8 July 2023 by Shahriar Lahouti. Last Update 14 September 2025.
CONTENTS
- Preface
- Etiology
- Pathophysiology
- Diagnosis
- Transthoracic echocardiography
- Approach to Management in ED
- Definitive treatment
- Other considerations
- Non-rheumatic calcific MS
- Multiple and Mixed Valvular Heart Disease
- Appendix
- Media
- Going further
- References
Preface
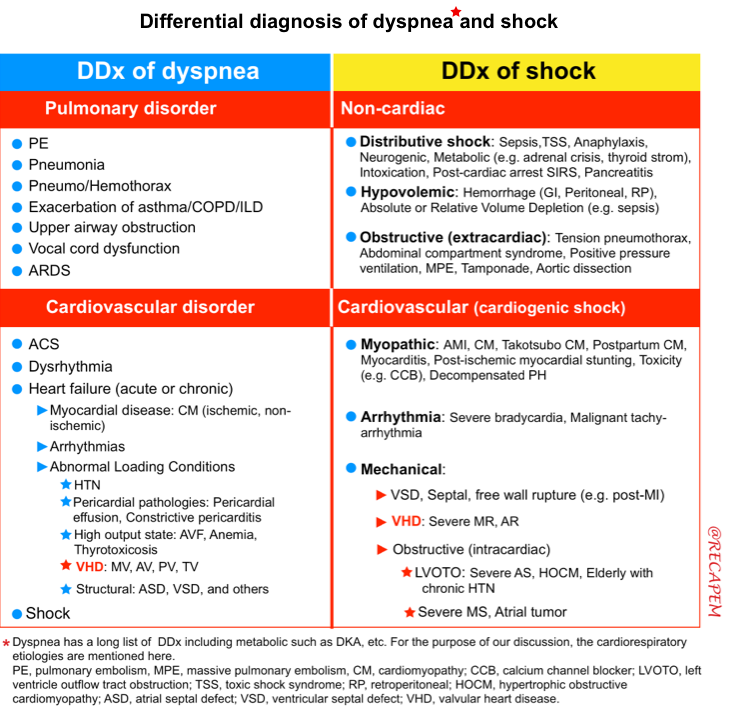
Valvular emergencies include acute deterioration of a stable lesion or the development of a new critical lesion. Severe mitral stenosis (MS) can produce heart failure and cardiogenic shock despite preserved systolic function that may not respond to standard medical therapy and typically necessitate surgical or percutaneous intervention. Diagnosis of critical valvular emergencies requires a high index of suspicion. These are often overlooked in clinical practice because the clinical history may mimic other disease entities, such as pneumonia or adult respiratory distress syndrome (ARDS) etc. Employment of bedside ultrasound and echocardiography is crucially important and can shift the diagnostic paradigm.
Structural (valvular) heart disease lesions can be broadly separated into stenotic lesions (AS, MS, LVOTO) and regurgitant lesions (AR, MR, VSD). In this post, rheumatic mitral stenosis is discussed.
Case scenario
A 48-year-old man with a history of COPD presents complaining of feeling short of breath and a productive cough…
- He had smoked 30 cigarettes per day for 25 years and had a family history of ischemic heart disease, but had no cardiac history.
- Vital signs are HR 105 beats/min; BP 100/75 mm Hg; RR 25 breaths/min; Temp 37.4°C; and SO2 91% on room air.
- Following initial treatment as per the COPD protocol, his clinical condition deteriorated within 2h. As you enter the room, you note the patient has an increased work of breathing. Despite receiving supplemental oxygen and medications, her oxygen saturation trended down to 87%.
- Lung ultrasound shows a bilateral B-profile.
- The bedside echo is shown below.
- What would be the best way to manage this patient?
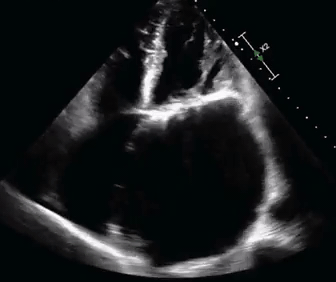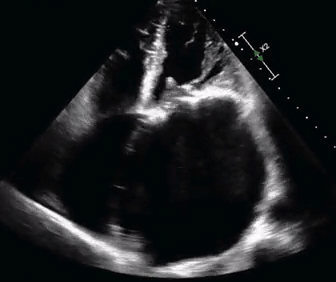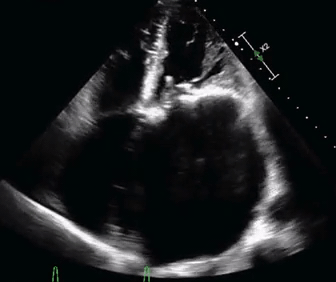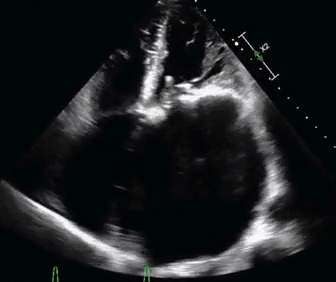
Etiology
- Rheumatic heart disease
- Mitral annular calcification (MAC). More on this below.
- Infective endocarditis
- Prosthetic valve thrombosis
- Other rare conditions
- Congenital mitral stenosis
- Radiation-associated valve disease
- Fabry disease, Whipple disease, mucopolysaccharidosis
- Carcinoid valve disease
- Endomyocardial fibrosis
- Systemic rheumatic diseases such as systemic lupus erythematosus and rheumatoid arthritis.
💡Note that almost all cases of mitral stenosis are primary MS (i.e., MS is caused by pathologies that affect the mitral valve apparatus).
Pathophysiology
The normal mitral valve orifice has a cross-sectional area of 4 to 6 cm2. Under normal physiologic conditions, the pressure in the left atrium and the left ventricle during diastole is equal (~5 mmHg). The mitral valve opens during left ventricular diastole to allow blood to flow from the left atrium to the left ventricle. More on the anatomy of MV, here.
- The left ventricle is filled with blood during early ventricular diastole.
- Only a small amount of blood remains in the left atrium. This small amount of blood fills the left ventricle with the contraction of the left atrium (the “atrial kick”) during late ventricular diastole.
