16 March 2023 by Shahriar Lahouti. Peer reviewed by Mojtaba Chardoli.
CONTENTS
- Preface
- Clinical presentation
- Epidemiology
- Causes
- Point-of-care ultrasound in cardiac arrest
- Management
- Appendix
- Media
- Going further
- References
Preface
Patients with pulseless electrical activity (PEA) account for up to 30% of cardiac arrest victims. The overall survival rate of patients with PEA is much worse than that of cardiac arrest patients with shockable rhythms. However PEA is a spectrum of disease and not all cases of PEA are in cardiac arrest state. Therefore lumping together all patients with pulseless electrical activity and treating them according to current ACLS guidelines is not appropriate. It is imperative for physicians to be able to distinguish pseudo-PEA from true PEA as the prognosis and management of these patients differ. A directed therapeutic approach to go along with aggressive resuscitation will provide patients with Pseudo PEA the best chance to survive.
Clinical presentation
Cardiac arrest
- It is the final common pathway of all life-threatening conditions and is referred to the sudden cessation of “mechanical” cardiac activity (pump failure), as confirmed by the absence of circulation.
- Presentation
- Unresponsiveness, absence of spontaneous breathing and no palpable pulses.
- Residual agonal breathing (i.e. deep, slow breaths at a rate of 1-2/min) or gasping may be present immediately after the collapse, which can delay the recognition of cardiac arrest.
- A brief generalized seizure or myoclonic jerks may be the initial manifestation of cardiac arrest, which can confuse the lay provider and delay the application of lifesaving BLS care.
- Rhythm: Generally, initial rhythm in cardiac arrest could be:
- Shockable rhythm (e.g. ventricular fibrillation “VF”; ploymorphic ventricular tachycardia “pMVT”, VT) or
- Non-shockable rhythm (asystole and pulseless electrical activity). The center of focus in this post is pulseless electrical activity (PEA).
- Unresponsiveness, absence of spontaneous breathing and no palpable pulses.
Impending to arrest
- Depending on the underlying cause and severity of the disease, these patients may deteriorate to full-blown cardiac arrest within a couple of minutes.
- Altered mental status
- Extreme tachycardia, terminal bradycardia (e.g. HR<30 bpm)
- Severe tachypnea, bradypenia
- Progressive hypotension (MAP<60), shock index >1
- Cold clammy extremities
- Rhythm: Any organized rhythm (even including VT).
- Rarely patients with VT may presents with extreme instability (but not in cardiac arrest).
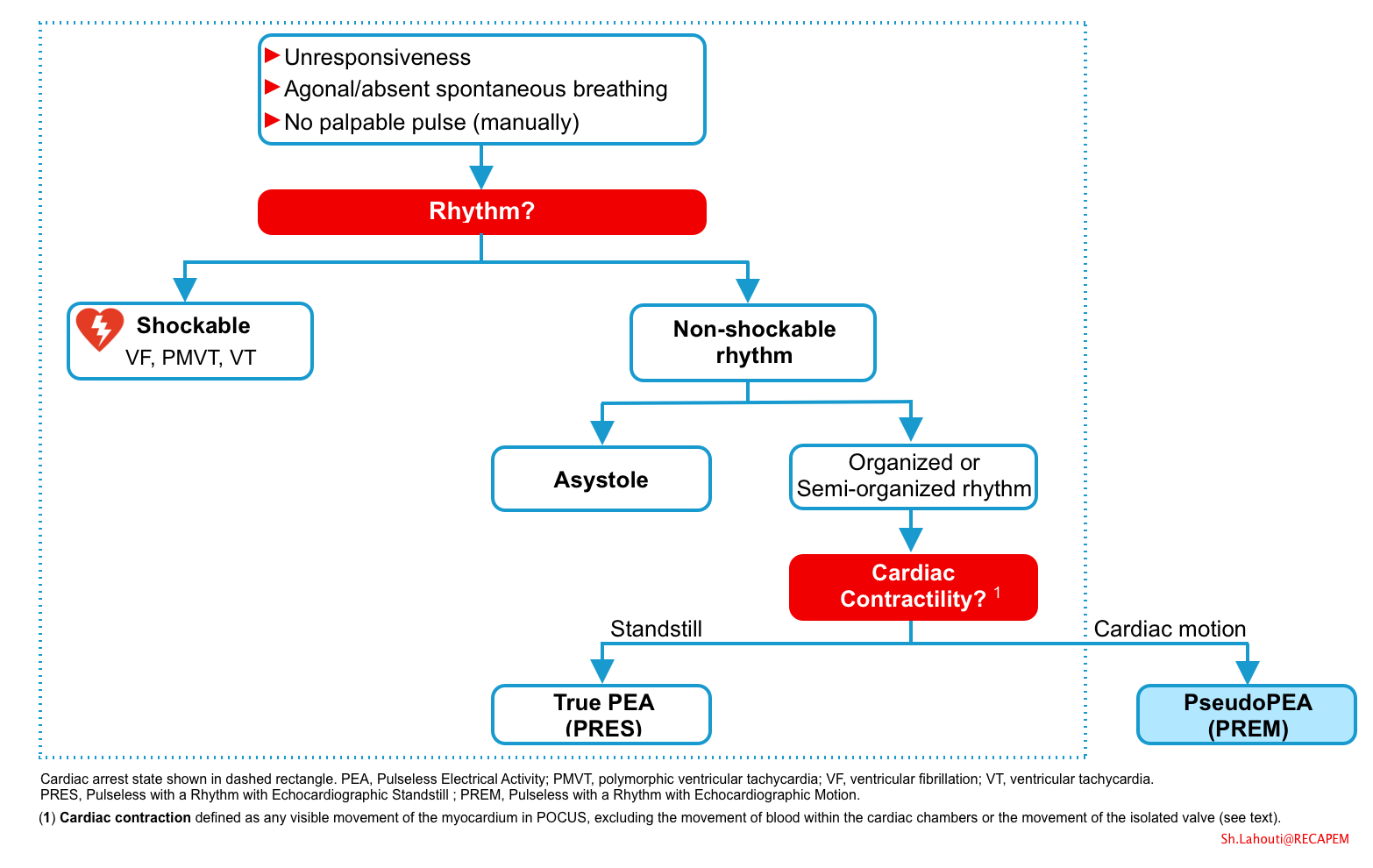
Pulseless electrical activity (PEA)
The diagnosis of PEA is based on a patient having an organized, non-shockable rhythm (e.g. bradycardia, normal sinus rhythm, etc.) and no palpable pulse. However not all PEA meet cardiac arrest by definition.
- In a study published in 1992 among patients with apparent PEA, a group of patients found to have aortic pulse pressure waveform using arterial line. Patients with pulse pressures were designated as having “pseudo-PEA”; and those without, as having “true PEA” *.
- Since this differentiation hinges on presence or absence of a pulse, it’s worth mentioning here that the palpation technique is poor at determining whether or not a patient has a pulse.
- In a study assessing the diagnostic accuracy of first responders of detecting a manual pulse in patient with and without a true pulse, the sensitivity for pulselessness approached 90%, however the specificity was only 55%. Only 15% of participants could accurately assess for the presence of a pulse within 10 seconds and only 2% identified pulselessness correctly within 10 sec *.
- Since this differentiation hinges on presence or absence of a pulse, it’s worth mentioning here that the palpation technique is poor at determining whether or not a patient has a pulse.
- Previously these two conditions were lumped together as they share presence of cardiac electrical activity on ECG monitor and undetectable pulses by manual technique *.
- Difference
Scott Weingart has suggested another lexicon because of ambiguity around definition of Pseudo PEA:
- PRES = Pulseless with a Rhythm with Echocardiographic Standstill = True PEA
- PREM = Pulseless with a Rhythm with Echocardiographic Motion = Pseudo-PEA
- Clinical significance: “Why differentiating True PEA from Pseudo PEA is clinically important”?
- Prognostication
- Pseudo PEA is associated with higher rate of ROSC and survival to hospital discharge. Understanding the patient’s prognosis will help guide the decision of terminating resuscitation efforts.
- For example, the identification of true PEA (cardiac standstill) could predict the failure of resuscitation and help inform the decision to terminate CPR, which allows for hospital resources to be utilized elsewhere.
- In contrast, the presence of cardiac activity in pseudo-PEA patients can encourage more aggressive resuscitation efforts and lead to interventions targeting specific ultrasound findings (e.g. empty hyperdynamic ventricle leading to fluid administration and search of cause of hypovolemia, or dilated right ventricle leading to the administration of thrombolytic therapy) *.
- Therapeutic value
- When pseudo-PEA is diagnosed, a directed therapeutic approach and aggressive resuscitation will improve the survival rate.
- Pseudo-PEA should be managed as profound shock state (see below).
- Pseudo PEA is associated with higher rate of ROSC and survival to hospital discharge. Understanding the patient’s prognosis will help guide the decision of terminating resuscitation efforts.
