Basic resuscitation
- Access: Establish IV access with a large-gauge needle and draw serum electrolytes and glucose. Do not wait for results to begin treatment!
- Consider sending cortisol and ACTH levels.
- For patients with possible infection, it may be reasonable to obtain cultures and start empiric antibiotic.
- Resuscitation
- The overall resuscitative strategy here is similar to a sepsis resuscitation (more on this here).
- Volume resuscitation is provided based on hemodynamic assessment (patients often depleted due to poor oral intake, vomiting, and renal sodium wasting).
- Early use of vasopressors should be considered, with subsequent weaning based on clinical parameters.
- Correct hypoglycemia with IV glucose as needed.
- Consider using D5NS for resuscitation if the patient has concomitant hypoglycemia.
- The overall resuscitative strategy here is similar to a sepsis resuscitation (more on this here).
Steroid
Choice of steroids
- Hydrocortisone is the agent of choice.
- Dexamethasone is preferred if rapid ACTH stimulation test is contemplated.
- Dexamethasone is useful because it does not interfere with measurement of cortisol levels (unlike most other steroids). Therefore it does not alter the result of cosyntropin test.
- If hydrocortisone is not available, other steroids such as dexamethasone, methylprednisolone, or prednisolone may be used (see the following table for comparison of systemic glucocorticoids) *.
- Note that mineralocorticoid replacement is not necessary acutely because it takes several days for its sodium-retaining effects to appear.
- Hydrocortisone has the most potent mineralocorticoid effects among the glucocorticoids. This is particularly beneficial in patients with primary adrenal insufficiency (since they have mineralocorticoid deficiency with more severe hyponatremia).
- In patient with severe hyperkalemia (serum K > 6 mEq/L) fludrocortisone at 0.2mg PO may be beneficial.
- Note that mineralocorticoid replacement is not necessary acutely because it takes several days for its sodium-retaining effects to appear.
Comparison of systemic glucocorticoids
| Equivalent dose (mg) | Anti-inflammatory activity relative to hydrocortisone | Mineralocorticoid effect relative to hydrocortisone | Duration of action (hours) | |
| Short acting Hydrocortisone (cortisol) | 20 | 1 | 1 | 8-12 |
| Intermediate acting Prednisone Prednisolone Methylprednisolone 🌟 Triamcinolone | 5 5 4 4 | 4 4 5 5 | 0.8 0.8 0.5 Zero | 12-36 12-36 12-36 12-36 |
| Long acting Dexamethasone Betamethasone | 0.75 0.6 | 30 30 | Zero Zero | 36-72 36-72 |
🛑Note that when given at replacement doses, triamcinolone, dexamethasone, and betamethasone have no clinically important mineralocorticoid activity.
🛑Prednisone or prednisolone given at anti-inflammatory doses ≥50 mg/d provide a mineralocorticoid effect that is equivalent to ~ 0.1 mg of fludrocortisone.
🌟Only methylprednisolone succinate formulation (Solu-Medrol) may be given IV. Methylprednisolone acetate (Depo-Medrol) is intended for IM or intra-articular use only.
Initial therapeutic regimen
- For patient with known adrenal insufficiency
- Hydrocortisone 100mg IV STAT as a loading dose, followed by 50 mg IV q6hr as a maintenance dose *.
- If hydrocortisone isn’t immediately available, another glucocorticoid may be used (steroid conversion calculator) e.g. methylprednisolone 40 mg IV daily.
- Once the patient has recovered, steroid may be tapered over 3 days to the patient’s baseline regimen.
- Hydrocortisone 100mg IV STAT as a loading dose, followed by 50 mg IV q6hr as a maintenance dose *.
- For patient with suspected adrenal insufficiency
- Do not delay steroid administration in suspected adrenal crisis.
- Dexamethasone 4-6 mg IV once can be preferred (if cosyntropin test is contemplated).
- May add fludrocortisone 0.1mg PO per day, as dexamethasone dose not have mineralocorticoid properties.
- Once the cortisol level is available, you can start standard treatment with hydrocortisone 50 mg IV q6hr; if they have adrenal insufficiency.
Re-evaluation
Following initial resuscitation and steroid administration, patients with adrenal crisis should rapidly improve with a marked resolution of hypotension within 1 hour and improvement in clinical symptoms over a period of 2 hours. Failure to improve suggest a missed diagnosis such as
- Occult infection
- Takotsubo cardiomyopathy (which can be associated with adrenal crisis). More on this here.
- Other diagnoses (DDx above)
Prevention
Maintenance regimen
- Common maintenance steroid regimens in adrenal insufficiency are as follows:
- Hydrocortisone: 20 mg qAM and 10 mg qHS, plus fludrocortisone 50-100 mcg/day.
- Prednisone: 5 mg qAM and 2.5 mg qHS, plus fludrocortisone 50-100 mcg/day.
Acute illness
- For patients with known adrenal insufficiency, steroid dose should be increased according to severity of illness or magnitude of stressor to prevent the development of adrenal crisis.