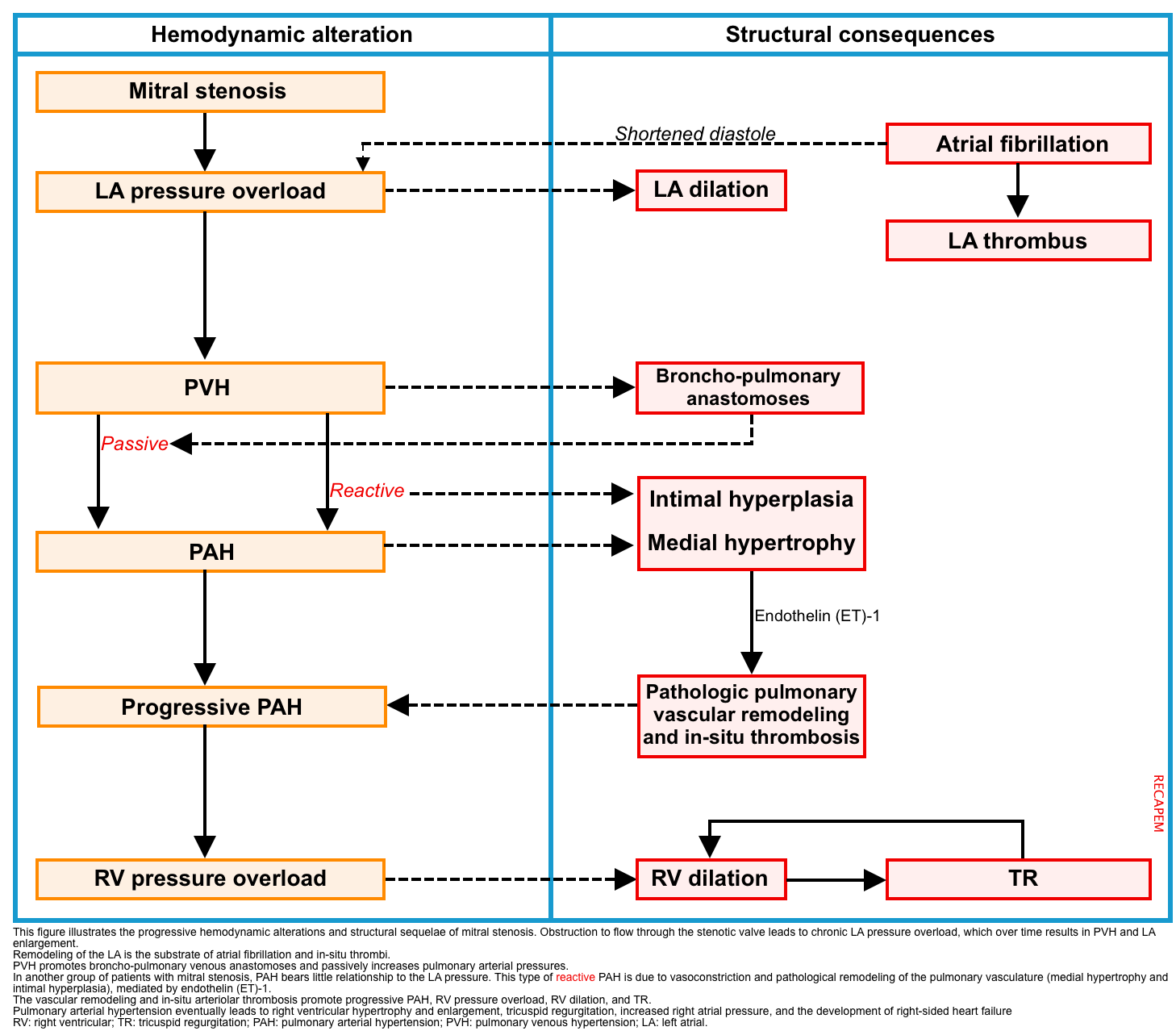
Progressive MS is associated with a gradual loss of cross-sectional valve area and leads to increased left atrial pressure. This has several hemodynamic and structural consequences (figure below), including:
- Left atrial enlargement
- Chronic pressure overload causes left atrial enlargement with an increased risk of atrial fibrillation and in situ thrombosis.
- In MS, the diastolic pressure gradient across the MV is increased. Therefore, the left ventricle requires the atrial kick to fill with blood. If atrial fibrillation develops, the atrial kick is lost and LV diastolic filling is reduced, leading to reductions in stroke volume and cardiac output.
- Chronic pressure overload causes left atrial enlargement with an increased risk of atrial fibrillation and in situ thrombosis.
- Pulmonary hypertension (PH)
- By and large, pulmonary hypertension in patients with MS is the direct consequence of chronically elevated pulmonary venous pressure *.
- Two subtypes of PH have been defined *:
- Post-capillary PH
- PVH promotes broncho-pulmonary venous anastomoses and passively increases pulmonary arterial pressures. In these patients, the pulmonary arteriovenous pressure gradient remains near normal *.
- Mean pulmonary artery pressure >20 mmHg.
- Pulmonary artery occlusion pressure of >15 mmHg.
- Diastolic pressure gradient (diastolic pulmonary artery pressure – pulmonary artery occlusion pressure) of < 7 mmHg.
- Pulmonary vascular resistance ≤3 Wood units.
- PVH promotes broncho-pulmonary venous anastomoses and passively increases pulmonary arterial pressures. In these patients, the pulmonary arteriovenous pressure gradient remains near normal *.
- Combined pre- and post-capillary PH
- This type of pulmonary hypertension is due to vasoconstriction and pathological remodeling of the pulmonary vasculature mediated by the potent vasoconstrictor endothelin-1 (ET-1) *. Hence, it is called reactive PH.
- In this relatively small group of patients with MS, pulmonary hypertension is of a different magnitude and bears little relationship to the LA pressure *.
- Post-capillary PH
Diagnosis
Diagnosis pitfalls
- Many patients with severe MS do not recognize symptoms because the slow progression of the disease is accompanied by a gradual reduction in activity and generalized deconditioning.
- Most patients often have other comorbidities, e.g., COPD and IHD, and the clinical manifestation of MS is mistakenly attributed to these comorbidities.
- For example, the acute onset of dyspnea in a patient with COPD and undiagnosed MS may be attributed to COPD exacerbation, while critical MS is the culprit.
- The most common clinical presentation of MS is dyspnea, which has a broad differential diagnosis.
| 💡Tips When patients present with complaints suspicious for acute coronary syndromes, tachycardia, heart failure, or COPD, it is important to consider a valvular cause. |
Clinical Manifestation
◾️Triggers of symptoms
- Any situation that increases the cardiac output (which raises transmitral flow) or causes tachycardia (which decreases diastolic filling time) can precipitate signs and symptoms of MS. Precipitants of MS include:
- AF
- LV diastolic filling in patients with MS is dependent on the atrial kick. In AF, loss of atrial kick leads to further underfilling of LV (↓↓cardiac output) and elevation of left atrial pressure (LAP).