- Prognostication
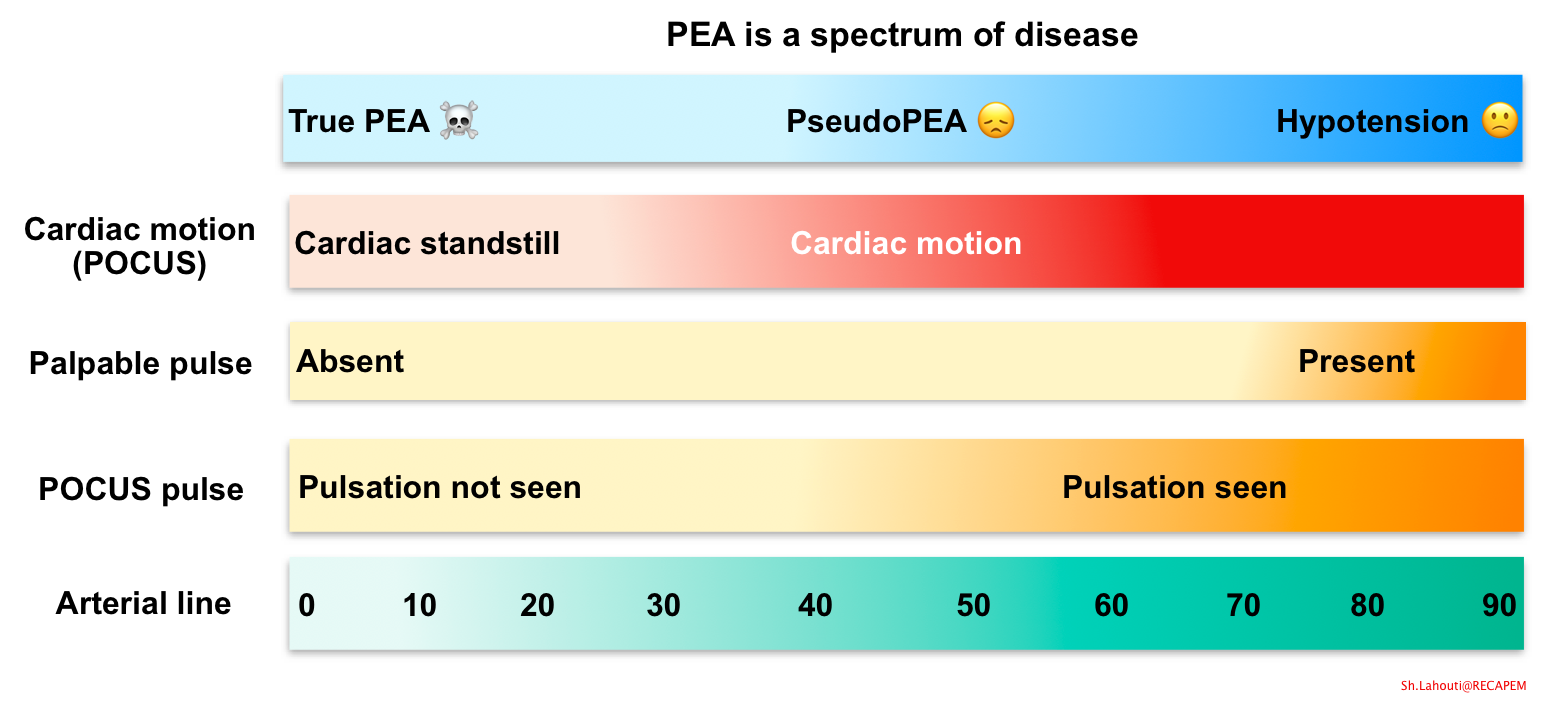
- Spectrum of PEA
- PEA is a heterogeneous state of circulatory collapse (shown below).
- True PEA: is a state of cardiac arrest with presence of electrical activity but no cardiac contraction. Therefore it is closer to the ‘dead patient’ end of the spectrum with worse prognosis.
- Pseudo-PEA: It can be considered as a state of cardiogenic shock state which does not adequately maintain perfusion pressure, ultimately resulting in a non-detectable pulse *.
- The patient looks like they might be dead, but there’s organized cardiac activity, a decent ETCO2, and there’s probably a blood pressure that can be detected with an arterial line.
- On the other end is just hypotension, in a patient who is very much alive, and you may or may not be able to palpate a pulse.
- PEA is a heterogeneous state of circulatory collapse (shown below).

- Evaluation. “How to determine where the patient is on the PEA spectrum”?
- Physiologic principles
- The physiologic backbone to understand the spectrum of full PEA arrest to ROSC encompasses 4 components as called by Weingart, “Chain of survival” from PEA arrest to ROSC.
- This denotes that for establishing “ROSC” from full PEA arrest, all 4 components are required.
- This approach provides a framework for integrating POCUS into cardiac arrest and can further guide the management.
- Chain of survival approach from PEA arrest to ROSC (figure below)
- Perfusable Rhythm.
- Is there a perfusion-capable rhythm including a number of organized cardiac rhythms that may be supraventricular in origin, sinus versus non sinus, or ventricular in origin such as accelerated idioventricular rhythm (virtually any rhythm except VF, pVT, VT).
- Note that sinusoidal pattern of sine wave in hyperkalemia results from widened QRS complexes with T wave in the absence of discernible P waves.
- Is there a perfusion-capable rhythm including a number of organized cardiac rhythms that may be supraventricular in origin, sinus versus non sinus, or ventricular in origin such as accelerated idioventricular rhythm (virtually any rhythm except VF, pVT, VT).
- Cardiac contraction.
- Is the rhythm capable to produce actual cardiac contractions.
- Is the rhythm capable to produce actual cardiac contractions.
- Generating a pulse (palpable or POCUS pulse).
- Are the cardiac contractions “sufficient enough” to generate a pulse?
- Sufficient blood pressure (perfusion pressure)
- Is the pulse generating a sufficient blood pressure enough to perfuse vital organs (heart and brain), so that the new condition does not degenerate into a worse situation (e.g. asystole or VF).
- Perfusable Rhythm.
- The physiologic backbone to understand the spectrum of full PEA arrest to ROSC encompasses 4 components as called by Weingart, “Chain of survival” from PEA arrest to ROSC.
- Physiologic principles

-
- Diagnostic tools
- The diagnostic modalities that can help us to determine where the patient is on the PEA spectrum (pseudo-PEA vs. true PEA) include:
- POCUS
- Is there sufficient cardiac contraction to generate a pulse?
- Is there a visible POCUS pulse?
- Arterial line (A-line)
- Invasive BP monitoring in the form of an A-line should be a top priority for the management of PEA.
- It is the ultimate diagnostic tool to determine where the patient is on the spectrum of PEA.
- End-tidal waveform capnography (more on this here)
- Oxygen saturation waveform
- POCUS
- The diagnostic modalities that can help us to determine where the patient is on the PEA spectrum (pseudo-PEA vs. true PEA) include:
- Patient’s status on the PEA spectrum
- Tools used to classify a patient’s status on the PEA disease spectrum is shown below.
- Diagnostic tools
Return of spontaneous circulation (ROSC)
Signs and symptoms of ROSC include:
- Spontaneous breathing, a palpable pulse, a measurable blood pressure, and/or moving.
- ROSC is also suggested if:
👉Persistent diastolic BP >40 mm Hg measured by A-line (best option).
👉Persistent oxygen saturation waveform (from a finger probe) plus persistent elevation of ETCO2 ≥30-40 mm Hg.
Epidemiology
In the setting of cardiac arrest, evidence over the past 2 decades suggests that the incidence of PEA has been increasing, whereas the occurrence of pulseless ventricular tachycardia and ventricular fibrillation has been decreasing *. However, many of these cases may be pseudo-PEA rather than true PEA, which would alter treatment strategies.
- Women are more likely to develop PEA as compared to the male population.
- The risk of PEA increases over the age of 70, especially in the female population.
Out-of-hospital cardiac arrest (OHCA)
- Incidence of PEA out-of-hospital: ~22% to 30% *.
- The overall prognosis is poor (2% and 5% )*.
- Although the prognosis for patients presenting with PEA is poor, the likelihood of survival for patients with pseudo-PEA is greater than those with true PEA *.
- Although the prognosis for patients presenting with PEA is poor, the likelihood of survival for patients with pseudo-PEA is greater than those with true PEA *.
In-hospital cardiac arrest (IHCA)
- Incidence of PEA in-hospital: ~35% to 40% of cardiac arrest events *.
- Overal prognosis is better than out-of-hospital PEA.
Causes
- The mechanisms of cardiorespiratory arrest differ between out and in-hospital.
- In-hospital cardiac arrest is frequently the result of clinical deterioration, which typically occurs gradually for hours or days in the context of a critically ill patient.
- The reversible causes of PEA arrest and their associated diagnostic tools and clues are mentioned in following table *.
| Condition Examples & risk factors | POCUS signs and symptoms | Considerations |
| Secondary dysrhythmias – IHD e.g. AMI – Cardiomyopathies | AMI: -LV hypokinetic or akinetic -RWMA -Severe MR -Free wall rupture | -Bradydysrhythmias are more commonly seen with non-ischemic cardiomyopathies. – VF and PMVT (without long QT) are more commonly due to acute MI. – Monomorphic VT often happen in patient with structural heart disease, e.g. previous MI, HF. ECG signs of AMI, 👉here. |
| Tamponade -Type A dissection – Cancer – Uremia | Arrest signs: -Pericardial effusion -Plethoric IVC Peri-arrest signs -Pericardial effusion -Plethoric IVC -Furiously contracting chamber -Systolic collapse of the right atrium -Collapse of the right ventricle in diastole | Echo signs of aortic dissection 👉here |
| Tension pneumothorax – CPR-related – COPD – Spontaneous | Arrest signs – Absence lung sliding (during artificial ventilation) Peri-arrest signs – Absence lung sliding – Absence B lines – Absence pulmonary pulse – Lung point | |
| Pulmonary embolism – Cancer – Immobility – Pregnancy/OCP | Arrest signs – Right heart thrombus – Right ventricular dilation – D sign of left ventricle – Deep vein thrombosis Peri-arrest signs – Pulmonary Hypertension – Hyperdynamic LV – Deep vein thrombosis – Paradoxical septal systolic movement – Septal flattening in diastole – McConnell sign – D sign of left ventricle | More on POCUS and ECG signs 👉 here. |
| Toxins such as – BBs – CCBs – TCA – Opioids | – BB & CCB intox: ECG and POCUS signs 👉here – TCA intoxication 👉 here | |
| Hypovolemia – Hemorrhage – Dehydration – Septic shock | Arrest signs – Obliteration of ventricles – Inferior vena cava diameter < 2 cm Peri-arrest signs – Hyperdynamic – Obliteration of ventricles – Kiss sign – Empty tank IVC < 2 cm collapse > 50% during inspiration) | Try to find the site of hemorrhage – Rupttured AAA: Enlarged abdominal aorta, abdominal free fluid – Ruptured ectopic pregnancy: Intra-abdominal free fluid – GI bleeding: Risk factors, melena, hematochezia, hematemesis |
| Hyperkalemia – CKD – Medications | ECG signs of ↑K👉 here | |
| Hypoxia – COPD – Drowning – OSA | ||
| Hydrogen ions – DKA – AKA – Metabolic acidosis |
Approach to differential diagnosis
- 5 H’s and T’s: This mnemonic is currently suggested in ACLS guidelines as teaching tool for underlying causes of PEA arrest *. This stands for a comprehensive list as it encompasses most causes of PEA arrest.