- Severe stress (e.g. major surgery, severe infection)
- Hydrocortisone 50 mg IV q6hr is given. This represents roughly eight times the normal physiologic output of steroid *.
- Moderate stress
- Severe stress (e.g. major surgery, severe infection)
Going further
References
1. PMID: 178180. Olefsky JM, Kimmerling G. Effects of glucocorticoids on carbohydrate metabolism. Am J Med Sci. 1976 Mar-Apr;271(2):202-10. doi: 10.1097/00000441-197603000-00009.
2. PMID: 23523565. Kuo T, Harris CA, Wang JC. Metabolic functions of glucocorticoid receptor in skeletal muscle. Mol Cell Endocrinol. 2013 Nov 5;380(1-2):79-88. doi: 10.1016/j.mce.2013.03.003. Epub 2013 Mar 21.
3.PMID: 26363354. Puar TH, Stikkelbroeck NM, Smans LC, Zelissen PM, Hermus AR. Adrenal Crisis: Still a Deadly Event in the 21st Century. Am J Med. 2016 Mar;129(3):339.e1-9. doi: 10.1016/j.amjmed.2015.08.021. Epub 2015 Sep 9.
4. PMID: 25419882. Hahner S, Spinnler C, Fassnacht M, Burger-Stritt S, Lang K, Milovanovic D, Beuschlein F, Willenberg HS, Quinkler M, Allolio B. High incidence of adrenal crisis in educated patients with chronic adrenal insufficiency: a prospective study. J Clin Endocrinol Metab. 2015 Feb;100(2):407-16. doi: 10.1210/jc.2014-3191. Epub 2014 Nov 24.
5. PMID: 31461595. Rushworth RL, Torpy DJ, Falhammar H. Adrenal Crisis. N Engl J Med. 2019 Aug 29;381(9):852-861. doi: 10.1056/NEJMra1807486.
6. PMID: 27995500. Rushworth RL, Torpy DJ, Falhammar H. Adrenal crises: perspectives and research directions. Endocrine. 2017 Feb;55(2):336-345. doi: 10.1007/s12020-016-1204-2. Epub 2016 Dec 19.
7. PMID: 26208266. Smans LC, Van der Valk ES, Hermus AR, Zelissen PM. Incidence of adrenal crisis in patients with adrenal insufficiency. Clin Endocrinol (Oxf). 2016 Jan;84(1):17-22. doi: 10.1111/cen.12865. Epub 2015 Aug 27.
8. PMID: 33762389. Hattersley R, Nana M, Lansdown AJ. Endocrine complications of immunotherapies: a review. Clin Med (Lond). 2021 Mar;21(2):e212-e222. doi: 10.7861/clinmed.2020-0827.
9. PMID: 31223468. Dineen R, Thompson CJ, Sherlock M. Adrenal crisis: prevention and management in adult patients. Ther Adv Endocrinol Metab. 2019 Jun 13;10:2042018819848218. doi: 10.1177/2042018819848218.
10. PMID: 10335792. Beishuizen A, Vermes I, Hylkema BS, Haanen C. Relative eosinophilia and functional adrenal insufficiency in critically ill patients. Lancet. 1999 May 15;353(9165):1675-6. doi: 10.1016/s0140-6736(99)01346-x.
11. PMID: 10335792. Beishuizen A, Vermes I, Hylkema BS, Haanen C. Relative eosinophilia and functional adrenal insufficiency in critically ill patients. Lancet. 1999 May 15;353(9165):1675-6. doi: 10.1016/s0140-6736(99)01346-x.
12.PMID: 28940011. Annane D, Pastores SM, Rochwerg B, Arlt W, Balk RA, Beishuizen A, Briegel J, Carcillo J, Christ-Crain M, Cooper MS, Marik PE, Umberto Meduri G, Olsen KM, Rodgers S, Russell JA, Van den Berghe G. Guidelines for the diagnosis and management of critical illness-related corticosteroid insufficiency (CIRCI) in critically ill patients (Part I): Society of Critical Care Medicine (SCCM) and European Society of Intensive Care Medicine (ESICM) 2017. Intensive Care Med. 2017 Dec;43(12):1751-1763. doi: 10.1007/s00134-017-4919-5. Epub 2017 Sep 21. Erratum in: Intensive Care Med. 2018 Feb 23.
13. PMID: 23506281. Becker DE. Basic and clinical pharmacology of glucocorticosteroids. Anesth Prog. 2013 Spring;60(1):25-31; quiz 32. doi: 10.2344/0003-3006-60.1.25.
14. PMID: 26760044. Bornstein SR, Allolio B, Arlt W, Barthel A, Don-Wauchope A, Hammer GD, Husebye ES, Merke DP, Murad MH, Stratakis CA, Torpy DJ. Diagnosis and Treatment of Primary Adrenal Insufficiency: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2016 Feb;101(2):364-89. doi: 10.1210/jc.2015-1710. Epub 2016 Jan 13.





Add comment