- Exertion, emotional stress
- Fever, infection (e.g., pneumonia, UTI)
- Pregnancy
- AF
◾️Signs and symptoms
- Dyspnea
- Progressive exertional dyspnea is the most common symptom.
- In some patients, the initial clinical presentation is acute dyspnea with hypoxic respiratory failure due to pulmonary edema precipitated by AF with rapid ventricular response.
- Atrial fibrillation
- AF occurs commonly in MS, affecting ~ 40% of all patients *.
- AF often begins in paroxysms and eventually becomes chronic.
- Embolic events, which may be the presenting complaint in 20% of cases, are most often cerebral and leave sequelae in 25% of cases.
- Hemoptysis
- Right heart failure
- Signs and symptoms of right heart failure usually develop at more advanced stages and may include:
- Peripheral edema
- Ascites & hydrothorax
- Congestive hepatomegaly
- Signs and symptoms of right heart failure usually develop at more advanced stages and may include:
- Fever due to infective endocarditis
Differential diagnosis
Physicians should include acute valvular disease in the differential diagnosis of any patient presenting with cardiopulmonary decompensation and consider bedside echocardiography promptly to exclude or confirm this possibility.
Diagnostic evaluation
◾️Bedside US/echo
- Diagnosis of MS is based on echocardiography (discussed below).
- Bedside US/echo also plays a key role in the evaluation of patients with suspected cardiopulmonary disease and helps to narrow down the differential diagnosis.
◾️ECG
- The most important finding on ECG in patients with MS is the presence of AF.
- Other findings on the surface ECG are nonspecific and may include
- P-mitral: a P wave that becomes broader (duration in lead II >0.12 s), is of increased amplitude, and is notched (due to the delay in left atrial activation). There is also a prominent negative terminal portion of the P wave in lead V1.
- This results from left atrial hypertrophy and enlargement
- ECG changes secondary to the development of pulmonary hypertension (PH) and right ventricular hypertrophy.
- The frontal axis shifts to the right (S>R in lead I and aVL), and a tall R wave develops in V1 and V2 (R>S or R/S ratio >1).
- P pulmonale: A P wave with an amplitude of > 2.5 mm in the inferior leads (II, III, and AVF), > 1.5 mm in V1 and V2
- P-mitral: a P wave that becomes broader (duration in lead II >0.12 s), is of increased amplitude, and is notched (due to the delay in left atrial activation). There is also a prominent negative terminal portion of the P wave in lead V1.
- Note that the QRS amplitude and morphology are normal unless there is MR or coexistent aortic valve disease.
◾️Labs
- Requested based on suspected diagnosis and also to evaluate the possible triggers or comorbidities
- CBC, electrolytes including Ca/Mg/Phos.
- Troponin only if ACS is suspected.
- Lactate level.
- Liver function tests (marked transaminase elevation suggests shocked liver with poor cardiac output).
- Brain natriuretic peptide (BNP) levels are unhelpful (cardiopulmonary ultrasonography is a superior test).
Transthoracic echocardiography (TTE)
Echocardiography plays a key role in the assessment of patients with cardiopulmonary signs and symptoms. The echocardiographic approach to the evaluation of mitral stenosis is discussed here (summary table 👉appendix)
| Objectives ✅ Diagnosis: Assessment of valve anatomy and CFD ✅ Evaluation of severity ✅ Assessment of structural and hemodynamic consequences ✅ Associated lesions |
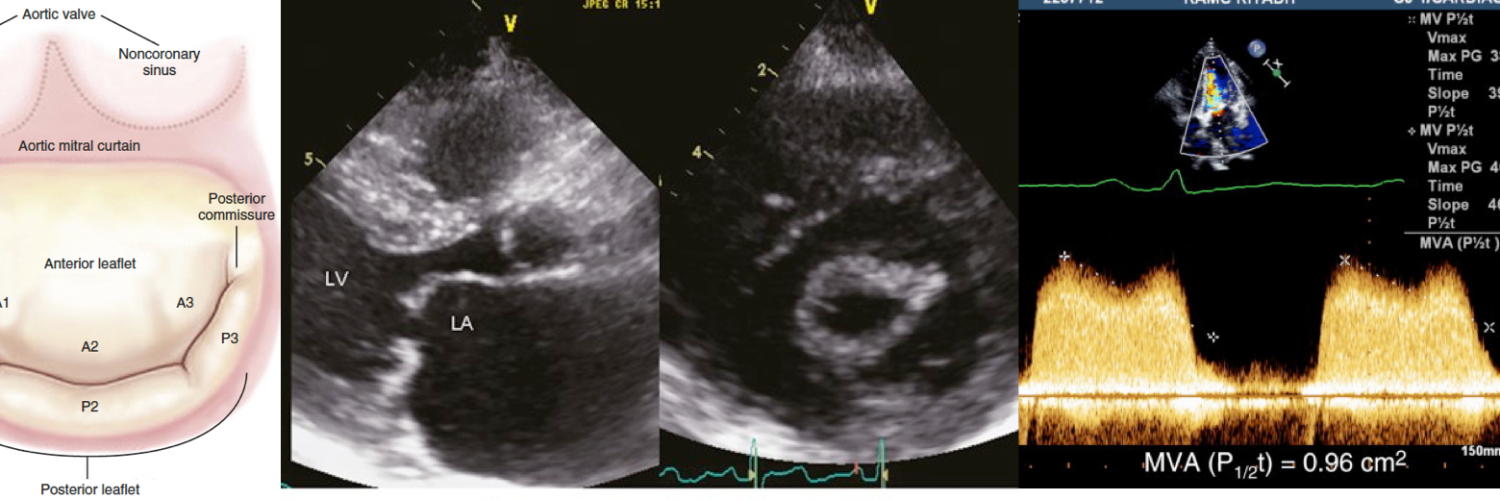
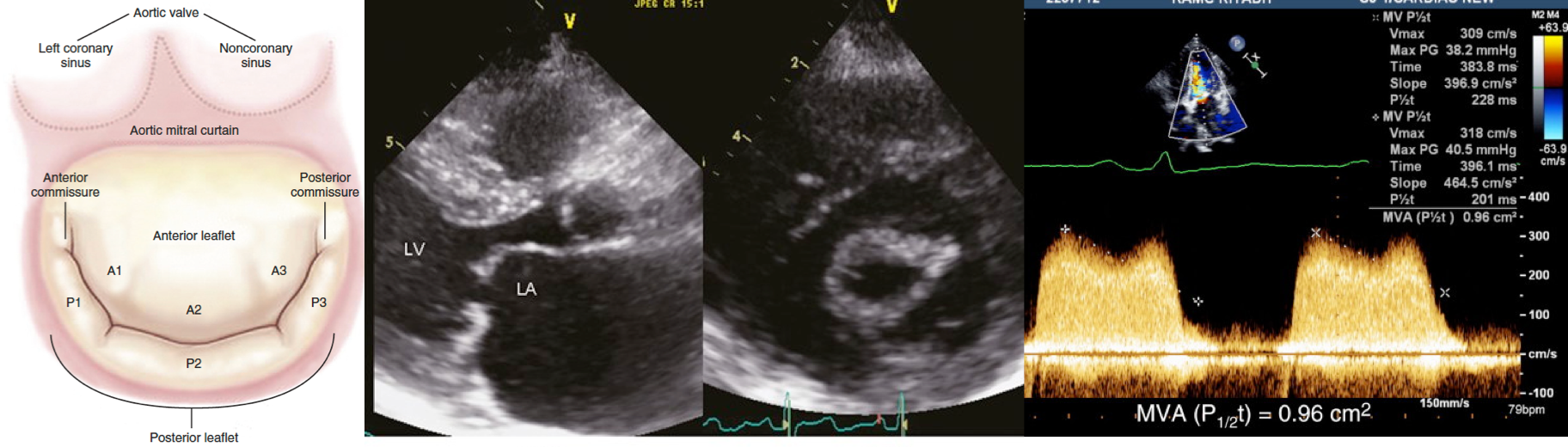
The diagnosis of rheumatic MS relies mainly on 2D echo, which shows leaflet thickening and restrictive motion (decreased mobility), commissural fusion, and involvement