- Caveats
- It is too complex, difficult to recall during resuscitation.
- It does not provide guidance to the most effective initial interventions.
- Caveats
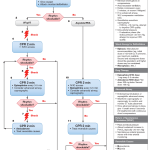
- Wide vs. narrow complex
- Littman in 2014 published an approach to PEA, dividing up patients into those with a wide QRS complex and those with a narrow QRS complex with their most likely causes and using POCUS to help differentiate the anatomical underlying causes of the PEA arrest (figure below) *.
- This approach was made in an effort to improve recall of the “5 Hs and Ts” underlying causes of PEA arrest.
- The idea was that narrow-complex PEA is generally due to mechanical problems caused by RV inflow or outflow obstruction and wide-complex PEA is typically due to metabolic problems. MI can have either wide or narrow complex QRS.
- However recent studies suggest that there are no unique ECG patterns associated with any particular underlying cause of PEA *. Most experts disagree on the usefulness of this approach.
- Littman in 2014 published an approach to PEA, dividing up patients into those with a wide QRS complex and those with a narrow QRS complex with their most likely causes and using POCUS to help differentiate the anatomical underlying causes of the PEA arrest (figure below) *.
- Dynamic approach
- The advanced management of PEA arrest integrates POCUS during cardiac arrest and information obtained from EMS or collateral. This enable us to identify and or empirically treat the most common causes of PEA arrest (5H’s and 5T’s).
- POCUS in cardiac arrest: Allows early diagnosis of pulmonary embolism, tamponade, tension pneumothorax, hypovolemia.
- RUSH exam: further identify the underlying causes e.g. AAA rupture
- Hypoxia is empirically treated following standard recommendation for advanced airway management.
- 🧪Bedside point-of-care testing is performed for all patients in cardiac arrest.
- This may be useful to identify specific metabolic etiologies of cardiac arrest (e.g. severe acidemia, electrolyte disturbances such as hyperkalemia, hypoglycemia, severe anemia).
- The advanced management of PEA arrest integrates POCUS during cardiac arrest and information obtained from EMS or collateral. This enable us to identify and or empirically treat the most common causes of PEA arrest (5H’s and 5T’s).
Point-of-care-ultrasound in cardiac arrest (POCUS-CA)
- POCUS is potentially helpful in diagnosis of reversible causes of PEA arrest (above), allowing for early decision making and could have a considerable impact on survival *.
- The third study in the Sonography in Hypotension and Cardiac Arrest in the Emergency Department (SHoC-ED) series examined potential relationships between POCUS use and the length of resuscitation, the frequency of interventions, and clinical outcomes during cardiac arrest.
- The ED cardiac arrest patients with cardiac activity on POCUS received longer resuscitation with higher rates of intervention as compared to those with negative findings or when no POCUS was performed.
- Patients with cardiac activity on POCUS had improved clinical outcomes as compared with patients not receiving POCUS, and patients with no activity on POCUS.
- The third study in the Sonography in Hypotension and Cardiac Arrest in the Emergency Department (SHoC-ED) series examined potential relationships between POCUS use and the length of resuscitation, the frequency of interventions, and clinical outcomes during cardiac arrest.
- Current literature support the use of POCUS in diagnosis of reversible causes of PEA and guiding the treatment. However synchronized use of POCUS in cardiac arrest needs to be further studies.
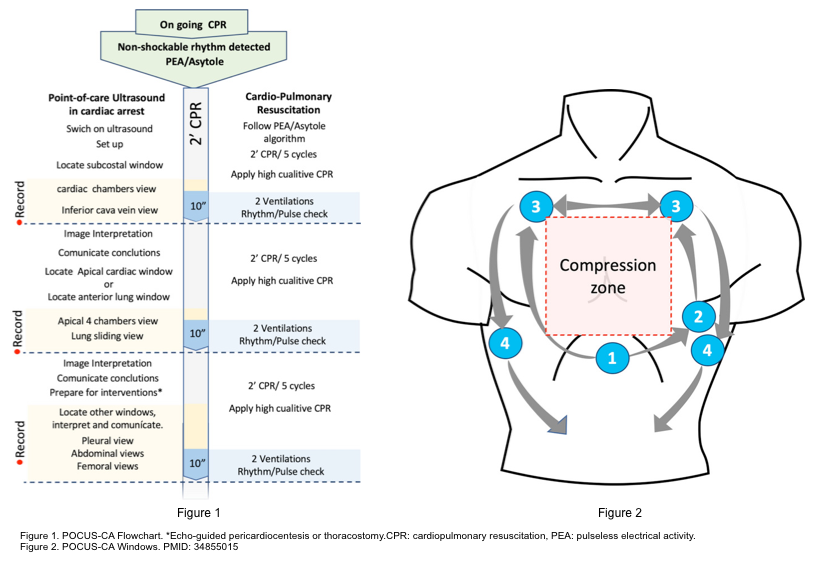
- There are several echocardiography protocols in a patient with cardiac arrest (below panel).
-The Focus-Assessed transthoracic echocardiography (FATE) protocol was performed on 210 patients in 20 ICU and evaluated the subcostal, apical, parasternal, and pleural windows.
-The Focused echocardiographic evaluation in resuscitation management (FEER) protocol (FEEL) evaluated the subcostal, parasternal and apical window, which showed an improvement in the results.
-The Cardiac arrest ultra-sound exam (CAUSE) protocol evaluates the subcostal or parasternal or apical window added to the evaluation of the pulmonary window and has been widely disseminated in clinical practice.
-The PEA protocol was proposed a simplified US protocol for non-shockable rhythms, called “PEA protocol” to remember the applications of the study (CA in Pulseless Electrical Activity, PEA) and the sequence of ultrasound scan includes Pulmonary, Epigastric, and Abdominal evaluation.
-The Sequential Echographic Scanning Assessing Mechanism Or Origin of Severe Shock of Indistinct Cause (SESAME) protocol, emphasized that a holistic approach, where ultrasound can be the modern stethoscope to complete the physiological examination of critically unstable patients should be employed.
-A consensus of an expert panel was published in 2017 and recommended the cardiac arrest protocol (CORE) at the time of POCUS and in specific clinical questions. These protocols represent a consensus on the prioritization of the examination for these critically ill patients, based on the probability of detecting the underlying pathology and the perceived impact on the patient’s treatment.
- POCUS in cardiac arrest should be performed by the most expert ultrasound clinician to interpret the findings correctly.
- The evaluation should not interfere with the development of resuscitation.
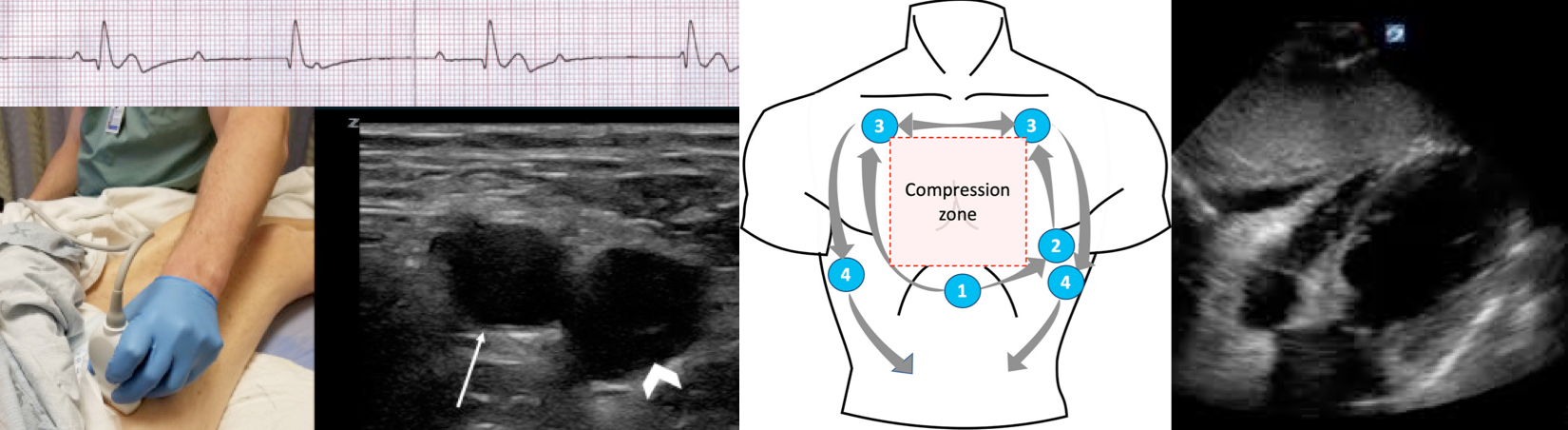
- Which POCUS window is recommended to be obtained first in cardiac arrest?
- Based on the most recent review article, a subxiphoid view for the initial assessment of cardiac contractility is recommended. Other views are used later in the resuscitation to rule out causes of PEA *.
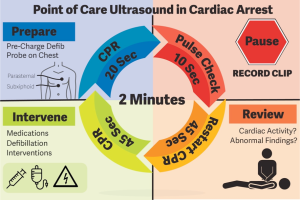
- How to prevent long pauses in chest compressions using POCUS?
- POCUS provider should locate the initial window (subxiphoid) during the chest compression. Generating an image at least 10 sec prior to pausing chest compressions and as chest compressions are stopped hit the video record button * (below figure).
- During the pause for rhythm & pulse check:
- Hit the video record button. Record the images for 5-10 sec maximum during the chest compression pause.
- Generally the evaluation of the images is not carried out during direct visualization, since this may delay the restart of the compressions *.
- Skilled ultrasound provider may be able to interpret images on real time, however this should not delay the CPR cycle.
- Hit the video record button. Record the images for 5-10 sec maximum during the chest compression pause.