of the subvalvular apparatus with thickened, fused, and shortened chordae. The parasternal short-axis view is of particular importance in assessing commissural fusion and the location of leaflet thickening or calcification
- Leaflet mobility and shape of leaflets
- Use parasternal long axis (PLAX) or apical 4-chamber (A4C) view.
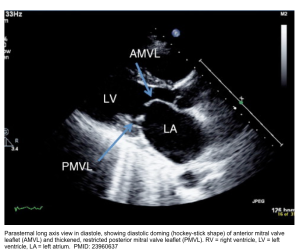
- MS alters the appearance of the valve on a 2D echo. The elevated left atrial pressure keeps the stenotic diastolic orifice opened to its maximum and causes the entire valve to dome or bulge into the ventricle throughout the diastole (🎥 clip below).
- In rheumatic MS, the anterior mitral leaflet shows diastolic doming or hockey-stick shape, and the posterior mitral leaflet has restricted motion or is totally immobile. This doming is due to the reduced mobility of the valve tips compared to the base of the leaflets (figure below).
- When the leaflet is calcified or severely thickened and therefore “stiff”, it will demonstrate mild or even no doming. Since leaflet thickening progresses with age, doming is less frequently encountered in the elderly.
- Leaflet thickening
- Use the parasternal short axis (PSAX) view.
- Leaflet thickening is considered significant if it is ≥ 5 mm *.
- Leaflet calcification
- It is suspected in the presence of increased echo brightness.
- Only acoustic shadowing is specific to calcification alone, whereas localized brightness can also be due to fibrosis.
- Use the parasternal short axis (PSAX) to assess the extent of calcification (figure here).
- The grading system for MAC is discussed here.
- It is suspected in the presence of increased echo brightness.
- The extent of commissural fusion
- Use parasternal short axis (PSAX) view.
- The assessment of commissural fusion is important to differentiate rheumatic MS from other causes, in particular degenerative calcific MS.
- Complete fusion of both commissures generally indicates severe rheumatic MS (🎥clip below).
- Conversely, the lack of commissural fusion does not exclude significant MS due to other causes or rheumatic MS in the case of restenosis due to valve rigidity with persistent commissural opening.

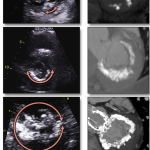
Apical long-axis view from a 2D echo shows thickened mitral valve leaflets, which have marked limitation of mobility and show doming, with failure to open normally during diastole.
Leaflet shape in rheumatic heart disease
Echographic analysis of mitral stenosis. Transthoracic echocardiography. Parasternal long-axis (left) and short-axis (right) views showing MS. Note on the short-axis view the bilateral commissural fusion without calcification.
◾️Color Doppler
- In the presence of 2D echocardiographic features of MS, Color Doppler can confirm the diagnosis of MS.
-
The diastolic flow acceleration across the MV in mitral stenosis is illustrated by a high-velocity narrow “jet” across a stenotic mitral valve during diastole into the left ventricle. Color flow aliasing and the formation of a proximal isovelocity surface area at the valve proximal to flow are shown below.

2D TTE, apical 2-chamber view without (left) and with (right) color flow Doppler, demonstrates mitral annular and leaflet calcification as well as restricted leaflet motion with diastolic flow acceleration across the MV exemplified by color flow aliasing and the formation of a proximal isovelocity surface area at the valve proximal to flow. PMID: 37325463
Severity of mitral stenosis
The assessment of the severity of MS is based on the estimation of valve area. Different techniques can be used to assess mitral valve area, e.g. planimetry, proximal isovelocity surface area (PISA).
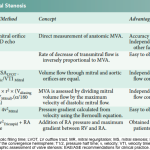
Planimetry of the mitral orifice
- This is the only direct measurement of valve area and correlates closely with anatomic findings (media 👉here).
- If MVA ≤ 1.5 cm2 mitral stenosis is considered severe.
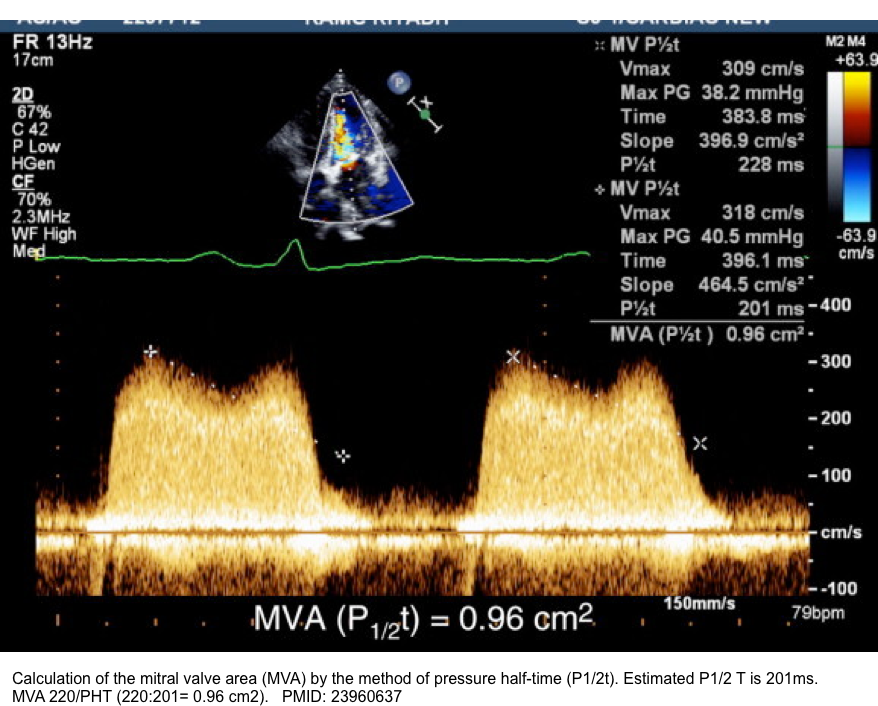
Pressure half-time method
- Pressure half-time is defined as the time (in milliseconds) that max pressure takes to half in early diastole (the gradient is half the maximum initial value) *.
- A high number means that the pressure never equilibrates between the LA and LV. A low number implies that the equilibration of pressures happens fast.
- Use CWD, apical four-chamber view.
- Estimate MV area by the formula: MV area = 220/PHT
- If PHT ≥150 ms, mitral stenosis is considered severe *.
- Note that pressure half-time is a load-dependent measure and should not be used as the single method for assessing MS severity.
| MS is defined severe if * 1. MVA ≤ 1.5 cm2 (direct planimetry of mitral orifice) 2. Diastolic pressure half time (PHT) ≥150 ms 3. Severe LAE and pulmonary artery systolic pressure >50 mmHg are usually accompanied with the above first two indices. 💡Note that MVA can be roughly estimated by 220/PHT |
👉Indirect methods to identify the severity of MS include noting the degree of:
- Left atrial enlargement (LAE).
- LV underloading (i.e., reduced left ventricle internal diameter in diastole, “LVIDD).
- Tricuspid regurgitation (TR) and pulmonary hypertension.
- Right ventricular and right atrial dilatation (RVE, RAE).
- Extent of leaflet calcification.
Structural and hemodynamic consequences
1.Left atrial size
- LA enlargement in mitral stenosis is due to chronic pressure overload.
- Method
- LA anterior-posterior (AP) diameter
- Eyeball assessment
- In the parasternal long axis view (PLAX), LA is estimated to be enlarged if it is larger than the aortic root.
- 2-D measurement using PLAX view (figure below)
- Measurements should be performed during end-LV systole with perpendicular orientation to the LA.
- M-mode: A diameter of < 50 mm and a ratio left atrium/aortic root of < 1.3 are considered normal. LA enlargement could be quantified as
- Mild: 51-60 mm
- Moderate: 61-70 mm
- Severe: >70 mm
- Eyeball assessment
- Volumetric methods
- These are the preferred methods to quantify left atrial size because they also account for variations in the shape of the LA.
- Left atrial volume is a strong predictor of events.
- There is evidence that patients with LA volumes >34 ml per m2 are at high risk for ischemic stroke, heart failure, AF, and death *.
- There are two different principles with which volumes can be obtained: The area-length method and the method of discs (Simpson method)
- LA anterior-posterior (AP) diameter
2.Mean mitral gradient
- Use CWD, apical 4-chamber view.
- The sample volume of the pulsed wave Doppler should be placed at the level of or just after the leaflet tips.
- The gradient can be measured by tracing the dense outline of mitral diastolic inflow, and the mean pressure gradient is automatically calculated.
- With more severe MS, the slower decline in transmitral flow velocity reflects the very slow decrease in left atrial pressure and slow increase in left ventricular diastolic pressure that occurs in mitral stenosis.
- In the case of atrial fibrillation, the mean gradient should be averaged over five cycles as close as possible to the normal heart rate (figure below).
- The severity can be assessed as mild (<5), moderate (5–10), and severe (>10) *.
👉Note that the gradient is not a reliable marker of the severity of valve stenosis, because it is highly dependent on heart rate and cardiac output. However, it gives information on the consequences of MS, because it is the main determinant of the increase in upstream pressures in the left atrium (LA) and pulmonary circulation.
3.Systolic pulmonary artery pressure
- This can be measured from tricuspid regurgitation velocity by the Bernoulli equation (more on this here).
- The severity can be assessed as mild (<30 mm Hg), moderate (30-50 mm Hg), and severe (>50 mm Hg) *.
Associated lesions
- Mitral regurgitation (MR)
- The detection and quantification of associated mitral regurgitation have important implications for the choice of intervention.
- The presence of MR does not alter the validity of the quantitation of MS (except in relation to the continuity equation).
- Aortic regurgitation (AR)
- The presence of AR results in a more rapid increase in LV pressure (during diastole) than would occur due to transmitral flow alone, thereby providing a misleadingly short PHT that will overestimate the MVA.
- Aortic stenosis (AS)
- LV enlargement and systolic dysfunction
- 🟠These are unusual in MS, and the initial step should be to search for an associated valvular regurgitation or coronary artery disease.
Approach to Management in ED
Clinical presentation of patients with MS can vary within a range of spectrum from exertional dyspnea to severe heart failure and cardiogenic shock.
Accurate diagnosis and appropriate hemodynamic assessment are significantly important since they have therapeutic implications.
- Caveats
- Evaluation might be challenging in critically ill patients with valvulopathy for the following reasons:
- Presence of multiple valvular diseases
- Extreme loading condition
- Determining whether the valvulopathy is the only factor responsible for a patient’s clinical deterioration may be challenging, since many patients have significant cardiac co-morbid conditions, including coronary artery disease, atrial fibrillation, and other valvular lesions, which may compromise a previously stable hemodynamic situation *.
- Evaluation might be challenging in critically ill patients with valvulopathy for the following reasons:
- Helpful clues
- Identifying the underlying etiology of valvulopathy is extremely helpful in determining the line of treatment (below table) *
- In acute conditions (e.g., acute prosthetic valve thrombosis, complicated endocarditis), the valvular problem is often at the forefront of clinical instability.
- Urgent surgery, with or without mechanical support, remains the treatment of choice in these high-risk situations.
- Conversely, in chronic situations (degenerative, rheumatic, and congenital diseases), instability is usually the result of a precipitating factor (e.g., AF) or as a result of a delayed diagnosis.