- Start the next cycle of chest compression immediately.
- Evaluate the obtained recording (to determine findings) only when the compressions are restarted.
👉Note that other windows (described in the e-FAST protocol ) to evaluate pneumothorax, pleural effusion, hemothorax, and free intra-abdominal fluid does not require synchronization with the CPR (can be obtained during chest compression) *.
Cardiac window
Goal
During the first pause in CPR, subxiphoid window should be obtained to answer two major questions.
- Is there actual cardiac contraction? (Differentiating true PEA vs. pseudo-PEA)
- Actual cardiac contraction is defined as any visible movement of the myocardium, excluding the movement of blood within the cardiac chambers or the movement of the isolated valve (such as mitral valve).
- Is there pericardial effusion (suggestive for tamponade)?
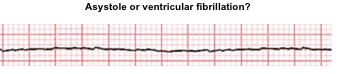
👉Cardiac POCUS also allows distinguishing VF from asystole (below).
Pt brought in for cardiac arrest with no pulse…is this…
— Salim R. Rezaie, MD (@srrezaie) July 7, 2022
A: PRES (Pulseless with a Rhythm and Echocardiographic Standstill)
OR
B: PREM (Pulseless with a Rhythm and Echocardiographic Motion)#FOAMed #FOAMcc #OHCA #PseudoPEA #PEA pic.twitter.com/Okc8KipuHC
Ventricular fibrillation vs. asystole
Fine ventricular fibrillation is often difficult to be distinguished from asystole. This differentiation has significant therapeutic implication as
- VF in cardiac arrest: should be electrically defibrillated, while
- Asystole: CPR should be continued based on ACLS guidelines {chest compression, epinephrine, identifying the cause(s)}
POCUS can be helpful in diagnosis of VF. The following finding are suggestive for VF:
Left: VF. Disorganized cardiac twitching and mitral valve quivering.
Right: Asystole
Another example of VF.
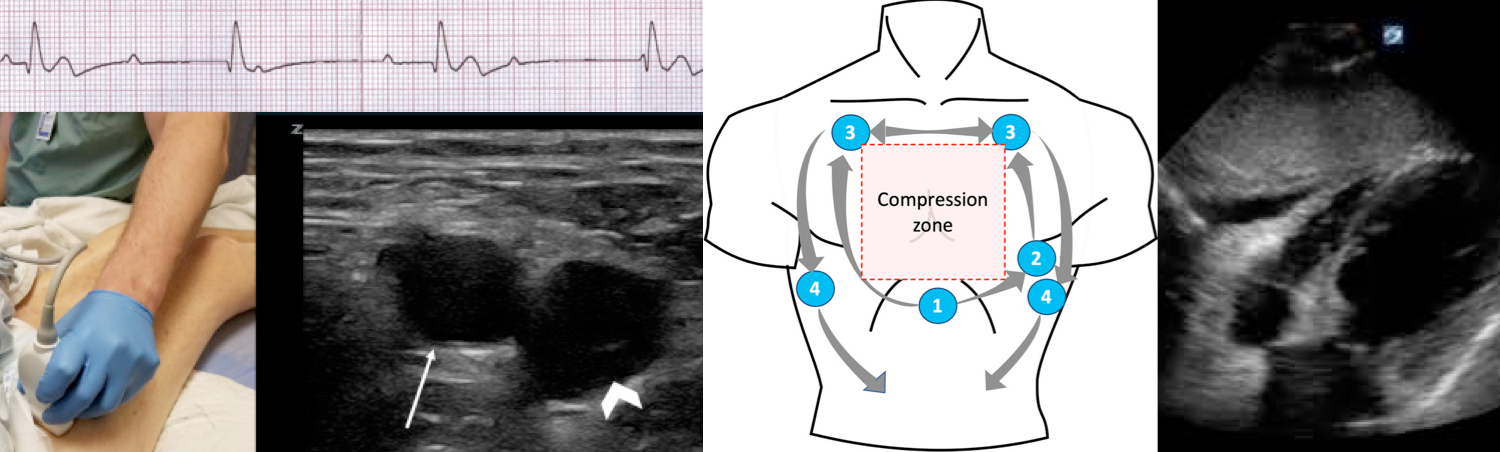
POCUS pulse
During CPR pulse checks must be rapid and accurate. Several studies have shown that health care providers have poor accuracy for detection of central pulses by palpation.
Significance
- An incorrect determination of the presence or absence of a pulse can have significant adverse consequences for patients.
- For example, the incorrect determination that a pulse is present may lead to delays in CPR for patients in cardiac arrest.
- Conversely, the incorrect determination of an absent pulse may lead to the unnecessary administration of arrest doses of epinephrine.
- POCUS pulse check has been shown a valuable adjunct that can help to change management for critically ill patients *.
Technique
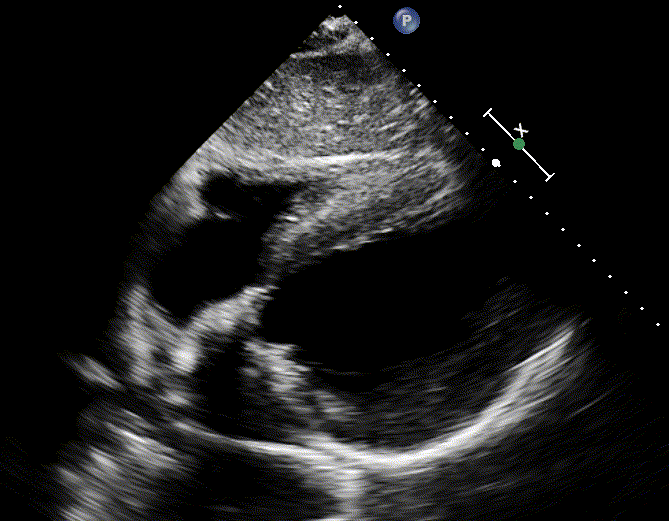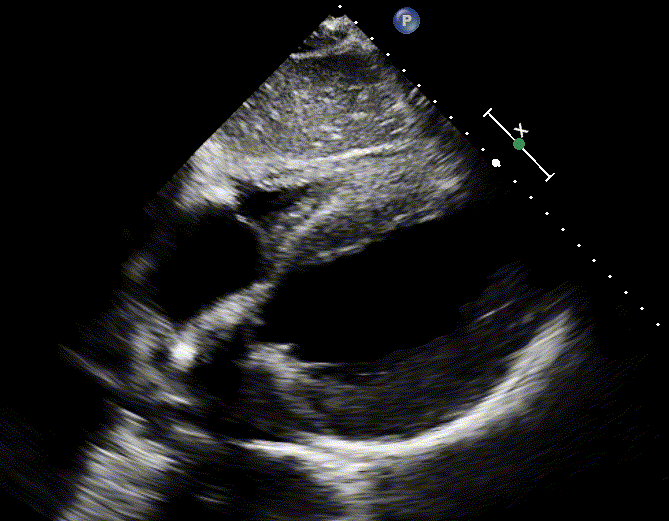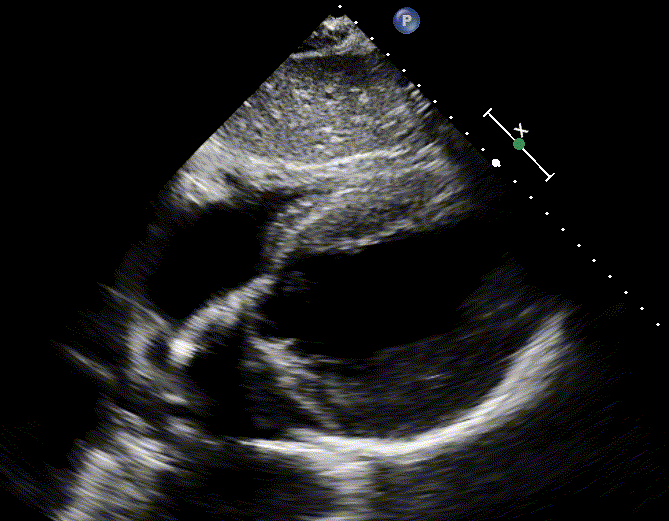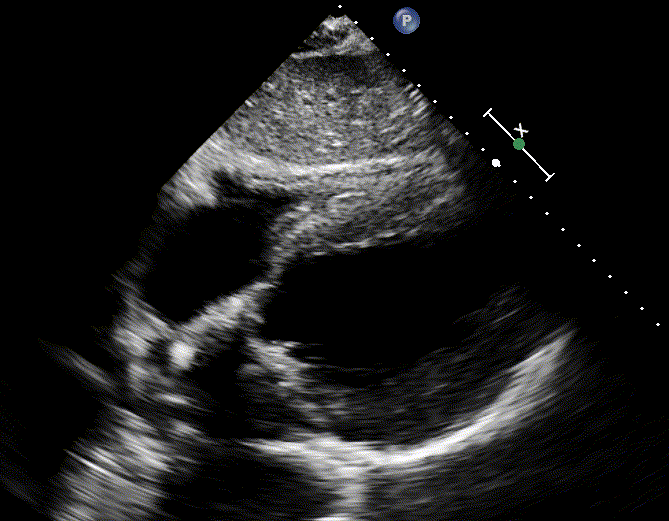
- Site: Either carotid or femoral artery can be assessed, depending on access to the patient.
- Transducer: Linear probe
- Position: The transducer should be placed in a transverse orientation, with the indicator pointing toward the patient’s right side.
- The carotid artery generally lies medial to the internal jugular vein and the common femoral artery lies lateral to the common femoral vein.
- Use 2D views, the pulsation of the artery should be visible. Gentle compression of the artery with the transducer will reveal whether the artery collapses, and whether pulsations are present *.