- These factors (coronary artery disease, AF) always require clinical insight for prompt management, including coronary revascularization or electrical cardioversion when deemed appropriate.
- In acute conditions (e.g., acute prosthetic valve thrombosis, complicated endocarditis), the valvular problem is often at the forefront of clinical instability.
- Identifying the underlying etiology of valvulopathy is extremely helpful in determining the line of treatment (below table) *
| Clinical context | Mechanisms | Therapeutic approaches: Heart Team evaluation |
| Acute setting | Endocarditis with new or worsening mitral/aortic valvulopathy Prosthetic valve failure Aortic dissection with severe AR Papillary muscle rupture with massive MR Acute prosthetic valve thrombosis | Coronary revascularization, restoration of sinus rhythm, etc. Surgical or percutaneous repair (e.g. PMBC for MS), w/wo temporary circulatory support |
| Chronic setting | The causes of instability are: 1. Delayed diagnosis of valvulopathy (chronic progressive) 2. Concomitant co-morbid conditions (AF/CAD, etc.) 3. Combined valvulopathies | Coronary revascularization, restoration of sinus rhythm, etc. Surgical or percutaneous repair (e.g., PMBC for MS), w/wo temporary circulatory support |
TCS: temporary circulatory support; PMBC, percutaneous mitral balloon commissurotomy.
Cardiogenic shock (CS)
Pathophysiologic background
- Mitral stenosis produces delayed and impaired LV filling (obstruction to LV filling). While the left atrial pressure is elevated, the LVEDP is low.
- A rapid HR will leave inadequate diastolic filling time and exacerbate hemodynamic compromise in the setting of MS.
- This is markedly exacerbated by loss of the atrial contribution to ventricular filling in AF, which is a common cause of deterioration among patients with chronic MS.
- Having said that, the main problem in MS is inadequate LV preload. Note that the LV systolic function is typically normal.
Basic principles
- Medical therapy
- Patients with critical valvular disease usually fail to respond to medical therapy alone *.
- The goal of medical therapy and mechanical circulatory support (MCS) is stabilization before definitive correction of the lesion.
- Generally, medical stabilization before definitive intervention differs substantially between stenotic lesions (e.g., mitral stenosis, LVOTO) and regurgitant lesions (aortic regurgitation, mitral regurgitation). See table below *.
- The effect of positive pressure ventilation (PPV) among patients with valvular emergencies is variable (table below).
- Mechanical circulatory device (MCS)
- When MCS is indicated, early application is preferred to reduce the need for vasopressors and inotropes, which may have unfavorable effects on cardiac loading conditions.
- Possible options include: aortic balloon pumps, percutaneous centrifugal pumps, and full veno-arterial ECMO
- Consultation and referral
- Cardiac surgeon consultation is warranted early for valvular emergencies.
- By and large, novel percutaneous interventions may be effective for selected patients. Therefore, consider referral to a comprehensive valve center with proficiency in both surgical and percutaneous interventions for valvular heart diseases *.
- Hemodynamic monitoring
- Patients with valvular emergencies (especially multiple or mixed valvular lesions) often have a very narrow window for hemodynamic optimization, warranting close monitoring *.
- Hemodynamic assessment can generally be made non-invasively (bedside ultrasound/echo/doppler, A-line BP monitoring, physical exams).
- Pulmonary arterial catheterization (PAC) might be considered for critical valvular emergencies if:
- Multiple or mixed valvulae lesions
- Unresponsiveness to initial therapy
- Poor transthoracic echocardiographic windows and inability to perform a transesophageal echocardiogram
Treatments for Decompensated Heart Failure and Cardiogenic Shock Due to Valvular Heart Disease Emergencies *
Critical MS with Low Blood Pressure (Wet and Cold)
- Control rhythm
- Control HR
- Goal: Resting HR of 60 bpm for patients who are not in AF *.
- Agents: Digoxin or amiodarone
- Protect MAP
- Hypotension requires prompt treatment to defend the coronary and end-organ perfusion.
- Agents: Vasopressin or phenylephrine *
- Intubation
- PPV has a risk of hypotension and cardiac arrest.
- If intubation is required, resuscitate (raise the MAP), then intubate.
Critical MS with Normal BP (Wet and Warm)
- Control rhythm
- AF (esp with rapid ventricular response) should be cardioverted *.
- Control HR
- Goal: Resting HR of 60 bpm for patients who are not in AF
- Agents: Beta-blocker (e.g., esmolol)*
- Volume
- Diuretics in patients with pulmonary edema are beneficial *.
- Intubation
- PPV has a risk of hypotension and cardiac arrest.
- If intubation is required, resuscitate (raise the MAP), then intubate.
Definitive treatment
- Urgent balloon mitral valvuloplasty (BMV), if feasible in critically ill patients, may be considered as the first-line therapy *.
- For patients who have contraindications for BMV (heavy calcification, concomitant MR), transcatheter mitral valve implantation should be considered *.
Symptomatic mitral stenosis
In hemodynamically stable patients, medical therapy may be helpful for symptomatic relief.
- Diuretics
- Indicated when there are manifestations of pulmonary vascular congestion, such as exertional shortness of breath, orthopnea, or, at a later stage of MS, when right heart failure can occur, with hepatic congestion and peripheral edema.
- Reducing the heart rate (in patients with sinus rhythm or AF)
- Beta-blockers, nondihydropyridine calcium channel blockers (verapamil or diltiazem), and ivabradine.
- Digoxin may help control the rapid ventricular rate during AF in patients with left and/or right ventricular systolic dysfunction.
- Management of atrial fibrillation
- A rate control strategy is preferred (rhythm control may not be feasible as MS progresses) *.
- However, conversion to sinus rhythm may be necessary if the heart rate cannot be adequately controlled with medications.
- In patients not requiring urgent cardioversion, the decision for rate control versus rhythm control is based upon multiple factors, including the duration of AF, hemodynamic response to AF, symptoms, and left atrial size *.
- Prevention of thromboembolism (TE)
Definitive treatment
Since MS is a mechanical disorder, once valve obstruction is present, only intervention provides sustained symptom control and improved survival. Therapeutic interventions include:
- Percutaneous mitral balloon commissurotomy (PMBC)
- Surgery: Surgical repair, commissurotomy, or valve replacement
Decisions on whether and how to intervene are largely based on the following criteria:
- The stage of MS
- The stages of MS are defined by patient symptoms, valve anatomy, valve hemodynamics, and the consequences of valve obstruction on the left atrium (LA) and pulmonary circulation * (figure below).
- Feasibility and risk of intervention (PMBC or mitral surgery).
- Appropriate candidates for PBMC are
- Less than moderate MR on TEE
- Favorable valve morphology
- No left atrial thrombus
- No prior failed PMBC
- Appropriate candidates for PBMC are
- Whether the patient is undergoing cardiac surgery for a concurrent condition (e.g., coronary artery disease).
Indications for intervention in patients with rheumatic MS
- Severe MS (MVA ≤ 1.5 cm2) who are either
- Symptomatic (stage D), or
- Asymptomatic (stage C) with elevated pulmonary artery systolic pressure (PASP; >50 mmHg) or AF.
- Nonsevere progressive MS (MVA > 1.5 cm2 , stage B) who have either
- Isolated nonsevere MS with exertional symptoms and evidence of hemodynamically significant MS with exercise (i.e., pulmonary artery wedge pressure >25 mmHg or mean mitral valve gradient >15 mmHg).
- Mixed nonsevere MS and moderate MR who are symptomatic. In these patients, surgical mitral valve replacement is considered.
Other consideration
Secondary prevention of rheumatic fever
- Patients with rheumatic MS should receive antibiotic prophylaxis for the secondary prevention of rheumatic fever.
Prevention of infective endocarditis
- Only patients with the highest risk of the development of endocarditis (e.g., patients with prosthetic heart valves, patients with prior endocarditis) are advised to receive antimicrobial prophylaxis *.
- Most patients with native valvular heart disease, including those with MS, are not included in this group and therefore do not require antimicrobial prophylaxis
Non-rheumatic calcific MS
Mitral annular calcification (MAC) is generally an incidental finding associated with aging (🎥 clip below), although it is occasionally prominent enough to cause mitral stenosis, mitral regurgitation, or both (mixed disease) *.
- The prevalence of MAC in the general population is estimated to be around 10%.
- Risk factors for MAC:

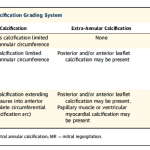
Posterior mitral annular calcification without restricted mitral leaflet and not a stenotic lesion
◾️Calcific MS (aka degenerative MS) occurs via progression of this calcification along the mitral annulus, causing degeneration of the fibrous mitral support *.
- Calcific MS is found with increasing frequency in the elderly population in high-income countries.
- ~8% of patients with MAC are found to have MS.
- Calcific MS is the result of calcification of the mitral annulus that extends into the leaflet bases, resulting in both the narrowing of the annulus and rigidity of the leaflets.
- 👉In contrast to rheumatic MS, there is no commissural fusion, and the leaflet tips are usually unaffected *.
- Determination of the severity of stenosis is difficult because of extensive calcification, which prevents measurement of an accurate planimetered area, and the significant abnormalities of LA and LV compliance, which cause a high gradient in the absence of severe obstruction.
- These patients are at high risk with any intervention because of the extensive calcification, as well as advanced age and multiple comorbidities.
- Thus, in patients with calcific MS, the indications for any intervention differ from those for rheumatic MS, and intervention for calcific MS should be performed only in highly symptomatic patients *.
Multiple and Mixed Valvular Heart Disease
- Multiple VHD refers to the combination of stenotic or regurgitant lesions occurring on ≥2 cardiac valves.
- Mixed VHD refers to the combination of stenotic and regurgitant lesions on the same valve *.
- The hemodynamic and clinical consequences of any given valvular lesion may be modulated by the concomitant presence of mixed or multiple VHD. These hemodynamic interactions can promote, exacerbate, or, in contrast, blunt the clinical expression of each singular lesion *.
- Conditions that reduce the effective LV forward flow in patients with MS:
- Increasing left atrial pressure: Concomitant MR
- ↑Afterload (e.g., Hypertension, Aortic stenosis)
- ↓Inotrope
- Tachycardia
- Hypotension →↓Coronary perfusion pressure→ Myocardial ischemia
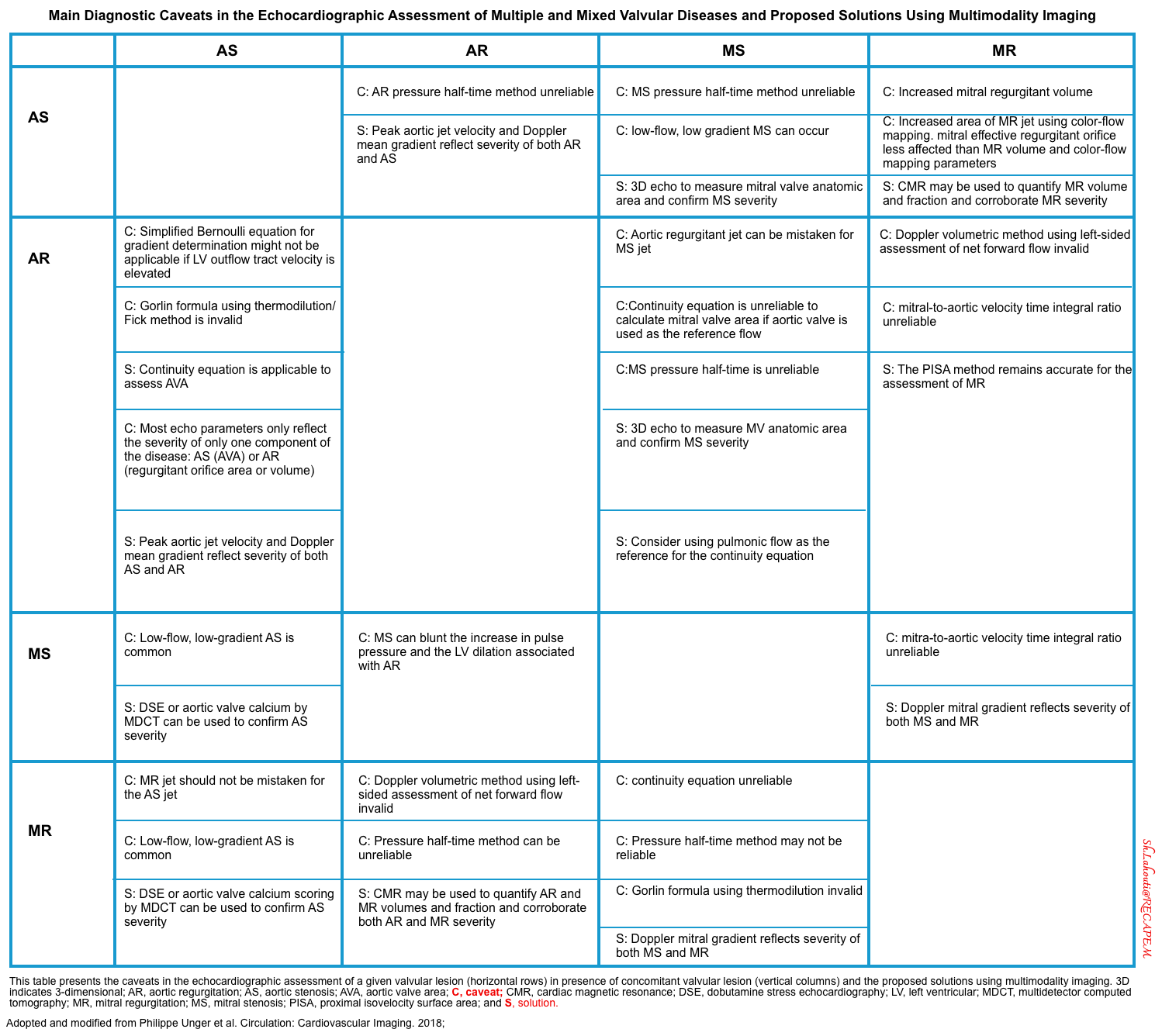
- The main diagnostic caveats in the echocardiographic assessment of multiple and mixed valvular diseases are summarized in the following table.