Two good reasons to #POCUS the femoral region during cardiac arrest:
— Trent She, MD (@trentshemd) January 18, 2021
1. Visualizing a femoral pulse, signaling ROSC.
2. Visualizing a DVT suggesting acute PE was the initial cause of arrest.
Both can be seen in this clip.#sonofirst #FOAMed #FOAMus #FOAMcc #MedEd #MedTwitter pic.twitter.com/QM2y9CuNZQ
Helpful diagnostic clues
By enlarge, the initial subxiphoid window in PEA provides useful diagnostic information
- Hypovolemia
- A hyperdynamic LV, underfilled RV in the absence of pericardial effusion suggest hypovolemia (media below)
- This is supported by collapsible IVC
- 👉Keep in mind that in severe hypovolemic shock, despite vigorous cardiac contractions, POCUS pulse may not be seen.
- Look for source of volume loss such as hemothorax, ruptured AAA, GI loss.
- Cardiac tamponade
- Pericardial effusion: However presence of pericardial effusion does not make a diagnosis of cardiac tamponade.
- Diagnosis of tamponade requires additional findings including right atrial and or right ventricular diastolic collapse.
- However, these signs are not seen in the patient with arrest, but they can occur prior to the arrest.
- Hyperdynamic cardiac chambers (furiously contracting chambers) is present in patients impending to arrest.
- This exaggerated cardiac movement making it challenging to assess for diastolic collapse of RA and RV.
- Pulmonary embolism
- A hyperdynamic, underfilled LV, RV enlragement and more specifically clot-in-transit (CIT) suggest pulmonary embolism (media below)
- An engorged, non-collapsible IVC support this.
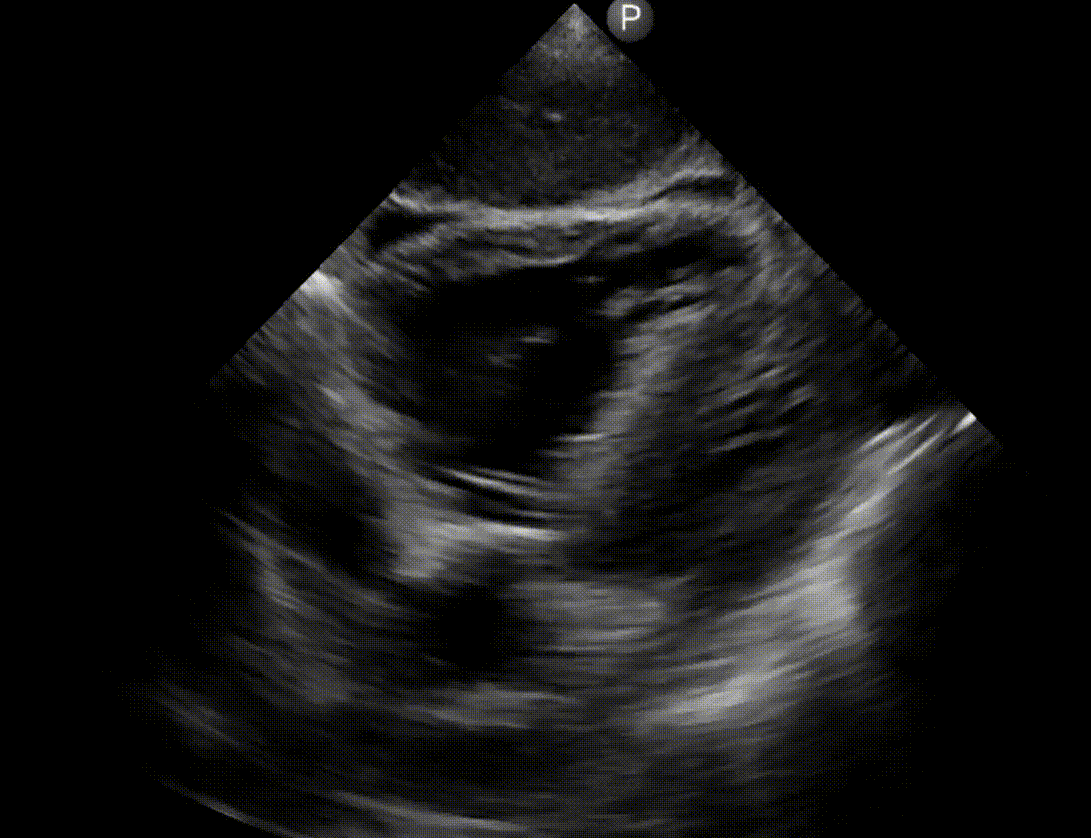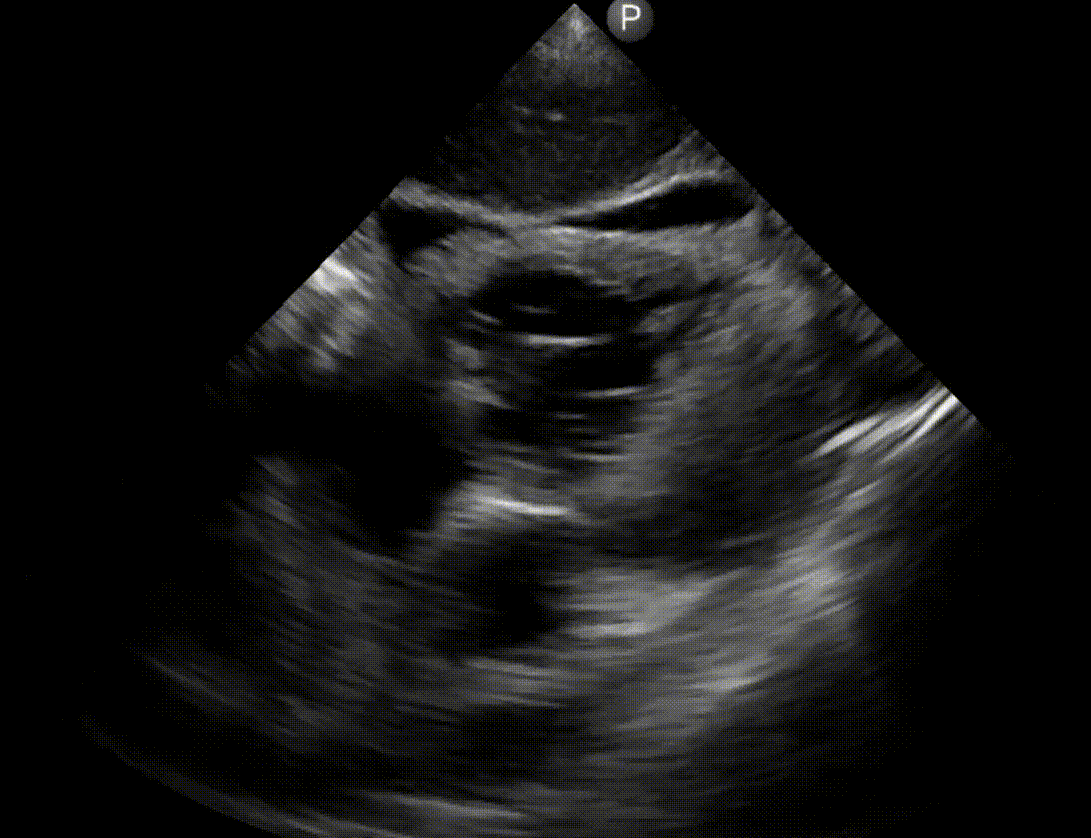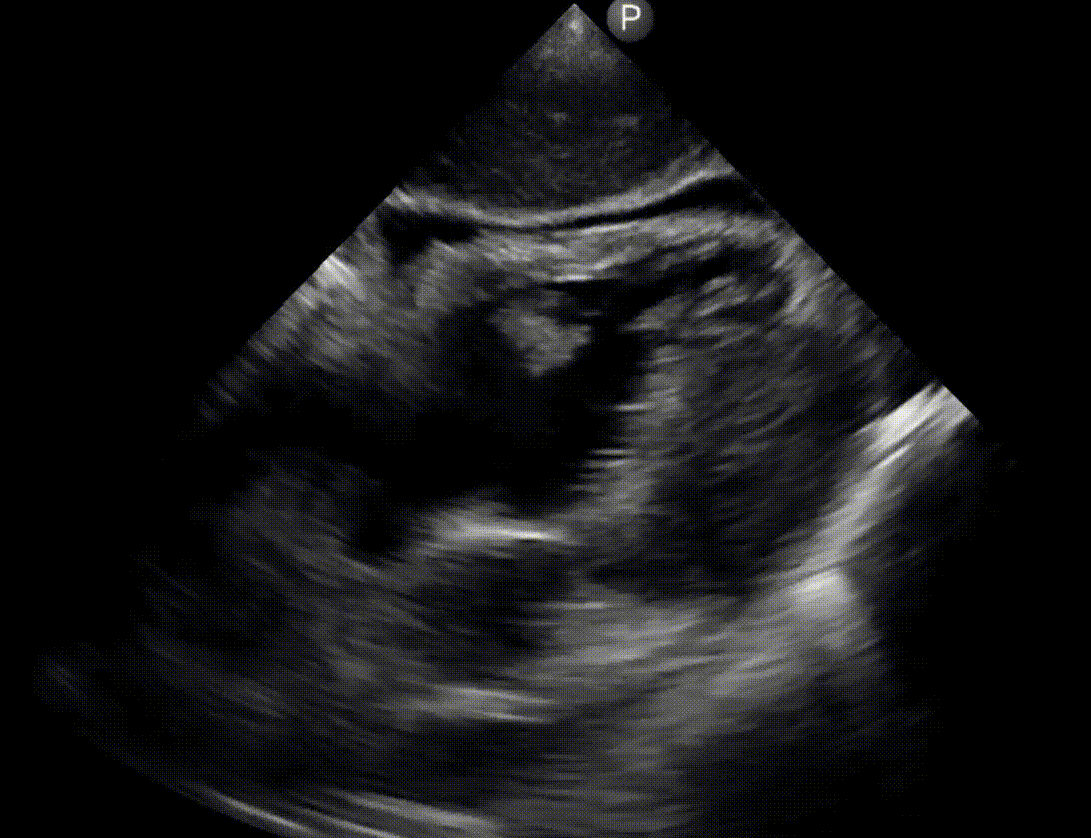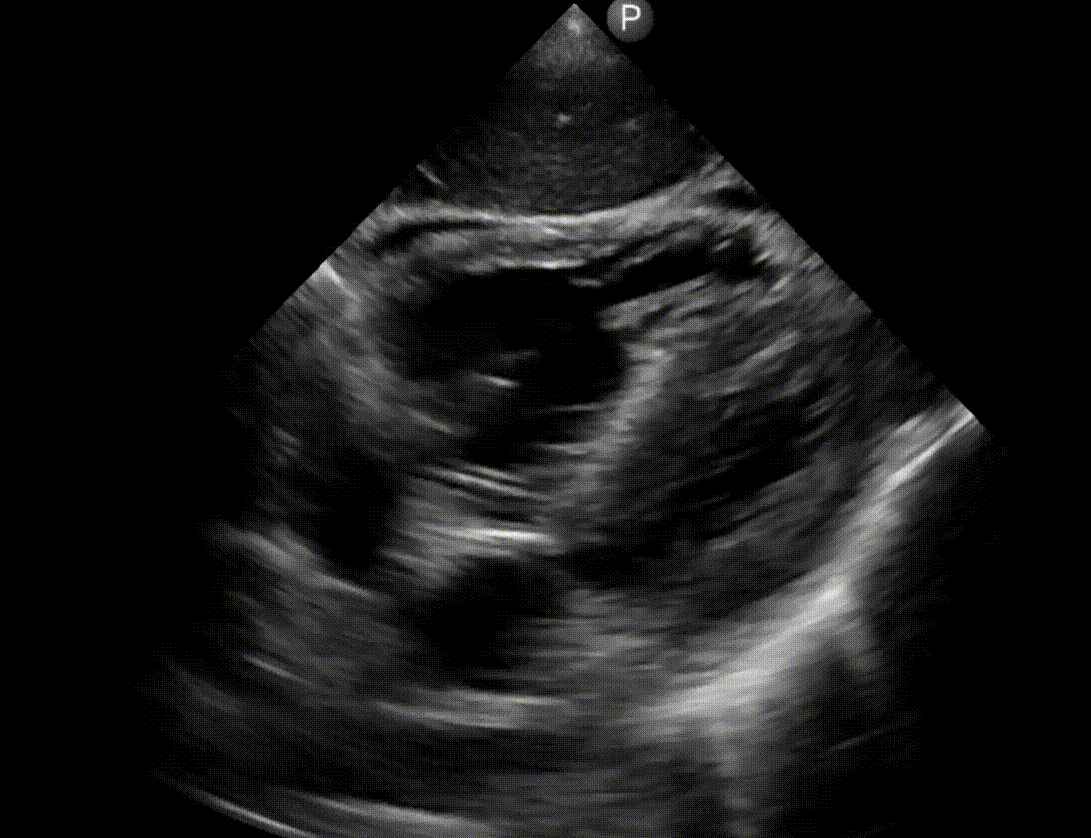
- LV myocardial infarction
- A hypokinetic LV, RWMA suggest AMI (media below).