Appendix
Proposed grading of mitral annular calcification
Approach to the evaluation of mitral stenosis
Media
Echocardiographic assessment of the mitral valve
Going further
- Mitral stenosis (show me the POCUS)
- Managing acute cardiac valvular emergencies in the ED
- Reference (normal) values for echocardiography
References
1. PMID: 29876357. Maeder MT, Weber L, Buser M, Gerhard M, Haager PK, Maisano F, Rickli H. Pulmonary Hypertension in Aortic and Mitral Valve Disease. Front Cardiovasc Med. 2018 May 23;5:40. doi: 10.3389/fcvm.2018.00040.
2. PMID: 30545968. Simonneau G, Montani D, Celermajer DS, Denton CP, Gatzoulis MA, Krowka M, Williams PG, Souza R. Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur Respir J. 2019 Jan 24;53(1):1801913. doi: 10.1183/13993003.01913-2018.
3. PMID: 29876357. Maeder MT, Weber L, Buser M, Gerhard M, Haager PK, Maisano F, Rickli H. Pulmonary Hypertension in Aortic and Mitral Valve Disease. Front Cardiovasc Med. 2018 May 23;5:40. doi: 10.3389/fcvm.2018.00040.
4. PMID: 16027271. Carabello BA. Modern management of mitral stenosis. Circulation. 2005 Jul 19;112(3):432-7. doi: 10.1161/CIRCULATIONAHA.104.532498.
5. PMID: 19130998. Baumgartner H, Hung J, Bermejo J, Chambers JB, Evangelista A, Griffin BP, Iung B, Otto CM, Pellikka PA, Quiñones M; American Society of Echocardiography; European Association of Echocardiography. Echocardiographic assessment of valve stenosis: EAE/ASE recommendations for clinical practice. J Am Soc Echocardiogr. 2009 Jan;22(1):1-23; quiz 101-2. doi: 10.1016/j.echo.2008.11.029. Erratum in: J Am Soc Echocardiogr. 2009 May;22(5):442. Erratum in: J Am Soc Echocardiogr. 2023 Apr;36(4):445.
6. PMID: 23960637. Omran AS, Arifi AA, Mohamed AA. Echocardiography in mitral stenosis. J Saudi Heart Assoc. 2011 Jan;23(1):51-8. doi: 10.1016/j.jsha.2010.07.007. Epub 2010 Sep 7.
7. PMID: 31492536. Akodad M, Schurtz G, Adda J, Leclercq F, Roubille F. Management of valvulopathies with acute severe heart failure and cardiogenic shock. Arch Cardiovasc Dis. 2019 Dec;112(12):773-780. doi: 10.1016/j.acvd.2019.06.009. Epub 2019 Sep 3.
8. PMID: 32314662. Jentzer JC, Ternus B, Eleid M, Rihal C. Structural Heart Disease Emergencies. J Intensive Care Med. 2021 Sep;36(9):975-988. doi: 10.1177/0885066620918776. Epub 2020 Apr 21.
9. PMID: 35912478. Bernard S, Deferm S, Bertrand PB. Acute valvular emergencies. Eur Heart J Acute Cardiovasc Care. 2022 Aug 9;11(8):653-665. doi: 10.1093/ehjacc/zuac086.
10. PMID: 33332149. Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP 3rd, Gentile F, Jneid H, Krieger EV, Mack M, McLeod C, O’Gara PT, Rigolin VH, Sundt TM 3rd, Thompson A, Toly C. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021 Feb 2;143(5):e35-e71. doi: 10.1161/CIR.0000000000000932. Epub 2020 Dec 17. Erratum in: Circulation. 2021 Feb 2;143(5):e228. Erratum in: Circulation. 2021 Mar 9;143(10):e784.
11. PMID: 27832900. Eleid MF, Foley TA, Said SM, Pislaru SV, Rihal CS. Severe Mitral Annular Calcification: Multimodality Imaging for Therapeutic Strategies and Interventions. JACC Cardiovasc Imaging. 2016 Nov;9(11):1318-1337. doi: 10.1016/j.jcmg.2016.09.001.
12. PMID: 33061148. Van Hemelrijck M, Taramasso M, Gülmez G, Maisano F, Mestres CA. Mitral annular calcification: challenges and future perspectives. Indian J Thorac Cardiovasc Surg. 2020 Jul;36(4):397-403. doi: 10.1007/s12055-019-00910-2. Epub 2020 Jan 27.
13. PMID: 16647710. Sharma R, Pellerin D, Gaze DC, Mehta RL, Gregson H, Streather CP, Collinson PO, Brecker SJ. Mitral annular calcification predicts mortality and coronary artery disease in end stage renal disease. Atherosclerosis. 2007 Apr;191(2):348-54. doi: 10.1016/j.atherosclerosis.2006.03.033. Epub 2006 May 2.
14. PMID: 37325463. Lamberg M, Devine A, Jewulski J, Smith A, Garster N, Sharma A, Mason PJ, Lewandowski D, Widlansky M, Mohananey D. Degenerative Mitral Stenosis: A Case-Based Review. CASE (Phila). 2023 Mar 13;7(5):189-196. doi: 10.1016/j.case.2022.12.014.












Add comment