- Such patients often develop cardiogenic pulmonary edema. Presence of diffuse bilateral B-lines is supporitve.
Management
ACLS guidelines
The resuscitation of adult patient with cardiac arrest should proceed as per ACLS guidelines (appendix). However, the ‘one size fits all’ approach of current ALS algorithms may not be appropriate for patients with PEA.
-
- High quality CPR
- Considering (early) advanced airway management
- Early administration of standard dose of epinephrine
- Identifying and treating the reversible causes (5 H’s and T’s)
Caveats
- “One-size-fit-all” approach
- The current ACLS approach to management of PEA arrest is ambiguous. Given the heterogeneity of PEA, it is critical to identify patients with pseudo-PEA since the treatment is different from those with true PEA as explained below.
- Epinephrine
- Current AHA and ILCOR guidelines for the management of PEA recommend giving 1mg of epinephrine q3-5 minutes. However, it is unclear whether this is really the best vasopressor choice for pseudo-PEA.
- This recommendation is based on interpretation of moderate-quality evidence from randomized data. The writing group prioritized the consistent improvements in ROSC, short-term and long-term survival, while acknowledging the continuing uncertainty about overall impact on neurologic outcome.
- The beta-adrenergic effects of epinephrine (↑cardiac rate, ↑contractility, and automaticity) are potentially harmful as they may lead to dysrhythmias and ↑myocardial oxygen demand and ischemia.
- ⚠️These potential deleterious effects of epinephrine are likely exaggerated in patients with “pseudo-PEA”.
- Current AHA and ILCOR guidelines for the management of PEA recommend giving 1mg of epinephrine q3-5 minutes. However, it is unclear whether this is really the best vasopressor choice for pseudo-PEA.
- Vasopressor
- Pseudo-PEA should be considered as a profound shock state. Delay in diagnosis and administration of appropriate vasopressor (to maintain perfusion pressure of vital organs) results in degeneration to a worse condition such as VF or asystole.
- Asynchronous chest compression
- Repeated cardiac chest compressions asynchronous with the patient’s cardiac cycle could raise the mean intrathoracic pressure, resulting in a reduction in cardiac filling and ultimately in cardiac output *.
- It has been shown that in pseudo-PEA, the synchronization of external chest compressions with cardiac systole resulted in a higher peak aortic pressure compared and higher coronary perfusion pressure which can lead to improving the low-flow state of those with pseudo-PEA * (This study was performed in a porcine model).
- Epinephrine
- The current ACLS approach to management of PEA arrest is ambiguous. Given the heterogeneity of PEA, it is critical to identify patients with pseudo-PEA since the treatment is different from those with true PEA as explained below.
- Diagnostic pitfalls
- Despite emphasizing the need to identify and correct reversible etiologies (5H’s and T’s), current ACLS guidelines does not provide a pragmatic approach.
- Recalling this broad list of differentials is a difficult task that is not always possible in the often hectic early minutes of cardiac arrest resuscitation.
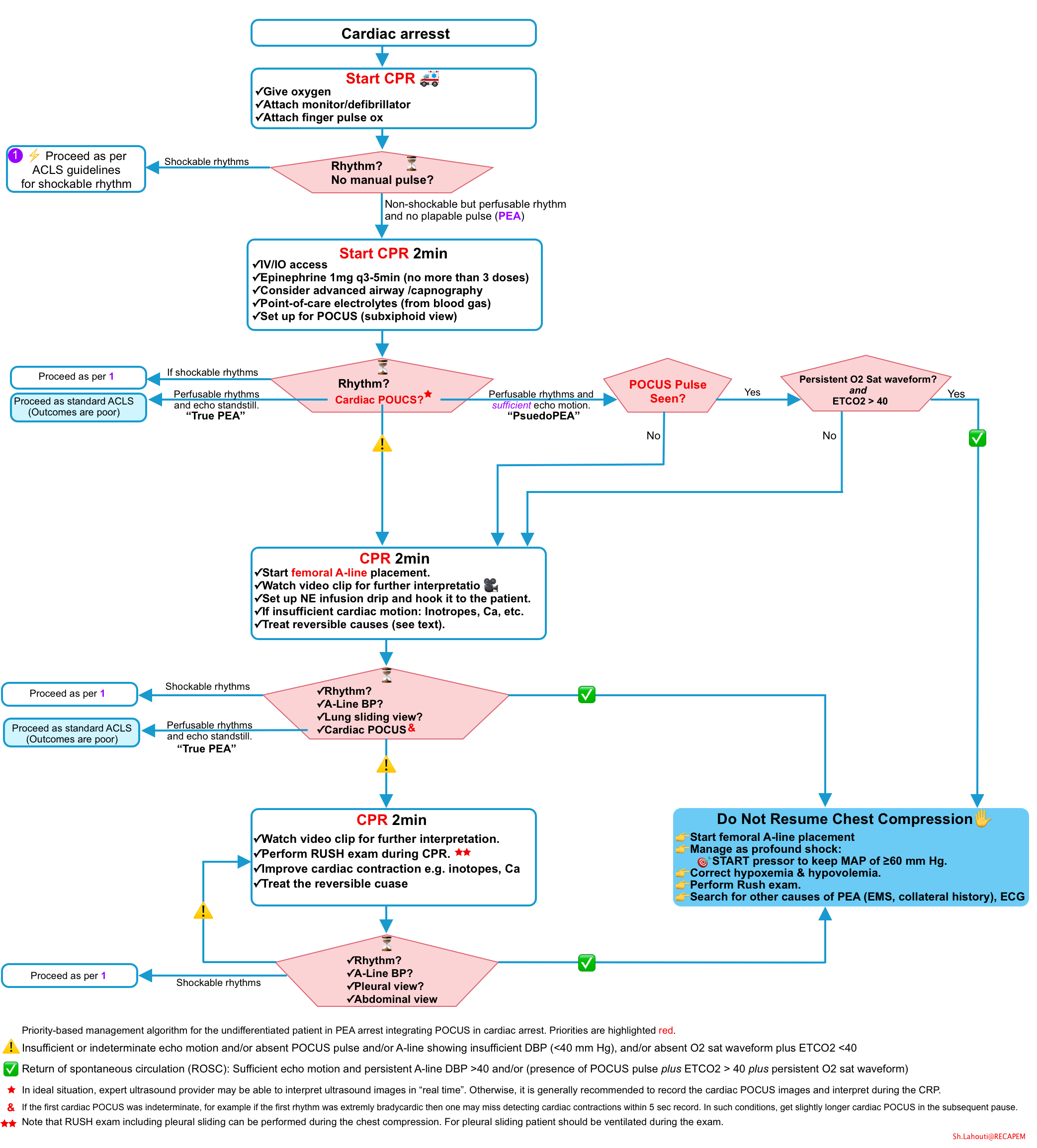
Advanced management of PEA arrest
Objectives
- In addition to priority given in resuscitation of PEA (above), the following goals should be considered.
- Early identification of pseudo-PEA while avoiding any delays in chest compressions.
- Timely diagnosis and directed treatment of underlying cause of PEA arrest.
The advanced management of PEA discussed here, is based on latest evidence of POCUS-CA *,* and expert opinions. The principles of this diagnostic and therapeutic approach is reviewed below.
Principles of approach to advanced PEA management
- Chain of survival approach from PEA arrest to ROSC is explained above. This provides a useful framework to guide an individualized dynamic management of PEA spectrum of disease.
- Chain of survival: Perfusable rhythm ➡️Sufficient cardiac contraction ➡️Pulse generation ➡️Sufficient BP
- Sufficient DBP to perfuse vital organs (A-line DBP > 40 mm Hg) is best indicator of ROSC.
- For sure POCUS pulse as well as cardiac contractions are sufficient, if A-line shows DBP >40.
- Presence of sufficient cardiac contractions is essential for detection of a pulse. However it is possible to have vigorous contractions with undetectable pulses. This condition is commonly seen in profound hypovolemic shock.
- Sufficient DBP to perfuse vital organs (A-line DBP > 40 mm Hg) is best indicator of ROSC.
- Diagnostic tools to classify a patient’s status on the PEA disease spectrum
- Rhythm monitoring: Is the rhythm perfusion-capable or not?
- Cardiac POCUS: Is there organized cardiac contraction? Are these contractions sufficient enough to generate a pulse?
- Pulse check (Palpation vs. POCUS)
- While POCUS pulse checks do not completely replace manual pulse checks, it is a useful adjunct to determine where the patient is on the PEA spectrum.
- The idea is that the cardiac output may be present but low enough that we miss detecting a pulse by manual check.
- POCUS pulse is more sensitive (i.e. pulsation of carotid or femoral artery can be seen even if the cardiac output is low).
- However seeing palpation on POCUS does not necessarily imply sufficient arterial BP.
- Evaluation of perfusion pressure (BP)
- Arterial line (A-line) BP measurement
- Is the optimal method to assess sufficiency of BP and ROSC.
- Oxygen saturation waveform and ETCO2
- Can be used to assess for sufficiency of perfusion pressure (BP) if A-line is not available.
- A persistent oxygen sat waveform (during CPR pause) and ETCO2 > 40 highly suggest adequacy of perfusion pressure.
- Arterial line (A-line) BP measurement
- Chain of survival: Perfusable rhythm ➡️Sufficient cardiac contraction ➡️Pulse generation ➡️Sufficient BP
- Diagnosis the underlying cause of PEA arrest
- History obtained from EMS or collateral.
- POCUS-CA
- POCUS-CA is a protocol to consider in management of PEA arrest.
- It employs POCUS synchronized with CPR using a rational “holistic” approach. It always starts with cardiac imaging (subxiphoid window) and proceed further as described earlier. POCUS-CA provides useful clues to:
- Differentiates true vs. pseudo-PEA
- Distinguishes asystole from VF
- Identifying reversible causes of PEA (explained above).
- It employs POCUS synchronized with CPR using a rational “holistic” approach. It always starts with cardiac imaging (subxiphoid window) and proceed further as described earlier. POCUS-CA provides useful clues to:
- POCUS-CA is a protocol to consider in management of PEA arrest.
- Point-of-care testing
Dynamic approach
Since the management of cardiac arrest is a dynamic process and there is no solid evidence for use of synchronized POCUS in cardiac arrest, it is difficult to provide an algorithmic approach to PEA. The dynamic approach to management of PEA depends on multiple factors including
- Local resources and ultrasound provider skills
- Assume at least 4 people in this approach. For teams <4 people, priorities are high quality chest compressions, IV access, POCUS pulse checks, ventilation and oxygenation.
- Patients clinical characteristics
- Other diagnostic clues obtained from EMS and collateral (e.g. risk factors for myocardial infarction or pulmonary embolism, severe fluid loss, risk of metabolic derangement).
- Be prepared before the PEA arrest patient arrives in the ED.
- Call for two dedicated people to perform chest compressions, preferably with in-line feedback or a metronome set to 110 bpm.
- Airway gear to the bedside including bougie, LMA and cric kit and call for a dedicated, skilled airway person if available.
- Turn on/gel up the POCUS machine and call for a dedicated, skilled user if available.
- Draw up “push dose epinephrine” and prepare a norepinephrine infusion to be started as the patient arrives running in the background at 10-50 micrograms/min (more on this below).
- Oxygen saturation, ETCO2, arterial line gear at the bedside and call for a dedicated person to place the arterial line if available
Distinguish True PEA from Pseudo PEA.
- ⏳First pause in chest compressions: Check the rhythm and perform cardiac POCUS.
- In the presence of perfusion-capable rhythm, ask yourself: 🤔Are there actual organized cardiac contractions (cardiac POCUS)?
- If cardiac standstill, then this is true PEA (the outcomes are poor).
- Note that since there are no contractions, pulse check is not required.
- Management should be proceeded as per ACLS guidelines for PEA arrest.
- If organized cardiac contractions are present, then this is called pseudo-PEA.
- To further guide the management, ask yourself: 🤔Are these contractions sufficient enough to generate a pulse?
- If yes, then it might be reasonable to go further and perform POCUS pulse check (algorithm below).
- If no, continue chest compression and start femoral arterial line placement.
- To further guide the management, ask yourself: 🤔Are these contractions sufficient enough to generate a pulse?
- If cardiac standstill, then this is true PEA (the outcomes are poor).
- In the presence of perfusion-capable rhythm, ask yourself: 🤔Are there actual organized cardiac contractions (cardiac POCUS)?
- ⏳Second pause in chest compression
-
- Check the rhythm and A-line BP; or (oxygen saturation waveform and ETCO2, if A-line not placed yet).
- Cardiac POCUS
- If first cardiac POCUS was indeterminate, for example in extremely bradycardic patients, cardiac contractions may be missed during a 5 sec record. Get a slightly longer cardiac POCUS within the subsequent pause.
- Lung sliding view (while the patient is being ventilated).
- 🤔Ask yourself “does the patient have a pulse from those contractions to generate a sufficient BP?
- If evidence of sufficient perfusion pressure is present, stop chest compression, and manage as profound shock state.
- Sufficient BP to provide perfusion is present if:
- A-Line DBP >40 mm Hg, or
- Persistent oxygen saturation waveform and ETCO2>40
- Sufficient BP to provide perfusion is present if:
- If evidence of sufficient perfusion pressure is absent, then continue chest compression.
- If evidence of sufficient perfusion pressure is present, stop chest compression, and manage as profound shock state.
-
- ⏳Third pause in chest compression
-
- Check rhythm
- Check A-line BP?
- Pleural and abdominal view (note that views used in eFAST does not require synchronization with CPR).
-
- If pseudo-PEA is suspected, the general management of patients usually changes from cardiac arrest management to profound shock management:
- Start arterial line placement as an ultimate monitoring tool to guide the resuscitation.
- If you determine that pseudo-PEA is present or if the patient has already received 3 doses of epinephrine in the field, rather than administering 1mg IV epinephrine, give volume and a titratable pressor: norepinephrine, vasopressin or push dose epinephrine (see below).
- Perform RUSH exam to find and treat the identifiable cause(s).
- Norepineprhine infusion
- Get your norepinephrine drip ready before before the patient arrives, so that you can start it as the patient arrives or as needed.
- Dosage: Start at 50mcg and titrating down to 20-30mcg/min as needed.
- Epinephrine (push dose pressor)
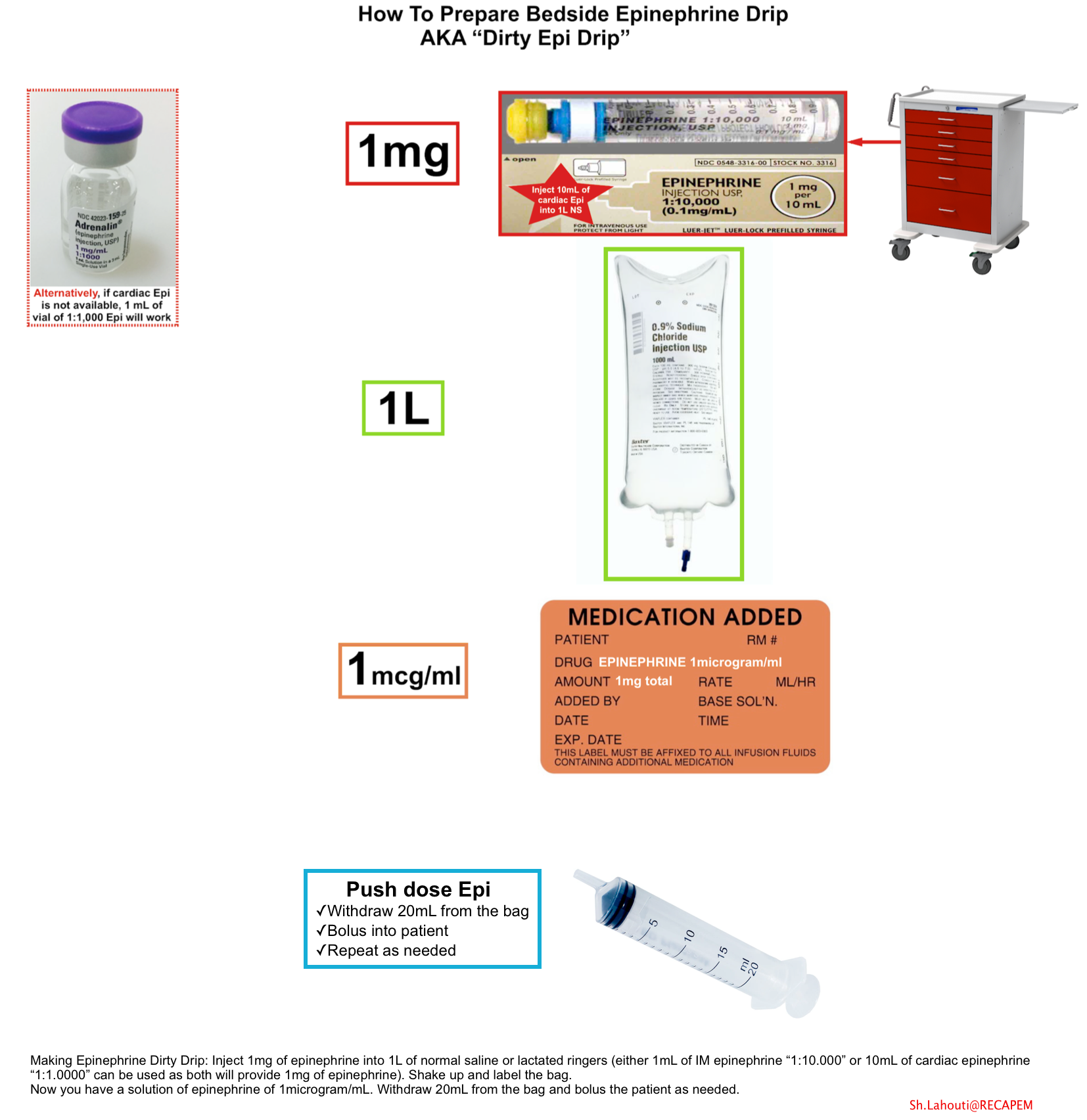
- If a pre-mixed bag of epinephrine isn’t immediately available, one may be created as follows (below figure):
- Inject 1mg epinephrine (either 10 mL of cardiac epinephrine “1:10.000” or a 1mL vial of “1:1.000” epinephrine) into a 1-liter bag of normal saline or lactated ringers.
- Either form of epinephrine will work perfectly here, since both contain a total of 1 mg of epinephrine.
- Inject 1mg epinephrine (either 10 mL of cardiac epinephrine “1:10.000” or a 1mL vial of “1:1.000” epinephrine) into a 1-liter bag of normal saline or lactated ringers.
- Shake it up. This creates a 1 mcg/ml solution of epinephrine. Figure below.
- Push dose epinephrine: 20mcg
- Fill up an empty 20 cc syringe with diluted (1 mcg/ml) epinephrine from your one-liter bag.
- Bolus the patient with 20 ml of this solution, which will deliver a bolus of 20 mcg epinephrine.
- Refill your 20 ml syringe and repeat as needed.
- If a pre-mixed bag of epinephrine isn’t immediately available, one may be created as follows (below figure):
- Vasopressin: Give vasopressin 20 IU as a bridge to getting your norepinephrine drip infusion *.
- Every effort should be made to determine reversible cause(s) of PEA and treatment should be directed accordingly.
- Tamponade
- In the patient with cardiac arrest presenting signs compatible with cardiac tamponade ultrasound-guided pericardiocentesis can be performed emergently (more on this👉 here).
- Tension pneumothorax
- Needle thoracostomy on the involved side will immediately relieve the pressure that obstructs venous return. A chest tube can then be placed to correct the pneumothorax at a more leisurely pace. More on the management of pneumothorax in ED,👉 here.
- Needle thoracostomy on the involved side will immediately relieve the pressure that obstructs venous return. A chest tube can then be placed to correct the pneumothorax at a more leisurely pace. More on the management of pneumothorax in ED,👉 here.
- Massive PE in cardiac arrest
- Systemic thrombolysis should be considered. Alteplase 50mg IV push over 2min. May repeat the dose after 15 min of CPR if ROSC is not established (more on this👉 here).
- Acute myocardial infarction (AMI) in cardiac arrest
- Systemic thrombolysis may be considered. Alteplase 50mg IV push over 2min. May repeat the dose after 15 min of CPR if ROSC is not established (more on this👉 here).
- Crashing patients on mechanical ventilation: see👉 here.
- Hyperkalemia in cardiac arrest or peri-arrest
- Push 1 amp (1 gram) of 10% calcium chloride through a large bore well-running peripheral IV or central line (preferable). Repeat as needed to achieve QRS <100ms and p waves re-appear.
- Sodium bicarbonate if acidosis
- Epinephrine: has the potential to treat hyperkalemia, bradycardia, and maintain coronary arterial perfusion pressure.
- Intra-arrest dialysis. More on this👉 here.
- Tamponade
Appendix
Adult cardiac arrest algorithm
ECG approach to underlying cause of PEA
Logistic of team-based resuscitation
Media
Cardiac standstill
Going further
- POCUS in ACLS
- POCUS in ACLS algorithm
- All PEA is not the same(REBELEM)
- Beyond ACLS: POCUS in cardiac arrest (REBELEM)
- Management of PEA: in need of resuscitation?
- PEA Arrest, PseudoPEA and PREM (With Simard and Weingart)
- VF during CPR (radiopaedia)
- 30 Year Old with Cardiac Arrest, PEA, then Cardiac Ultrasound (Dr.Smith’s Blog)
References
1.PMID: 1729058. Paradis NA, Martin GB, Goetting MG, Rivers EP, Feingold M, Nowak RM. Aortic pressure during human cardiac arrest. Identification of pseudo-electromechanical dissociation. Chest. 1992 Jan;101(1):123-8. doi: 10.1378/chest.101.1.123.
2. PMID: 11098941. Dick WF, Eberle B, Wisser G, Schneider T. The carotid pulse check revisited: what if there is no pulse? Crit Care Med. 2000 Nov;28(11 Suppl):N183-5. doi: 10.1097/00003246-200011001-00002.
3. PMID: 31740090. Rabjohns J, Quan T, Boniface K, Pourmand A. Pseudo-pulseless electrical activity in the emergency department, an evidence based approach. Am J Emerg Med. 2020 Feb;38(2):371-375. doi: 10.1016/j.ajem.2019.158503. Epub 2019 Oct 14.
4. PMID: 29364925. Wu C, Zheng Z, Jiang L, Gao Y, Xu J, Jin X, Chen Q, Zhang M. The predictive value of bedside ultrasound to restore spontaneous circulation in patients with pulseless electrical activity: A systematic review and meta-analysis. PLoS One. 2018 Jan 24;13(1):e0191636. doi: 10.1371/journal.pone.0191636.
5. PMID: 20970286. Mehta C, Brady W. Pulseless electrical activity in cardiac arrest: electrocardiographic presentations and management considerations based on the electrocardiogram. Am J Emerg Med. 2012 Jan;30(1):236-9. doi: 10.1016/j.ajem.2010.08.017. Epub 2010 Oct 20.
6. PMID: 10924315. Herlitz J, Andersson E, Bång A, Engdahl J, Holmberg M, lindqvist J, Karlson BW, Waagstein L. Experiences from treatment of out-of-hospital cardiac arrest during 17 years in Göteborg. Eur Heart J. 2000 Aug;21(15):1251-8. doi: 10.1053/euhj.2000.2150.
7. PMID: 11719169. Engdahl J, Bång A, Lindqvist J, Herlitz J. Factors affecting short- and long-term prognosis among 1069 patients with out-of-hospital cardiac arrest and pulseless electrical activity. Resuscitation. 2001 Oct;51(1):17-25. doi: 10.1016/s0300-9572(01)00377-x.
8. PMID: 11114451. Parish DC, Dane FC, Montgomery M, Wynn LJ, Durham MD, Brown TD. Resuscitation in the hospital: relationship of year and rhythm to outcome. Resuscitation. 2000 Nov;47(3):219-29. doi: 10.1016/s0300-9572(00)00231-8.
9. PMID: 18216597. Desbiens NA. Simplifying the diagnosis and management of pulseless electrical activity in adults: a qualitative review. Crit Care Med. 2008 Feb;36(2):391-6. doi: 10.1097/CCM.0b013e318161f504.
10. PMID: 16314375. ECC Committee, Subcommittees and Task Forces of the American Heart Association. 2005 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2005 Dec 13;112(24 Suppl):IV1-203. doi: 10.1161/CIRCULATIONAHA.105.166550. Epub 2005 Nov 28.
11. PMID: 23949188. Littmann L, Bustin DJ, Haley MW. A simplified and structured teaching tool for the evaluation and management of pulseless electrical activity. Med Princ Pract. 2014;23(1):1-6. doi: 10.1159/000354195. Epub 2013 Aug 13.
12. PMID: 27143124. Bergum D, Skjeflo GW, Nordseth T, Mjølstad OC, Haugen BO, Skogvoll E, Loennechen JP. ECG patterns in early pulseless electrical activity-Associations with aetiology and survival of in-hospital cardiac arrest. Resuscitation. 2016 Jul;104:34-9. doi: 10.1016/j.resuscitation.2016.03.029. Epub 2016 Apr 30.
13. PMID: 29269162. Long B, Alerhand S, Maliel K, Koyfman A. Echocardiography in cardiac arrest: An emergency medicine review. Am J Emerg Med. 2018 Mar;36(3):488-493. doi: 10.1016/j.ajem.2017.12.031. Epub 2017 Dec 16.
14. PMID: 34855015. Ávila-Reyes D, Acevedo-Cardona AO, Gómez-González JF, Echeverry-Piedrahita DR, Aguirre-Flórez M, Giraldo-Diaconeasa A. Point-of-care ultrasound in cardiorespiratory arrest (POCUS-CA): narrative review article. Ultrasound J. 2021 Dec 2;13(1):46. doi: 10.1186/s13089-021-00248-0.
15. PMID: 31003817. Simard RD, Unger AG, Betz M, Wu A, Chenkin J. The POCUS Pulse Check: A Case Series on a Novel Method for Determining the Presence of a Pulse Using Point-of-Care Ultrasound. J Emerg Med. 2019 Jun;56(6):674-679. doi: 10.1016/j.jemermed.2019.02.013. Epub 2019 Apr 16.
16. PMID: 22809840. Hogan TS. External cardiac compression may be harmful in some scenarios of pulseless electrical activity. Med Hypotheses. 2012 Oct;79(4):445-7. doi: 10.1016/j.mehy.2012.06.018. Epub 2012 Jul 17.
17. PMID: 20926019. Prosen G, Križmarić M, Završnik J, Grmec S. Impact of modified treatment in echocardiographically confirmed pseudo-pulseless electrical activity in out-of-hospital cardiac arrest patients with constant end-tidal carbon dioxide pressure during compression pauses. J Int Med Res. 2010 Jul-Aug;38(4):1458-67. doi: 10.1177/147323001003800428.





Add comment